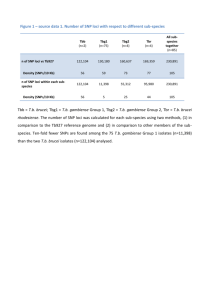
GIANT_BMI_2013_Main_Text_Revision.041014
advertisement

1 LARGE-SCALE GENETIC STUDIES OF BODY MASS INDEX PROVIDE INSIGHT 2 INTO THE BIOLOGICAL BASIS OF OBESITY 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 37 38 39 40 41 42 43 44 45 46 47 48 Adam E Locke1,*, Bratati Kahali2,*, Sonja I Berndt3,*, Anne E Justice4,*, Tune H Pers5,6,7,8,*, Felix R Day9, Corey Powell2, Sailaja Vedantam5,6, Martin L Buchkovich10, Jian Yang11,12, Damien C Croteau-Chonka10,13, Tonu Esko5,6,7,14, Tove Fall15,16, Teresa Ferreira17, Stefan Gustafsson15, Zoltan Kutálik18,19,20, Jian'an Luan9, Reedik Mägi14,17, Joshua C Randall17,21, Thomas W Winkler22, Andrew R Wood23, Tsegaselassie Workalemahu24, Jessica D Faul25, Jennifer A Smith26, Jing Hua Zhao9, Wei Zhao26, Jin Chen27, Rudolf Fehrmann28, Åsa K Hedman15,17, Juha Karjalainen28, Ellen M Schmidt29, Devin Absher30, Najaf Amin31, Denise Anderson32, Marian Beekman33,34, Jennifer L Bolton35, Jennifer L BraggGresham1, Steven Buyske36,37, Ayse Demirkan31,38, Guohong Deng39,40,41, Georg B Ehret42,43, Bjarke Feenstra44, Mary F Feitosa45, Krista Fischer14, Anuj Goel17,46, Jian Gong47, Anne U Jackson1, Stavroula Kanoni48, Marcus E Kleber49,50, Kati Kristiansson51, Unhee Lim52, Vaneet Lotay53, Massimo Mangino54, Irene Mateo Leach55, Carolina Medina-Gomez56,57,58, Sarah E Medland59, Michael A Nalls60, Cameron D Palmer5,6, Dorota Pasko23, Sonali Pechlivanis61, Marjolein J Peters56,58, Inga Prokopenko17,62,63, Dmitry Shungin64,65,66, Alena Stančáková67, Rona J Strawbridge68, Yun Ju Sung69, Toshiko Tanaka70, Alexander Teumer71, Stella Trompet72,73, Sander W van der Laan74, Jessica van Setten75, Jana V Van Vliet-Ostaptchouk76, Zhaoming Wang77,78, Loïc Yengo79,80,81, Weihua Zhang39,82, Aaron Isaacs31,83, Eva Albrecht84, Johan Ärnlöv15,85, Gillian M Arscott86, Antony P Attwood87,88, Stefania Bandinelli89, Amy Barrett62, Isabelita N Bas90, Claire Bellis91, Amanda J Bennett62, Christian Berne92, Roza Blagieva93, Matthias Blüher94,95, Stefan Böhringer33,96, Lori L Bonnycastle97, Yvonne Böttcher94, Heather A Boyd44, Marcel Bruinenberg98, Ida H Caspersen99, Jin Chen27, Robert Clarke100, E Warwick Daw45, Anton JM de Craen73, Graciela Delgado de Moissl49, Josh C Denny101, Maria Dimitriou102, Alex SF Doney103, Niina Eklund51,104, Karol Estrada6,58,105, Elodie Eury79,80,81, Lasse Folkersen68, Ross M Fraser35, Melissa E Garcia106, Frank Geller44, Vilmantas Giedraitis107, Bruna Gigante108, Alan S Go109, Alain Golay110, Alison H Goodall111,112, Scott D Gordon59, Mathias Gorski22,113, Hans-Jörgen Grabe114,115, Harald Grallert116, Tanja B Grammer49, Jürgen Gräßler117, Henrik Grönberg16, Christopher J Groves62, Gaëlle Gusto118, Jeffrey Haessler47, Per Hall16, Toomas Haller14, Goran Hallmans119, Catharina A Hartman120, Maija Hassinen121, Caroline Hayward122, Nancy L Heard-Costa123,124, Quinta Helmer33,96,125, Christian Hengstenberg126,127, Oddgeir Holmen128, Jouke-Jan Hottenga129, Alan L James130,131, Janina M. Jeff53, Åsa Johansson132, Jennifer Jolley87,88, Thorhildur Juliusdottir17, Abel N Kho133, Leena Kinnunen51, Wolfgang Koenig50, Markku Koskenvuo134, Wolfgang Kratzer135, Jaana Laitinen136, Claudia Lamina137, Karin Leander108, Nanette R Lee90, Peter Lichtner138, Lars Lind139, Jaana Lindström51, Ken Sin Lo140, Stéphane Lobbens79,80,81, Roberto Lorbeer141, Yingchang Lu53,142, François Mach43, Patrik KE Magnusson16, Anubha Mahajan17, Wendy L McArdle143, Stela McLachlan35, Cristina Menni54, Sigrun Merger93, Evelin Mihailov14,144, Lili Milani14, Alireza Moayyeri54, Keri L Monda4,145, Mario A Morken97, Antonella Mulas146, Gabriele Müller147, Martina Müller-Nurasyid84,148,149, Arthur W Musk150, Ramaiah Nagaraja151, Markus M Nöthen152,153, Ilja M Nolte154, Stefan Pilz155,156, Nigel W Rayner17,21,62, Frida Renstrom64, Rainer Rettig157, Janina S Ried84, Stephan Ripke105,158, Neil R Robertson17,62, Lynda M Rose159, Serena Sanna146, Hubert Scharnagl160, Salome Scholtens154, Fredrick R Schumacher161, William R Scott39,82, Thomas Seufferlein135, Jianxin Shi162, Albert Vernon Smith163,164, Joanna Smolonska28,154, Alice V Stanton165, Page 1 of 68 49 50 51 52 53 54 55 56 57 58 59 60 61 62 63 64 65 66 67 68 69 70 71 72 73 74 75 76 77 78 79 80 81 82 83 84 85 86 87 88 89 90 91 92 93 94 95 96 97 98 99 Valgerdur Steinthorsdottir166, Kathleen Stirrups21,48, Heather M Stringham1, Johan Sundström139, Morris A Swertz28, Amy J Swift97, Ann-Christine Syvänen15, Sian-Tsung Tan39,167, Bamidele O Tayo168, Barbara Thorand169, Gudmar Thorleifsson166, Jonathan P Tyrer170, Hae-Won Uh33,96, Floor VA van Oort171, Liesbeth Vandenput172, Sita H Vermeulen173,174, Niek Verweij55, Judith M Vonk154, Lindsay L Waite30, Helen R Warren175, Dawn Waterworth176, Michael N Weedon23, Lynne R Wilkens52, Christina Willenborg177,178, Tom Wilsgaard179, Mary K Wojczynski45, Andrew Wong180, Alan F Wright122, Qunyuan Zhang45, The LifeLines Cohort Study181,182, Eoin P. Brennan183, Murim Choi184, Zari Dastani185, Alexander W Drong17, Per Eriksson68, Anders FrancoCereceda186, Jesper Gådin68, Ali G Gharavi187, Michael E Goddard188,189, Robert E Handsaker6,7, Jinyan Huang190, Fredrik Karpe62,191, Sekar Kathiresan6,192, Sarah Keildson17, Krzysztof Kiryluk187, Michiaki Kubo193, Jong-Young Lee194, Liming Liang190,195, Richard P Lifton196, Baoshan Ma190,197, Steven A McCarroll6,7,158, Amy J McKnight198, Josine L Min143, Miriam F Moffatt167, Grant W Montgomery59, Joanne M Murabito123,199, George Nicholson200,201, Dale R Nyholt59, Yukinori Okada202,203, John RB Perry17,23,54, Rajkumar Dorajoo204, Eva Reinmaa14, Rany M Salem5,6,7, Niina Sandholm205,206,207, Robert A Scott9, Lisette Stolk33,58, Atsushi Takahashi202, Toshihiro Tanaka203,208,209, Ferdinand M van ´t Hooft68, Anna AE Vinkhuyzen11, Harm-Jan Westra28, Wei Zheng210, Krina T Zondervan17,211, The ADIPOGen Consortium182,212, The AGEN-BMI Working Group182,213, The CARDIOGRAMplusC4D Consortium, The CKDGen Consortium, The eMERGE Consortium214, The GLGC215, The ICBP182,216, The MAGIC Investigators217, The MuTHER Consortium182,218, The MIGen Consortium182,219, The PAGE Consortium182,220, The ReproGen Consortium, The GENIE Consortium182,221, The International Endogene Consortium182, Andrew C Heath222, Dominique Arveiler223, Stephan JL Bakker224, John Beilby86,225, Richard N Bergman226, John Blangero91, Pascal Bovet227,228, Harry Campbell35, Mark J Caulfield175, Giancarlo Cesana229, Aravinda Chakravarti42, Daniel I Chasman159,230, Peter S Chines97, Francis S Collins97, Dana C Crawford231,232, L Adrienne Cupples233, Daniele Cusi234,235, John Danesh236, Ulf de Faire108, Hester M den Ruijter74,237, Anna F Dominiczak238, Raimund Erbel239, Jeanette Erdmann177,178, Johan G Eriksson51,240,241, Martin Farrall17,46, Stephan B Felix77,242, Ele Ferrannini243,244, Jean Ferrières245, Ian Ford246, Nita G Forouhi9, Terrence Forrester247, Oscar H Franco56,57, Ron T Gansevoort224, Pablo V Gejman248, Christian Gieger84, Omri Gottesman53, Vilmundur Gudnason163,164, Ulf Gyllensten132, Alistair S Hall249, Tamara B Harris106, Andrew T Hattersley250, Andrew A Hicks251,252, Lucia A Hindorff253, Aroon D Hingorani254, Albert Hofman56,57, Georg Homuth71, G Kees Hovingh255, Steve E Humphries256, Steven C Hunt257, Elina Hypponen258,259,260, Thomas Illig116,261, Kevin B Jacobs3,262, Marjo-Riitta Jarvelin82,263,264,265,266,267, Karl-Heinz Jöckel61, Berit Johansen99, Pekka Jousilahti51, J Wouter Jukema72,268,269, Antti M Jula51, Jaakko Kaprio51,104,134, John JP Kastelein255, Sirkka M Keinanen-Kiukaanniemi270,271, Lambertus A Kiemeney173,272, Paul Knekt51, Jaspal S Kooner39,167,273, Charles Kooperberg47, Peter Kovacs94,95, Aldi T Kraja45, Meena Kumari274, Johanna Kuusisto275, Timo A Lakka121,276, Claudia Langenberg9,274, Loic Le Marchand52, Terho Lehtimäki277, Valeriya Lyssenko278, Satu Männistö51, André Marette279,280, Tara C Matise37, Colin A McKenzie247, Barbara McKnight281, Frans L Moll282, Andrew D Morris103, Andrew P Morris14,17,283, Jeffrey C Murray284, Mari Nelis14, Claes Ohlsson172, Albertine J Oldehinkel120, Ken K Ong9,180, Pamela AF Madden222, Gerard Pasterkamp74, John F Peden285, Annette Peters116,126,169, Dirkje S Postma286, Peter P Pramstaller251,252,287, Jackie F Price35, Lu Qi13,288, Olli T Raitakari289,290, Tuomo Rankinen291, DC Rao45,69,222, Treva K Rice69,222, Paul M Ridker159,230, John D Rioux140,292, Marylyn D. Ritchie293, Igor Rudan35,294, Veikko Salomaa51, Nilesh J Samani111,112, Jouko Saramies295, Mark A Sarzynski291, Heribert Schunkert126,127, Peter EH Schwarz117,296, Peter Sever297, Alan R Shuldiner298,299, Juha Page 2 of 68 100 101 102 103 104 105 106 107 108 109 110 111 112 113 114 115 116 117 118 119 120 121 122 123 124 125 126 127 128 129 130 131 132 133 134 135 136 137 138 139 140 141 142 143 144 145 146 147 148 149 150 Sinisalo300, Ronald P Stolk154, Konstantin Strauch84,149, Anke Tönjes94,95, DavidAlexandre Trégouët301,302,303,Angelo Tremblay304, Elena Tremoli305, Jarmo Virtamo51, Marie-Claude Vohl280,306, Uwe Völker71,77, Gérard Waeber307, Gonneke Willemsen129, Jacqueline C Witteman57, Wolfgang Koenig50, M Carola Zillikens56,58, Linda S Adair308, Philippe Amouyel309, Folkert W Asselbergs254,268,310 Themistocles L Assimes311, Murielle Bochud227,228, Bernhard O Boehm93,312, Eric Boerwinkle313, Stefan R Bornstein117, Erwin P Bottinger53, Claude Bouchard291, Stéphane Cauchi79,80,81, John C Chambers39,82,273, Stephen J Chanock3, Richard S Cooper168, Paul IW de Bakker75,314,315, George Dedoussis102, Luigi Ferrucci70, Paul W Franks64,65,288, Philippe Froguel63,79,80,81, Leif C Groop104,316, Christopher A Haiman161, Anders Hamsten68, M Geoffrey Hayes133, Jennie Hui86,225,258, David J. Hunter13,190,288, Kristian Hveem128, Robert C Kaplan317, Mika Kivimaki274, Diana Kuh180, Markku Laakso275, Yongmei Liu318, Nicholas G Martin59, Winfried März49,160,319, Mads Melbye44, Andres Metspalu14,144, Susanne Moebus61, Patricia B Munroe175, Inger Njølstad179, Ben A Oostra31,83,320, Colin NA Palmer103, Nancy L Pedersen16, Markus Perola14,51,104, Louis Pérusse280,304, Ulrike Peters47, Chris Power260, Thomas Quertermous311, Rainer Rauramaa121,321, Fernando Rivadeneira56,57,58, Timo E Saaristo322,323, Danish Saleheen236,324,325, Naveed Sattar326, Eric E Schadt327, David Schlessinger151, P Eline Slagboom33,34, Harold Snieder154, Tim D Spector54, Kari Stefansson166,328, Michael Stumvoll94,95, Jaakko Tuomilehto51,329,330,331, André G Uitterlinden56,57,58, Matti Uusitupa332,333, Pim van der Harst28,55,268, Mark Walker334, Henri Wallaschofski77,78, Nicholas J Wareham9, Hugh Watkins17,46, David R Weir25, H-Erich Wichmann335,336,337, James F Wilson35, Pieter Zanen338, Ingrid B Borecki45, Panos Deloukas21,48,339, Caroline S Fox123, Iris M Heid22,84, Jeffrey R O'Connell298, David P Strachan340, Unnur Thorsteinsdottir166,328, Cornelia M van Duijn31,56,57,83, Gonçalo R Abecasis1, Lude Franke28, Timothy M Frayling23, Mark I McCarthy17,62,341, Peter M Visscher11,12, André Scherag61,342, Cristen J Willer27,29,343, Michael Boehnke1, Karen L Mohlke10, Cecilia M Lindgren6,17, Jacques S. Beckmann19,20,344, Inês Barroso21,345,346, Kari E North4,347,§, Erik Ingelsson15,17,§, Joel N Hirschhorn5,6,7,§, Ruth JF Loos9,53,142,348,§, Elizabeth K Speliotes2,§ Affiliations 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. Center for Statistical Genetics, Department of Biostatistics, University of Michigan, Ann Arbor, MI 48109, USA Department of Internal Medicine, Division of Gastroenterology, and Department of Computational Medicine and Bioinformatics, University of Michigan, Ann Arbor, MI 48109 Division of Cancer Epidemiology and Genetics, National Cancer Institute, National Institutes of Health, Bethesda, MD 20892, USA Department of Epidemiology, University of North Carolina at Chapel Hill, Chapel Hill, NC 27599, USA Divisions of Endocrinology and Genetics and Center for Basic and Translational Obesity Research, Boston Children's Hospital, Boston, MA 02115, USA Broad Institute of the Massachusetts Institute of Technology and Harvard University, Cambridge 02142, MA, USA Department of Genetics, Harvard Medical School, Boston, MA 02115, USA Center for Biological Sequence Analysis, Department of Systems Biology, Technical University of Denmark, Lyngby 2800, Denmark MRC Epidemiology Unit, University of Cambridge, Institute of Metabolic Science, Addenbrooke’s Hospital, Hills Road, Cambridge, CB2 0QQ, UK Department of Genetics, University of North Carolina, Chapel Hill, NC 27599, USA Page 3 of 68 151 152 153 154 155 156 157 158 159 160 161 162 163 164 165 166 167 168 169 170 171 172 173 174 175 176 177 178 179 180 181 182 183 184 185 186 187 188 189 190 191 192 193 194 195 196 197 198 199 200 201 11. 12. 13. 14. 15. 16. 17. 18. 19. 20. 21. 22. 23. 24. 25. 26. 27. 28. 29. 30. 31. 32. 33. 34. 35. 36. 37. 38. 39. Queensland Brain Institute, The University of Queensland, Brisbane 4072, Australia The University of Queensland Diamantina Institute, The Translation Research Institute, Brisbane 4012, Australia Channing Division of Network Medicine, Department of Medicine, Brigham and Women's Hospital and Harvard Medical School, Boston, MA 02115, USA Estonian Genome Center, University of Tartu, Tartu 51010, Estonia Department of Medical Sciences, Molecular Epidemiology and Science for Life Laboratory, Uppsala University, Uppsala 75185, Sweden Department of Medical Epidemiology and Biostatistics, Karolinska Institutet, Stockholm 17177, Sweden Wellcome Trust Centre for Human Genetics, University of Oxford, Oxford OX3 7BN, UK Institute of Social and Preventive Medicine (IUMSP), Centre Hospitalier Universitaire Vaudois (CHUV), Lausanne 1010, Switzerland Swiss Institute of Bioinformatics, Lausanne 1015, Switzerland Department of Medical Genetics, University of Lausanne, Lausanne 1005, Switzerland Wellcome Trust Sanger Institute, Hinxton, Cambridge CB10 1SA, UK Department of Genetic Epidemiology, Institute of Epidemiology and Preventive Medicine, University of Regensburg, Regensburg, Germany, D-93053 Regensburg, Germany Genetics of Complex Traits, University of Exeter Medical School, University of Exeter, Exeter EX1 2LU, UK Harvard School of Public Health, Department of Nutrition, Harvard University, Boston, MA 2115, USA Survey Research Center, Institute for Social Research, University of Michigan, Ann Arbor, MI 48104, USA Department of Epidemiology, University of Michigan, Ann Arbor, MI 48109, USA Department of Internal Medicine, Division of Cardiovascular Medicine, University of Michigan, Ann Arbor, MI, USA Department of Genetics, University Medical Center Groningen, University of Groningen, 9700 RB Groningen, The Netherlands Department of Computational Medicine and Bioinformatics, University of Michigan, Ann Arbor, MI, USA HudsonAlpha Institute for Biotechnology, Huntsville, AL 35806, USA Genetic Epidemiology Unit, Department of Epidemiology, Erasmus University Medical Center, 3015 GE Rotterdam, The Netherlands Telethon Institute for Child Health Research, Centre for Child Health Research, The University of Western Australia, Western Australia 6008, Australia Netherlands Consortium for Healthy Aging (NCHA), Leiden University Medical Center, Leiden 2300 RC, The Netherlands Department of Molecular Epidemiology, Leiden University Medical Center, 2300 RC Leiden, The Netherlands Centre for Population Health Sciences, University of Edinburgh, Teviot Place, Edinburgh, EH8 9AG, Scotland, UK Department of Statistics & Biostatistics, Rutgers University, Piscataway, N.J. USA Department of Genetics, Rutgers University, Piscataway, N.J. USA. Department of Human Genetics, Leiden University Medical Center, 2333 ZC Leiden, The Netherlands Ealing Hospital NHS Trust, Middlesex UB1 3HW, UK Page 4 of 68 202 203 204 205 206 207 208 209 210 211 212 213 214 215 216 217 218 219 220 221 222 223 224 225 226 227 228 229 230 231 232 233 234 235 236 237 238 239 240 241 242 243 244 245 246 247 248 249 250 251 252 40. 41. 42. 43. 44. 45. 46. 47. 48. 49. 50. 51. 52. 53. 54. 55. 56. 57. 58. 59. 60. 61. 62. 63. 64. 65. 66. Department of Gastroenterology and Hepatology, Imperial College London, London W2 1PG, UK Institute of infectious Diseases, Southwest Hospital, Third Military Medical University, Chongqing, China Center for Complex Disease Genomics, McKusick-Nathans Institute of Genetic Medicine, Johns Hopkins University School of Medicine, Baltimore, MD 21205, USA Cardiology, Department of Specialties of Internal Medicine, Geneva University Hospital, Geneva 1211, Switzerland Department of Epidemiology Research, Statens Serum Institut, Copenhagen DK2300, Denmark Department of Genetics, Washington University School of Medicine, St. Louis, MO 63110, USA Division of Cardiovacular Medicine, Radcliffe Department of Medicine, University of Oxford, Oxford OX3 9DU, UK Division of Public Health Sciences, Fred Hutchinson Cancer Research Center, Seattle, WA 98109, USA William Harvey Research Institute, Barts and The London School of Medicine and Dentistry, Queen Mary University of London, EC1M 6BQ UK Institute of Public Health, Social and Preventive Medicine, Mannheim Medical Faculty, Heidelberg University, D-68167 Mannheim, Germany Department of Internal Medicine II, Ulm University Medical Centre, D-89081 Ulm, Germany National Institute for Health and Welfare, FI-00271 Helsinki, Finland Epidemiology Program, University of Hawaii Cancer Center, Honolulu, HI USA The Charles Bronfman Institute for Personalized Medicine, Icahn School of Medicine at Mount Sinai, New York, NY 10029, USA Department of Twin Research and Genetic Epidemiology, King’s College London, London SE1 7EH, UK Department of Cardiology, University Medical Center Groningen, University of Groningen, 9700RB Groningen, The Netherlands Netherlands Consortium for Healthy Aging (NCHA), 3015GE Rotterdam, The Netherlands Department of Epidemiology, Erasmus Medical Center, 3015GE Rotterdam, The Netherlands Department of Internal Medicine, Erasmus Medical Center, 3015GE Rotterdam, The Netherlands QIMR Berghofer Medical Research Institute, Queensland 4006, Australia Laboratory of Neurogenetics, National Institute on Aging, National Institutes of Health, Bethesda, MD 20892, USA Institute for Medical Informatics, Biometry and Epidemiology (IMIBE), University Hospital Essen, Essen, Germany Oxford Centre for Diabetes, Endocrinology and Metabolism, University of Oxford, Oxford OX3 7LJ, UK Department of Genomics of Common Disease, School of Public Health, Imperial College London, Hammersmith Hospital, London, UK Department of Clinical Sciences, Genetic & Molecular Epidemiology Unit, Lund University Diabetes Center, Skåne University Hosptial, Malmö 205 02, Sweden Department of Public Health and Clinical Medicine, Unit of Medicine, Umeå University, Umeå 901 87, Sweden Department of Odontology, Umeå University, Umeå 901 85, Sweden Page 5 of 68 253 254 255 256 257 258 259 260 261 262 263 264 265 266 267 268 269 270 271 272 273 274 275 276 277 278 279 280 281 282 283 284 285 286 287 288 289 290 291 292 293 294 295 296 297 298 299 300 301 302 67. 68. 69. 70. 71. 72. 73. 74. 75. 76. 77. 78. 79. 80. 81. 82. 83. 84. 85. 86. 87. 88. 89. 90. 91. 92. 93. 94. 95. 96. University of Eastern Finland, FI-70210 Kuopio, Finland Atherosclerosis Research Unit, Center for Molecular Medicine, Department of Medicine, Karolinska Institutet, Stockholm 17176, Sweden Division of Biostatistics, Washington University School of Medicine, St. Louis, MO 63110, USA Translational Gerontology Branch, National institute on Aging, Baltimore MD 21225, USA Interfaculty Institute for Genetics and Functional Genomics, University Medicine Greifswald, D-17475 Greifswald, Germany Department of Cardiology, Leiden University Medical Center, 2300 RC Leiden, The Netherlands Department of Gerontology and Geriatrics, Leiden University Medical Center, 2300 RC Leiden, The Netherlands Experimental Cardiology Laboratory, Division Heart and Lungs, University Medical Center Utrecht, 3584 CX Utrecht, The Netherlands Department of Medical Genetics, University Medical Center Utrecht, 3584 CX Utrecht, The Netherlands Department of Endocrinology, University of Groningen, University Medical Center Groningen, Groningen, 9700 RB, The Netherlands Deutsches Forschungszentrum für Herz-Kreislauferkrankungen (DZHK) (German Centre for Cardiovascular Research), partner site Greifswald, D-17475 Greifswald, Germany Institute of Clinical Chemistry and Laboratory Medicine, University Medicine Greifswald, D-17475 Greifswald, Germany CNRS UMR 8199, F-59019 Lille, France European Genomic Institute for Diabetes, F-59000 Lille, France Université de Lille 2, F-59000 Lille, France Department of Epidemiology and Biostatistics, Imperial College London, London W2 1PG, UK Center for Medical Sytems Biology, Leiden, The Netherlands Institute of Genetic Epidemiology, Helmholtz Zentrum München - German Research Center for Environmental Health, D-85764 Neuherberg, Germany School of Health and Social Studies, Dalarna University, Falun, Sweden PathWest Laboratory Medicine of Western Australia, NEDLANDS, Western Australia 6009, Australia Department of Haematology, University of Cambridge, Cambridge CB2 0PT, UK NHS Blood and Transplant, Cambridge CB2 0PT, UK Geriatric Unit, Azienda Sanitaria Firenze (ASF), Florence, Italy USC-Office of Population Studies Foundation, Inc., University of San Carlos, Cebu City 6000, Philippines Department of Genetics, Texas Biomedical Research Institute, San Antonio, TX, USA Department of Medical Sciences, Endocrinology, Diabetes and Metabolism, Uppsala University, Uppsala 75185, Sweden Division of Endocrinology, Diabetes and Metabolism, Ulm University Medical Centre, D-89081 Ulm, Germany IFB Adiposity Diseases, University of Leipzig, D-04103 Leipzig, Germany Department of Medicine, University of Leipzig, D-04103 Leipzig, Germany Department of Medical Statistics and Bioinformatics, Leiden University Medical Center, 2300 RC Leiden, The Netherlands Page 6 of 68 303 304 305 306 307 308 309 310 311 312 313 314 315 316 317 318 319 320 321 322 323 324 325 326 327 328 329 330 331 332 333 334 335 336 337 338 339 340 341 342 343 344 345 346 347 348 349 350 351 352 97. 98. 99. 100. 101. 102. 103. 104. 105. 106. 107. 108. 109. 110. 111. 112. 113. 114. 115. 116. 117. 118. 119. 120. 121. 122. 123. Genome Technology Branch, National Human Genome Research Institute, NIH, Bethesda, MD 20892, USA LifeLines, University Medical Center Groningen, University of Groningen, 9700 RB Groningen, The Netherlands Department of Biology, Norwegian University of Science and Technology, Trondheim, Norway Clinical Trial Service Unit and Epidemiological Studies Unit, Nuffield Department of Population Health, University of Oxford, Oxford OX3 7LF, UK Department of Biomedical Informatics, Vanderbilt University, Nashville, TN 37232, USA Department of Dietetics-Nutrition, Harokopio University, Athens, Greece Medical Research Institute, University of Dundee, Ninewells Hospital and Medical School, Dundee DD1 9SY, UK Institute for Molecular Medicine, University of Helsinki, FI-00014 Helsinki, Finland Analytic and Translational Genetics Unit, Massachusetts General Hospital and Harvard Medical School, Boston, MA, USA Laboratory of Epidemiology and Population Sciences, National Institute on Aging, NIH, Bethesda, MD 20892, USA Department of Public Health and Caring Sciences, Geriatrics, Uppsala University, Uppsala 75185, Sweden Division of Cardiovascular Epidemiology, Institute of Environmental Medicine, Karolinska Institutet, Stockholm, Sweden, Stockholm 17177, Sweden Kaiser Permanente, Division of Research, Oakland, CA 94612, USA Service of Therapeutic Education for Diabetes, Obesity and Chronic Diseases, Geneva University Hospital, Geneva CH-1211, Switzerland Department of Cardiovascular Sciences, University of Leicester, Glenfield Hospital, Leicester LE3 9QP, UK National Institute for Health Research (NIHR) Leicester Cardiovascular Disease Biomedical Research Unit, Glenfield Hospital, Leicester, LE3 9QP, UK Department of Internal Medicine II, University Medical Center Regensburg, Regensburg, Germany, D-93053 Regensburg, Germany Department of Psychiatry and Psychotherapy, University Medicine Greifswald, HELIOS-Hospital Stralsund, D-17475 Greifswald, Germany German Center for Neurodegenerative Diseases (DZNE), Rostock, Greifswald, D17475 Greifswald, Germany Research Unit of Molecular Epidemiology, Helmholtz Zentrum München - German Research Center for Environmental Health, D-85764 Neuherberg, Germany Department of Medicine III, University of Dresden, Medical Faculty Carl Gustav Carus, D-01307 Dresden, Germany Institut inter Régional pour la Santé, Synergies, F-37520 La Riche, France Department of Public Health and Primary Care, Units of Nutritional Research, Umeå University Hosptial, Umeå 90187, Sweden Department of Psychiatry, University of Groningen, University Medical Center Groningen, Groningen, The Netherlands Kuopio Research Institute of Exercise Medicine, Kuopio, Finland MRC Human Genetics Unit, Institute of Genetics and Molecular Medicine, University of Edinburgh, Western General Hospital, Edinburgh, EH4 2XU, Scotland, UK National Heart, Lung, and Blood Institute, the Framingham Heart Study, Framingham MA 01702, USA Page 7 of 68 353 354 355 356 357 358 359 360 361 362 363 364 365 366 367 368 369 370 371 372 373 374 375 376 377 378 379 380 381 382 383 384 385 386 387 388 389 390 391 392 393 394 395 396 397 398 399 400 401 402 124. Department of Neurology, Boston University School of Medicine, Boston, MA 02118, USA 125. Faculty of Psychology and Education, VU University Amsterdam, Amsterdam, The Netherlands 126. Deutsches Forschungszentrum für Herz-Kreislauferkrankungen (DZHK) (German Research Centre for Cardiovascular Research), Munich Heart Alliance, D-80636 Munich, Germany 127. Deutsches Herzzentrum München, Technische Universität München, D-80636 Munich, Germany 128. Department of Public Health and General Practice, Norwegian University of Science and Technology, Trondheim 7489, Norway 129. Biological Psychology, VU University Amsterdam, 1081BT Amsterdam, The Netherlands 130. Department of Pulmonary Physiology and Sleep Medicine, NEDLANDS, Western Australia 6009, Australia 131. School of Medicine and Pharmacology, University of Western Australia, CRAWLEY 6009, Australia 132. Uppsala University, Department of Immunology, Genetics, Pathology, SciLifeLab, Rudbeck Laboratory, SE-751 85, Uppsala, Sweden 133. Department of Medicine, Northwestern University Feinberg School of Medicine, Chicago, IL 60611, USA 134. Hjelt Institute Department of Public Health, University of Helsinki, FI-00014 Helsinki, Finland 135. Department of Internal Medicine I, Ulm University Medical Centre, D-89081 Ulm, Germany 136. Finnish Institute of Occupational Health, Oulu, Finland 137. Division of Genetic Epidemiology, Department of Medical Genetics, Molecular and Clinical Pharmacology, Innsbruck Medical University, 6020 Innsbruck, Austria 138. Institute of Human Genetics, Helmholtz Zentrum München - German Research Center for Environmental Health, D-85764 Neuherberg, Germany 139. Department of Medical Sciences, Cardiovascular Epidemiology, Uppsala University, Uppsala 75185, Sweden 140. Montreal Heart Institute, Montreal, Quebec H1T 1C8, Canada 141. Institute for Community Medicine, University Medicine Greifswald, D-17475 Greifswald, Germany 142. The Genetics of Obesity and Related Metabolic Traits Program, The Icahn School of Medicine at Mount Sinai, New York, NY 10029, USA 143. School of Social and Community Medicine, University of Bristol, Bristol BS8 2BN, UK 144. Institute of Molecular and Cell Biology, University of Tartu, Tartu 51010, Estonia 145. The Center for Observational Research, Amgen, Inc., Thousand Oaks, CA 91320, USA 146. Istituto di Ricerca Genetica e Biomedica (IRGB), Consiglio Nazionale delle Ricerche, Cagliari, Sardinia 09042, Italy 147. Institute for Medical Informatics and Biometrics, University of Dresden, Medical Faculty Carl Gustav Carus, D-01307 Dresden, Germany 148. Department of Medicine I, University Hospital Grosshadern, Ludwig-MaximiliansUniversität, D-81377 Munich, Germany 149. Institute of Medical Informatics, Biometry and Epidemiology, Chair of Genetic Epidemiology, Ludwig-Maximilians-Universität, D-85764 Neuherberg, Germany Page 8 of 68 403 404 405 406 407 408 409 410 411 412 413 414 415 416 417 418 419 420 421 422 423 424 425 426 427 428 429 430 431 432 433 434 435 436 437 438 439 440 441 442 443 444 445 446 447 448 449 450 451 452 453 150. Department of Respiratory Medicine, Sir Charles Gairdner Hospital, NEDLANDS, Western Australia 6009, Australia 151. Laboratory of Genetics, National Institute on Aging, Baltimore, MD 21224, USA 152. Department of Genomics, Life & Brain Center, University of Bonn, Bonn, Germany 153. Institute of Human Genetics, University of Bonn, Bonn, Germany 154. Department of Epidemiology, University Medical Center Groningen, University of Groningen, 9700 RB Groningen, The Netherlands 155. Department of Epidemiology and Biostatistics, EMGO Institute for Health and Care Research, VU University Medical Center, Amsterdam, The Netherlands 156. Department of Internal Medicine, Division of Endocrinology and Metabolism, Medical University of Graz, 8036 Graz, Austria 157. Institute of Physiology, University Medicine Greifswald, D-17495 Karlsburg, Germany 158. Stanley Center for Psychiatric Research, Broad Institute of MIT and Harvard, Cambridge, MA 02142, USA 159. Division of Preventive Medicine, Brigham and Women's Hospital, Boston, MA 02215, USA 160. Clinical Institute of Medical and Chemical Laboratory Diagnostics, Medical University of Graz, Graz 8036, Austria 161. Department of Preventive Medicine, Keck School of Medicine, University of Southern California, Los Angeles, CA, USA 162. National Cancer Institute, Bethesda, MD, USA 163. Icelandic Heart Association, Kopavogur 201, Iceland 164. University of Iceland, Reykjavik 101, Iceland 165. Molecular & Cellular Therapeutics, Royal College of Surgeons in Ireland, 123 St Stephens Green, Dublin 2, Ireland 166. deCODE Genetics, Amgen inc., Reykjavik 101, Iceland 167. National Heart and Lung Institute, Imperial College, London W12 0NN, UK 168. Department of Public Health Sciences, Stritch School of Medicine, Loyola University of Chicago, Maywood, IL 61053, USA 169. Institute of Epidemiology II, Helmholtz Zentrum München - German Research Center for Environmental Health, Neuherberg, Germany, D-85764 Neuherberg, Germany 170. Department of Ocology, University of Cambridge, Cambridge CB2 0QQ, UK 171. Department of Child and Adolescent Psychiatry, Psychology, Erasmus University Medical Centre, 3000 CB Rotterdam, The Netherlands 172. Department of Internal Medicine, Institute of Medicine, Sahlgrenska Academy, University of Gothenburg, Gothenburg 413 45, Sweden 173. Department for Health Evidence, Radboud University Medical Centre, 6500 HB Nijmegen, The Netherlands 174. Department of Genetics, Radboud University Medical Centre, 6500 HB Nijmegen, The Netherlands 175. Clinical Pharmacology and Barts and The London Genome Centre, William Harvey Research Institute, Barts and The London School of Medicine and Dentistry, Queen Mary University of London, Charterhouse Square, London EC1M 6BQ, UK 176. Genetics, GlaxoSmithKline, King of Prussia, PA, USA 177. Deutsches Forschungszentrum für Herz-Kreislauferkrankungen (DZHK) (German Centre for Cardiovascular Research), partner site Hamburg, Kiel, D-23562 Lübeck, Germany 178. Institut für Integrative und Experimentelle Genomik, Universität zu Lübeck, D23562 Lübeck, Germany Page 9 of 68 454 455 456 457 458 459 460 461 462 463 464 465 466 467 468 469 470 471 472 473 474 475 476 477 478 479 480 481 482 483 484 485 486 487 488 489 490 491 492 493 494 495 496 497 498 499 500 501 502 503 504 179. Department of Clinical Medicine, Faculty of Health Sciences, University of Tromsø, Tromsø, Norway 180. MRC Unit for Lifelong Health and Ageing at University College London, London WC1B 5JU, UK 181. The LifeLines Cohort Study, University of Groningen, University Medical Center Groningen, Groningen, The Netherlands 182. Membership to this consortium is provided below. 183. Diabetes Complications Research Centre, Conway Institute, School of Medicine and Medical Sciences, University College Dublin, Dublin, Ireland 184. Department of Biomedical Sciences, Seoul National University College of Medicine, Seoul, Korea 185. Lady Davis Institute, Departments of Human Genetics, Epidemiology and Biostatistics, McGill University, Montréal, Québec H3T1E2, Canada 186. Cardiothoracic Surgery Unit, Department of Molecular Medicine and Surgery, Karolinska Institutet, Stockholm 17176, Sweden 187. Department of Medicine, Columbia University College of Physicians and Surgeons, New York NY, USA 188. Biosciences Research Division, Department of Primary Industries, Victoria 3083, Australia 189. Department of Food and Agricultural Systems, University of Melbourne, Victoria 3010, Australia 190. Harvard School of Public Health, Department of Epidemiology, Harvard University, Boston, MA 2115, USA 191. NIHR Oxford Biomedical Research Centre, OUH Trust, Oxford OX3 7LE, UK 192. Cardiovascular Research Center, Massachusetts General Hospital, Harvard Medical School, Boston, MA, USA 193. Laboratory for Genotyping Development, RIKEN Center for Integrative Medical Sciences , Yokohama, Japan 194. Center for Genome Science, National Institute of Health, Chungcheongbuk-do, Chungbuk 363-951, Republic of Korea 195. Harvard School of Public Health, Department of Biostatistics, Harvard University, Boston, MA 2115, USA 196. Department of Genetics, Howard Hughes Medical Institute, Yale University School of Medicine, New Haven, New Haven CT, USA 197. College of Information Science and Technology, Dalian Maritime University, Dalian, Liaoning 116026, China 198. Nephrology Research, Centre for Public Health, Queen's University of Belfast, Belfast, Co. Down BT9 7AB, UK 199. Section of General Internal Medicine, Boston University School of Medicine, Boston, MA 2118, USA 200. Department of Statistics, University of Oxford, 1 South Parks Road, Oxford OX1 3TG, UK 201. MRC Harwell, Harwell Science and Innovation Campus, Harwell, UK 202. Laboratory for Statistical Analysis, RIKEN Center for Integrative Medical Sciences, Yokohama, Japan 203. Department of Human Genetics and Disease Diversity, Graduate School of Medical and Dental Sciences, Tokyo Medical and Dental University, Tokyo, Japan 204. Genome Institute of Singapore, Agency for Science, Technology and Research, Singapore 205. Department of Biomedical Engineering and Computational Science, Aalto University School of Science, Helsinki, Finland Page 10 of 68 505 506 507 508 509 510 511 512 513 514 515 516 517 518 519 520 521 522 523 524 525 526 527 528 529 530 531 532 533 534 535 536 537 538 539 540 541 542 543 544 545 546 547 548 549 550 551 552 553 554 555 206. Department of Medicine, Division of Nephrology, Helsinki University Central Hospital, FI-00290 Helsinki, Finland 207. Folkhälsan Institute of Genetics, Folkhälsan Research Center, FI-00290 Helsinki, Finland 208. Laboratory for Cardiovascular Diseases, RIKEN Center for Integrative Medical Sciences, Yokohama, Japan 209. Division of Disease Diversity, Bioresource Research Center, Tokyo Medical and Dental University 210. Division of Epidemiology, Department of Medicine; Vanderbilt Epidemiology Center; and Vanderbilt-Ingram Cancer Center, Vanderbilt University Medical Center, Nashville, TN 37075, USA 211. Nuffield Department of Obstetrics & Gynaecology, University of Oxford, Oxford OX3 7BN, UK 212. Adiponectin Genetic Consortium 213. The Asian Genetic Epidemiology Network - BMI Working Group 214. The electronic medical records and genomics (eMERGE) consortium 215. The Global Lipids Genetics Consortium 216. The International Consortium for Blood Pressure Genome-Wide Association Studies 217. Meta-Analyses of Glucose and Insulin-related traits Consortium Investigators 218. The Multiple Tissue Human Expression Resource Consortium 219. Myocardial Infarction Genetics (MIGen) Consortium 220. Population Architecture using Genomics and Epidemiology Consortium 221. GEnetics of Nephropathy - an International Effort Consortium 222. Department of Psychiatry, Washington University School of Medicine, St. Louis, MO 63110, USA 223. Department of Epidemiology and Public Health, EA3430, University of Strasbourg, Faculty of Medicine, Strasbourg, France 224. Department of Internal Medicine, University Medical Center Groningen, University of Groningen, 9700RB Groningen, The Netherlands 225. Pathology and Laboratory Medicine, The University of Western Australia, Western Australia 6009, Australia 226. Diabetes and Obesity Research Institute, Cedars-Sinai Medical Center, Los Angeles, California, USA 227. Institute of Social and Preventive Medicine (IUMSP), Centre Hospitalier Universitaire Vaudois and University of Lausanne, Lausanne, Switzerland 228. Ministry of Health, Victoria, Republic of Seychelles 229. University of Milano, Bicocca, Italy 230. Harvard Medical School, Boston, MA 02115, USA 231. Center for Human Genetics Research, Vanderbilt University Medical Center, Nashville TN 37203, USA 232. Department of Molecular Physiology and Biophysics, Vanderbilt University, Nashville, TN 37232, USA 233. Department of Biostatistics, Boston University School of Public Health, Boston, MA 02118, USA 234. Department of Health Sciences, University of Milano, I 20142, Italy 235. Fondazione Filarete, Milano I 20139, Italy 236. Department of Public Health and Primary Care, University of Cambridge, Cambridge, UK 237. Julius Center for Health Sciences and Primary Care, University Medical Center Utrecht, 3584 CX Utrecht, The Netherlands Page 11 of 68 556 557 558 559 560 561 562 563 564 565 566 567 568 569 570 571 572 573 574 575 576 577 578 579 580 581 582 583 584 585 586 587 588 589 590 591 592 593 594 595 596 597 598 599 600 601 602 603 604 605 606 238. Institute of Cardiovascular and Medical Sciences, Faculty of Medicine, University of Glasgow, Glasgow G12 8TA, UK 239. Clinic of Cardiology, West-German Heart Centre, University Hospital Essen, Essen, Germany 240. Department of General Practice and Primary Health Care, University of Helsinki, FI-00290 Helsinki, Finland 241. Unit of General Practice, Helsinki University Central Hospital, Helsinki 00290, Finland 242. Department of Internal Medicine B, University Medicine Greifswald, D-17475 Greifswald, Germany 243. Department of Internal Medicine, University of Pisa, Pisa, Italy 244. CNR Institute of Clinical Physiology, University of Pisa, Pisa, Italy 245. Department of Cardiology, Toulouse University School of Medicine, Rangueil Hospital, Toulouse, France 246. Robertson Center for Biostatistics, University of Glasgow, Glasgow, UK 247. Tropical Metabolism Research Unit, Tropical Medicine Research Institute, The University of the West Indies, Mona, Kingston 7, Jamaica 248. NorthShore University HealthSystem, Evanston, IL, University of Chicago, Chicago, IL, USA 249. Division of Epidemiology, Leeds Institute of Genetics, Health and Therapeutics, University of Leeds, UK 250. Institute of Biomedical & Clinical Science, University of Exeter, Barrack Road, Exeter, EX2 5DW 251. Center for Biomedicine, European Academy Bozen, Bolzano (EURAC), Bolzano 39100, Italy 252. Affiliated Institute of the University of Lübeck, D-23562 Lübeck, Germany 253. Division of Genomic Medicine, National Human Genome Research Institute, National Institutes of Health, Bethesda, MD, USA 254. Institute of Cardiovascular Science, University College London, WC1E 6BT, UK 255. Department of Vascular Medicine, Academic Medical Center, Amsterdam, The Netherlands 256. Centre for Cardiovascular Genetics, Institute Cardiovascular Sciences, University College London, London WC1E 6JJ, UK 257. Cardiovascular Genetics Division, Department of Internal Medicine, University of Utah, Salt Lake City, Utah 84108, USA 258. School of Population Health and Sansom Institute for Health Research, University of South Australia, Adelaide 5000, Australia 259. South Australian Health and Medical Research Institute, Adelaide, Australia 260. Centre for Paediatric Epidemiology and Biostatistics, UCL Institute of Child Health, London WC1N 1EH, UK 261. Hannover Unified Biobank, Hannover Medical School, Hannover, Germany, D30625 Hannover, Germany 262. Core Genotyping Facility, SAIC-Frederick, Inc., NCI-Frederick, Frederick, MD 21702, USA 263. National Institute for Health and Welfare, FI-90101 Oulu, Finland 264. Department of Epidemiology and Biostatistics, MRC Health Protection Agency (HPE) Centre for Environment and Health, School of Public Health, Imperial College London, UK 265. Unit of Primary Care, Oulu University Hospital, FI-90220 Oulu, Finland 266. Biocenter Oulu, University of Oulu, FI-90014 Oulu, Finland 267. Institute of Health Sciences, FI-90014 University of Oulu, Finland Page 12 of 68 607 608 609 610 611 612 613 614 615 616 617 618 619 620 621 622 623 624 625 626 627 628 629 630 631 632 633 634 635 636 637 638 639 640 641 642 643 644 645 646 647 648 649 650 651 652 653 654 655 656 657 268. Durrer Center for Cardiogenetic Research, Interuniversity Cardiology Institute Netherlands-Netherlands Heart Institute, 3501 DG Utrecht, The Netherlands 269. Interuniversity Cardiology Institute of the Netherlands (ICIN), Utrecht, the Netherlands 270. Faculty of Medicine, Institute of Health Sciences, University of Oulu, Oulu, Finland 271. Unit of General Practice, Oulu University Hospital, Oulu, Finland 272. Department of Urology, Radboud University Medical Centre, 6500 HB Nijmegen, The Netherlands 273. Imperial College Healthcare NHS Trust, London W12 0HS, UK 274. Department of Epidemiology and Public Health, UCL London, WC1E 6BT, UK 275. Department of Medicine, University of Eastern Finland and Kuopio University Hospital, FI-70210 Kuopio, Finland 276. Department of Physiology, Institute of Biomedicine, University of Eastern Finland, Kuopio Campus, Kuopio, Finland 277. Department of Clinical Chemistry, Fimlab Laboratories and School of Medicine University of Tampere, FI-33520 Tampere, Finland 278. Steno Diabetes Center A, S, Gentofte DK-2820, Denmark 279. Institut Universitaire de Cardiologie et de Pneumologie de Québec, Faculty of Medicine, Laval University, Quebec, QC G1V 0A6, Canada 280. Institute of Nutrition and Functional Foods, Laval University, Quebec, QC G1V 0A6, Canada 281. Department of Biostatistics, University of Washington, Seattle, WA 98195, USA 282. Department of Surgery, University Medical Center Utrecht, 3584 CX Utrecht, The Netherlands 283. Department of Biostatistics, University of Liverpool, Liverpool L69 3GA, UK 284. Department of Pediatrics, University of Iowa, Iowa City, Iowa IA 52242, USA 285. Illumina, Inc, Little Chesterford CB10 1XL, UK 286. University of Groningen, University Medical Center Groningen, Department of Pulmonary Medicine and Tuberculosis, Groningen, The Netherlands 287. Department of Neurology, General Central Hospital, Bolzano 39100, Italy 288. Department of Nutrition, Harvard School of Public Health, Boston, Massachusetts, USA 289. Department of Clinical Physiology and Nuclear Medicine, Turku University Hospital, FI-20521 Turku, Finland 290. Research Centre of Applied and Preventive Cardiovascular Medicine, University of Turku, FI-20521 Turku, Finland 291. Human Genomics Laboratory, Pennington Biomedical Research Center, Baton Rouge, LA 70808, USA 292. Université de Montréal, Montreal, Quebec H1T 1C8, Canada 293. Center for Systems Genomics, The Pennsylvania State University, University Park, PA 16802, USA 294. Croatian Centre for Global Health, Faculty of Medicine, University of Split, 21000 Split, Croatia 295. South Carelia Central Hospital. 53130 Lappeenranta. Finland 296. Paul Langerhans Institute Dresden, German Center for Diabetes Research (DZD), Dresden, Germany" 297. International Centre for Circulatory Health, Imperial College London, London W2 1PG, UK 298. Program for Personalized and Genomic Medicine, and Division of Endocrinology, Diabetes and Nutrition, University of Maryland School of Medicine, Baltimore, MD 21201, USA Page 13 of 68 658 659 660 661 662 663 664 665 666 667 668 669 670 671 672 673 674 675 676 677 678 679 680 681 682 683 684 685 686 687 688 689 690 691 692 693 694 695 696 697 698 699 700 701 702 703 704 705 706 707 708 299. Geriatric Research and Education Clinical Center, Vetrans Administration Medical Center, Baltimore, MD 21201, USA 300. HUCH Heart and Lungcenter, Department of Medicine, Helsinki University Central Hospital, FI-00290 Helsinki, Finland 301. Sorbonne Universités, UPMC Univ Paris 06, UMR_S 1166 ,F-75013, Paris, France 302. INSERM, UMR S 1166, Team Genomics and Physiopathology of Cardiovascular Diseases, F-75013, Paris, France 303. ICAN Institute for Cardiometabolism And Nutrition, F-75013, Paris, France 304. Department of Kinesiology, Laval University, Quebec, QC G1V 0A6, Canada 305. Dipartimento di Scienze Farmacologiche e Biomolecolari, Università di Milano & Centro Cardiologico Monzino, IRCCS, Milan 20133, italy 306. Department of Food Science and Nutrition, Laval University, Quebec, QC G1V 0A6, Canada 307. Department of Internal Medicine, University Hospital (CHUV) and University of Lausanne, 1011, Switzerland 308. Department of Nutrition, University of North Carolina, Chapel Hill, NC 27599, USA 309. Institut Pasteur de Lille; INSERM, U744; Université de Lille 2; F-59000 Lille, France 310. Department of Cardiology, Division Heart and Lungs, University Medical Center Utrecht, 3584 CX Utrecht, The Netherlands 311. Department of Medicine, Stanford University School of Medicine, Stanford, CA 94305, USA 312. Lee Kong Chian School of Medicine, Imperial College London and Nanyang Technological University, Singapore, 637553 Singapore, Singapore 313. Health Science Center at Houston, University of Texas, Houston, TX, USA 314. Department of Medicine, Division of Genetics, Brigham and Women's Hospital, Harvard Medical School, Boston, MA 02115, USA 315. Department of Epidemiology, University Medical Center Utrecht, Utrecht, The Netherlands 316. Lund University Diabetes Centre and Department of Clinical Science, Diabetes & Endocrinology Unit, Lund University, Malmö 221 00, Sweden 317. Albert Einstein College of Medicine. Department of epidemiology and population health, Belfer 1306, NY 10461, USA 318. Center for Human Genetics, Division of Public Health Sciences, Wake Forest School of Medicine, Winston-Salem, NC 27157, USA 319. Synlab Academy, Synlab Services GmbH, Mannheim, Germany 320. Department of Clinical Genetics, Erasmus University Medical Center, Rotterdam, The Netherlands 321. Department of Clinical Physiology and Nuclear Medicine, Kuopio University Hospital, Kuopio, Finland 322. Finnish Diabetes Association, Kirjoniementie 15, FI-33680 Tampere, Finland 323. Pirkanmaa Hospital District, Tampere, Finland 324. Center for Non-Communicable Diseases, Karatchi, Pakistan 325. Department of Medicine, University of Pennsylvania, Philadelphia, USA 326. BHF Glasgow Cardiovascular Research Centre, Division of Cardiovascular and Medical Sciences, University of Glasgow, Glasgow G12 8TA, UK 327. Icahn Institute for Genomics and Multiscale Biology, Icahn School of Medicine at Mount Sinai, NY 10580, USA 328. Faculty of Medicine, University of Iceland, Reykjavik 101, Iceland 329. Instituto de Investigacion Sanitaria del Hospital Universario LaPaz (IdiPAZ), Madrid, Spain 330. Diabetes Research Group, King Abdulaziz University, Jeddah, Saudi Arabia Page 14 of 68 709 710 711 712 713 714 715 716 717 718 719 720 721 722 723 724 725 726 727 728 729 730 731 732 733 734 735 736 737 738 739 740 741 742 743 744 331. Centre for Vascular Prevention, Danube-University Krems, 3500 Krems, Austria 332. Department of Public Health and Clinical Nutrition, University of Eastern Finland, Finland 333. Research Unit, Kuopio University Hospital, Kuopio, Finland 334. Institute of Cellular Medicine, Newcastle University, Newcastle NE1 7RU, UK 335. Institute of Medical Informatics, Biometry and Epidemiology, Chair of Epidemiology, Ludwig-Maximilians-Universität, D-85764 Munich, Germany 336. Klinikum Grosshadern, D-81377 Munich, Germany 337. Institute of Epidemiology I, Helmholtz Zentrum München - German Research Center for Environmental Health, Neuherberg, Germany, D-85764 Neuherberg, Germany 338. Department of Pulmonology, University Medical Center Utrecht, Utrecht, The Netherlands 339. King Abdulaziz University, Jeddah 21589, Saudi Arabia 340. Division of Population Health Sciences & Education, St George's, University of London, London SW17 0RE, UK 341. Oxford NIHR Biomedical Research Centre, Oxford University Hospitals NHS Trust, Oxford, OX3 7LJ, UK 342. Clinical Epidemiology, Integrated Research and Treatment Center, Center for Sepsis Control and Care (CSCC), Jena University Hospital, Jena, Germany 343. Department of Human Genetics, University of Michigan, Ann Arbor, MI, USA 344. Service of Medical Genetics, CHUV University Hospital, Lausanne, Switzerland 345. University of Cambridge Metabolic Research Laboratories, Institute of Metabolic Science, Addenbrooke’s Hospital, Cambridge CB2 OQQ, UK 346. NIHR Cambridge Biomedical Research Centre, Institute of Metabolic Science, Addenbrooke’s Hospital, Cambridge CB2 OQQ, UK 347. Carolina Center for Genome Sciences, University of North Carolina at Chapel Hill, Chapel Hill, NC 27599, USA 348. The Mindich Child Health and Development Institute, Icahn School of Medicine at Mount Sinai, New York, NY 10029, USA * These authors contributed equally to this work. § These authors jointly directed this work. Correspondence should be addressed to E.K.S. (espeliot@med.umich.edu), R.J.F.L. (ruth_loos@mssm.edu), and J.N.H. (joelh@broadinstitute.org). Page 15 of 68 745 746 SUMMARY Obesity is heritable and predisposes to many diseases. To better understand the genetic 747 basis of obesity, we conducted a genome-wide association study and Metabochip meta- 748 analysis of body mass index (BMI), a measure commonly used to define obesity and 749 assess adiposity, in up to 339,224 individuals. This analysis identified 97 BMI-associated 750 loci (P < 5×10-8), of which 56 were novel. Five loci demonstrate clear evidence of 751 multiple independent association signals, and many loci have significant effects on other 752 metabolic phenotypes. The 97 loci account for ~2.7% of BMI variation, and genome-wide 753 estimates suggest common variation accounts for >20% of BMI variation. Pathway 754 analyses provide strong support for a role of the central nervous system in obesity 755 susceptibility and implicate new genes and pathways, including those related to synaptic 756 function, glutamate signaling, insulin secretion/action, energy metabolism, lipid biology, 757 and adipogenesis. 758 Page 16 of 68 759 Obesity is a worldwide epidemic associated with increased morbidity and mortality that 760 imposes an enormous burden on individual and public health. Forty to 70% of inter- 761 individual variability in body mass index (BMI), commonly used to assess obesity, has 762 been attributed to genetic factors1-3. At least 77 loci have previously been associated 763 with any obesity measure4-23, including 32 loci from our previous meta-analysis of BMI 764 genome-wide association studies (GWAS)24. Nevertheless, much of the genetic 765 variability in BMI remains unexplained. Moreover, while analyses of previous genetic 766 association results have suggested intriguing biological processes underlying obesity 767 susceptibility, only a few specific genes supported these pathways8,24. For the vast 768 majority of loci, the likely causal gene(s) and relevant pathways remain unknown. 769 770 To further expand the catalog of BMI susceptibility loci and gain a better understanding 771 of the genes and biological pathways influencing obesity, we performed the largest 772 GWAS meta-analysis to date for BMI. Compared to our previous BMI meta-analysis24, 773 this work doubles of the number of individuals contributing GWAS results, incorporates 774 results from >100,000 samples genotyped with the Metabochip25, and nearly doubles the 775 number of loci associated with BMI. In addition, comprehensive assessment of meta- 776 analysis results using integrative approaches provides multiple lines of evidence 777 supporting candidate genes at many loci and highlights pathways that both reinforce and 778 expand our understanding of the biological processes underlying obesity. 779 780 RESULTS 781 Identification of 97 genome-wide significant loci 782 This BMI meta-analysis included genetic association results for up to 339,224 individuals 783 from 125 separate studies. Of these, 82 studies contributed GWAS results for up to 784 236,231 individuals and 43 additional studies contributed BMI association results for up Page 17 of 68 785 to 103,047 individuals based on the Metabochip (Extended Data Table 1, 786 Supplementary Tables 1-3). Taking age and sex into account and after inverse normal 787 transformation of the residuals, we carried out association analyses with genotypes or 788 imputed genotype dosages. GWAS were meta-analyzed together, as were Metabochip 789 studies, followed by a combined GWAS + Metabochip meta-analysis (Extended Data 790 Fig. 1). In total, we analyzed data from up to 322,154 individuals of European-descent 791 (88,137 with Metabochip data) and up to 17,072 individuals, mostly of non-European- 792 descent (14,910 with Metabochip data). 793 794 Our primary meta-analysis of individuals of European-descent from both GWAS and 795 Metabochip (GWAS+Metabochip) studies (N=322,154) identified 77 loci reaching 796 genome-wide significance (GWS) and separated by at least 500 kb (Table 1 and 797 Supplementary Figs. 1 & 2). We carried out additional analyses to explore the effects 798 of power and heterogeneity. The inclusion of 17,072 non-European-descent individuals 799 (total N=339,224) identified an additional ten loci, while secondary analyses of 800 European-descent men (N=152,893) and women (N=171,977) separately, and of 801 population-based studies only (N=209,521) identified another ten GWS loci (Table 2, 802 Supplementary Tables 4-8, Supplementary Figs. 3-9). Of the 97 BMI associated loci, 803 41 have previously been associated with one or more obesity measure11,20,22-24,26. Thus, 804 our current analyses identified 56 novel loci associated with BMI (Tables 1 & 2). 805 806 Effects of associated loci on BMI 807 Newly identified loci generally have lower minor allele frequency (MAF) and/or smaller 808 effect size estimates than previously known loci (Figs. 1A & B). Based on effect 809 estimates from the discovery data set, which can be inflated due to winner’s curse, the 810 97 loci account for 2.7% of the phenotypic variance in BMI (Tables 1 & 2; Figs. 1A & B). Page 18 of 68 811 We conservatively used only GWS SNPs after strict double genomic control (GC) 812 correction, which likely over-corrects association statistics given the lack of evidence for 813 population stratification in family-based analyses (Extended Data Figs. 2 & 3, 814 Extended Data Table 1; A.R.W. et al., submitted). Polygene analyses suggested that 815 SNPs with association P values well below the GWS threshold added significantly to the 816 phenotypic variance explained. For example, 2,346 SNPs selected from conditional and 817 joint multiple SNP analysis with P < 5×10-3 explained 6.6% (SE = 1.1%) of variance, 818 compared to 21.6% (SE = 2.2%) of the variance explained by all SNPs in HapMap3 (31- 819 54% of heritability, Fig. 1C). Further, of the 1,909 independent SNPs (pairwise distance 820 >500 kb and r2 < 0.1) included on Metabochip for replication of potential BMI 821 associations, 1,458 (76.4%) have directionally consistent effects with our previous 822 GWAS meta-analysis24 and the non-overlapping samples in the current meta-analysis 823 (see Methods, Fig. 1D). Based on the significant excess of these directionally consistent 824 observations (sign test P = 2.5×10-123), we estimate approximately 1,007 of the 1,909 825 SNPs represent true associations with BMI (see Methods). 826 827 We compared the effects of our 97 BMI-associated SNPs between the sexes, between 828 ethnicities, and across multiple cross-sections of our data (Methods, Supplementary 829 Tables 4-11, Extended Data Fig. 4). Two previously identified loci, near SEC16B (P = 830 5.2×10-5) and ZFP64 (P = 9.1×10-5) showed evidence of heterogeneity between men 831 and women. Both have stronger effects in women (Supplementary Table 10). We also 832 observed significantly stronger effects at TCF7L2 in T2D case/control studies than in 833 population-based studies (Phet = 5.7×10-7), likely driven by effects of ascertainment in the 834 T2D cases (Phet = 1.3×10-11; Supplementary Tables 10 & 11). We also observed 835 marginally stronger effects at KCNK3 in T2D case/control studies (Phet = 2.6×10-4) and 836 stronger effects in population-based studies than in ascertained studies at CALCR (Phet Page 19 of 68 837 = 3.9×10-4). Two SNPs, near NEGR1 (P = 9.1×10-5) and PRKD1 (P = 1.9×10-5), 838 exhibited significant evidence for heterogeneity of effect between European and African- 839 descent samples, and one SNP, near GBE1 (P = 1.3×10-4), exhibited evidence for 840 heterogeneity between European and East Asian individuals (Supplementary Table 9). 841 These findings may reflect true heterogeneity at these loci, but is most likely due to 842 linkage disequilibrium (LD) differences across ancestries. Effect estimates for 79% of 843 BMI-associated SNPs in African-descent samples (P = 9.2×10-9) and 91% in East Asian 844 samples (P = 1.8×10-15) showed directional consistency with those observed in our 845 European-only analyses. These results suggest that common SNPs associated with BMI 846 have comparable effects across ancestry groups and between sexes. 847 848 We also took advantage of LD differences across populations to fine-map association 849 signals using the Bayesian methods of Maller et al.27 and Wakefield28. At ten of 27 loci 850 fine-mapped for BMI on Metabochip, the addition of non-European individuals into the 851 meta-analysis either 1) narrowed the genomic region containing the 99% credible set, or 852 2) decreased the number of SNPs in the credible set (Supplementary Table 12 and 853 Supplementary Fig. 10). At TCF7L2, the region decreased from >54 kb to 14 kb, 854 containing 4 SNPs, where the most likely SNP corresponds with the type 2 diabetes 855 (T2D) associated variant rs7093146. At the SEC16B and FTO loci, the all ancestries 856 credible set includes a single SNP, though the SNP we highlight at FTO (rs1558902) 857 differs from that identified by a recent fine-mapping effort in African American cohorts29. 858 Fine-mapping efforts using larger, more diverse study samples will further narrow 859 association signals. Regardless of fine-mapping methodology or sample diversity, a 860 complete catalogue of variation for the region of interest is necessary to identify causal 861 variants. For example, a known missense variant at the SH2B1 locus thought to be the Page 20 of 68 862 causal variant was not captured by the Metabochip, and thus is not included in the 863 credible interval. 864 865 We examined the combined effects of the lead SNP at the 97 loci in an independent 866 sample of 8,164 European-descent individuals from the Health and Retirement Study30. 867 We observed an average increase of 0.1 BMI units (kg/m2) per BMI-increasing allele, 868 equivalent to 260 to 320 grams for an individual 160-180 cm in height. There was a 1.8 869 kg/m2 difference in mean BMI between the 145 individuals (1.78%) carrying the most of 870 BMI-increasing alleles (>104 alleles) and those carrying the mean number of BMI- 871 increasing alleles in the sample (91 alleles, Fig. 1E), corresponding to a difference of 4.6 872 to 5.8 kg for an individual 160 to 180 cm in height, and a 1.5 kg/m2 difference (3.8 to 4.9 873 kg difference) in mean BMI between the 95 individuals (1.16%) carrying the fewest BMI- 874 increasing alleles (<78 alleles) and those carrying the mean number. Such differences 875 are medically significant in predisposing to development of metabolic disease31. For 876 predicting obesity (BMI ≥ 30 kg/m2), adding the genetic risk score to a model also 877 including age, age2, sex, and four genotype-based principal components significantly 878 increases the area under the receiver-operating characteristic (ROC) curve from 0.576 879 to 0.601. 880 881 Additional associated variants at BMI loci 882 To identify additional SNPs with independent BMI associations at the 97 established loci, 883 we used GCTA32 to perform an approximate joint and conditional association analysis33 884 using summary level results from European sex-combined meta-analysis after removing 885 family-based validation studies (TwinGene and QIMR; See Methods). GCTA analysis 886 confirmed two signals at MC4R that had previously been identified using exact 887 conditional analyses in individual cohorts24, and identified a total of five loci harboring Page 21 of 68 888 variants with independent evidence of association from the index SNPs (Table 3): 889 second signals near FLJ30838, NLRC3/ADCY9, GPRC5B/GP2, and BDNF, and a third 890 signal near MC4R (rs9944545) located between the two known signals (Fig. 1F). Joint 891 conditional analyses at two genomic regions separated by >500 kb (AGBL4/ELAVL4 on 892 chromosome 1 and ATP2A1/SBK1 on chromosome 16), indicate that these pairs of 893 signals may not be independent due to extended LD. 894 895 Effects of BMI variants on other traits 896 We tested for associations between our 97 BMI-associated index SNPs and other 897 metabolic phenotypes (Supplementary Tables 13-15, Fig. 2, Extended Data Fig. 5). 898 Thirteen of the 23 phenotypes tested had more SNPs with effects in the anticipated 899 direction than expected by chance (Supplementary Table 16). These results 900 corroborate the epidemiological relationships of BMI with metabolic traits. Whether this 901 reflects a common genetic etiology or a causal relationship of BMI on these traits 902 requires further investigation. 903 904 Interestingly, some individual loci showed significant association with traits in the 905 opposite direction than expected based on their phenotypic correlation with BMI (Fig. 2). 906 For example, at HHIP, the BMI-increasing allele is associated with decreased risk for 907 T2D and higher high-density lipoprotein (HDL). At LOC646736/IRS1, the BMI-increasing 908 allele is associated with reduced risk of CAD and diabetic nephropathy, decreased 909 triglyceride (TG) levels, increased HDL, higher adiponectin, and lower fasting insulin. 910 This may be due to increased subcutaneous fat and possible production of metabolic 911 mediators that are protective against the development of metabolic disease despite 912 increased adiposity22. These unexpected associations may help us to better understand 913 the complex pathophysiology underlying these traits and may indicate potential Page 22 of 68 914 additional benefits or potential side effects if these regions are targets of therapeutic 915 intervention. Further, of our 97 GWS loci, 35 (binomial P = 0.0019) were in high LD (r2 > 916 0.7) with one or more GWS SNPs in the NHGRI GWAS catalog (P < 5×10-8), even after 917 removing SNPs associated with anthropometric traits. These SNPs were associated not 918 only with cardiometabolic traits, but also with schizophrenia, smoking behavior, irritable 919 bowel syndrome, and Alzheimer’s disease, suggesting intriguing genetic links between 920 BMI and a diverse group of traits (Supplementary Tables 17A & B, Extended Data Fig. 921 5). 922 923 Relevant BMI tissues, biological pathways, and gene sets 924 We anticipated that the expanded sample size would not only identify additional BMI- 925 associated variants and loci, but would also more clearly highlight the biology implicated 926 by genetic studies of BMI. By applying multiple complementary methods, we identified 927 biologically relevant tissues, pathways and gene sets, and also highlighted genes likely 928 to be causal contributors at associated loci. These approaches included systematic 929 methods incorporating diverse data types, including a novel approach called DEPICT 930 (Data-driven Enrichment-Prioritized Integration for Complex Traits, see Methods), and 931 extensive manual review of the published literature. 932 933 DEPICT used 37,000 human gene expression microarray samples to identify tissues 934 and cell types in which genes near top BMI-associated SNPs are highly expressed, and 935 then tested for enrichment of specific tissues by comparing results with randomly 936 selected loci matched for gene density (see Methods). Twenty-seven of 31 significantly 937 enriched tissues were in the central nervous system (out of 290 tested). Current results 938 are not sufficient to isolate specific brain regions important in regulating BMI, however 939 we observe enrichment not only in the hypothalamus/pituitary gland -- key sites of Page 23 of 68 940 central appetite regulation -- but even more strongly in the hippocampus/limbic system, 941 tissues that play a role in learning, cognition, emotion, and memory (Fig. 3A; 942 Supplementary Table 18). Similar analyses for other anthropometric traits showed 943 enrichment in different tissues suggesting these results are specific to BMI (A.R.W.et al. 944 and D. Shungin et al.,submitted). 945 946 As a complementary approach to evaluate whether specific cell types/tissues could be 947 implicated by BMI-associated variants, we examined overlap of associated variants at 948 the 97 loci (r2 > 0.7 with the lead SNP) with five regulatory marks found in most of the 14 949 selected cell types from brain, blood, liver, pancreatic islet, and adipose tissue from the 950 Encyclopedia of DNA Elements (ENCODE) Consortium34 and Roadmap Epigenomics 951 Project35 (Supplementary Tables 19A, B & C). We found evidence of enrichment (P < 952 1.2×10-3) in 24 of 41 datasets examined. The strongest enrichment was observed with 953 promoter (H3K4me3, H3K9ac) and enhancer (H3K4me1, HeK27ac) marks detected in 954 mid-frontal lobe, anterior caudate, astrocytes, and substantia nigra, further corroborating 955 neuronal tissues in regulation of BMI. 956 957 To identify pathways or gene sets implicated by the BMI-associated loci, we first used 958 MAGENTA36, which takes as input pre-annotated gene sets, and then tests for 959 overrepresentation of the genes in each gene set at BMI-associated loci. We found 960 enrichment (false discovery rate (FDR) < 0.05) of seven gene sets, including 961 neurotrophin signaling. Other highlighted gene sets were related to general growth and 962 patterning: basal cell carcinoma, acute myeloid leukemia, and hedgehog signaling 963 (Supplementary Tables 20 A & B). 964 Page 24 of 68 965 Second, we employed DEPICT to perform gene set enrichment analysis. DEPICT first 966 reconstituted predefined gene sets based on data from over 77,000 gene expression 967 microarrays for close to 20,000 genes. DEPICT then incorporated all genes near top 968 BMI-associated SNPs and used the reconstituted gene sets as a reference to prioritize 969 genes and reconstituted gene sets at and across BMI-associated loci (see Methods). 970 After merging highly correlated gene sets, nearly 500 gene sets were significantly 971 enriched (FDR < 0.05) for genes in BMI-associated loci (Fig. 3B; Extended Data Fig. 6; 972 Supplementary Tables 21A & B). The most strongly enriched gene sets highlight 973 potentially novel pathways in the central nervous system (CNS). These include gene 974 sets related to synaptic function, long term potentiation, and neurotransmitter signaling 975 (glutamate signaling in particular, but also norepinephrine, dopamine, and serotonin 976 release cycles, and gamma-aminobutyric acid receptor activity; Fig. 3C). Potentially 977 relevant mouse behavioral phenotypes, such as physical activity and impaired 978 coordination were also highly enriched by DEPICT (Fig. 3B; Supplementary Table 979 21A). Several gene sets previously linked to obesity, such as integration of energy 980 metabolism, polyphagia, secretion and action of insulin and related hormones (e.g., 981 “regulation of insulin secretion by glucagon-like peptide-1” and “glucagon signaling in 982 metabolic regulation”), MTOR signaling (which affects cell growth in response to nutrient 983 intake via insulin and growth factors37), and gene sets overlapping the neurotrophin 984 signaling pathway identified by MAGENTA were also enriched in DEPICT, though not as 985 significantly as CNS processes (Fig. 3D). DEPICT also identified significant enrichment 986 for other cellular components and processes: calcium channels, MAP kinase activity, 987 chromatin organization/modification, and ubiquitin ligases. 988 989 Finally, we manually reviewed published literature related to 405 genes within the 97 990 associated loci (all genes within 500 kb and r2 > 0.2 to the index SNPs; see Methods). Page 25 of 68 991 We classified these genes into one or more biological categories (see Methods), and 992 observed 25 categories containing three or more genes (Supplementary Table 22). The 993 largest category comprised genes involved in neuronal processes, including monogenic 994 obesity genes involved in hypothalamic function/energy homeostasis and genes involved 995 in neuronal transmission and neuronal development. Other processes highlighted by the 996 manual literature review included glucose and lipid homeostasis as well as limb 997 development, which were less notable in the above methods, but may still be related to 998 the underlying biology of BMI. 999 1000 To identify specific genes that may account for the BMI association, we considered each 1001 of the following to represent supportive evidence for a gene within a locus: a) the gene 1002 nearest to the index SNP38,39, b) genes harboring missense, nonsense, or copy number 1003 variants, or a cis-expression quantitative trait locus (eQTL) in LD with the index SNP40,41, 1004 c) prioritized by integrative methods implemented in DEPICT, d) prioritized by 1005 connections in published abstracts as implemented in GRAIL42, or e) biologically related 1006 to obesity, related metabolic disease, or energy expenditure based on manual literature 1007 review (Table 1 & 2, Extended Data Tables 2 & 3, Supplementary Tables 23, 24, & 1008 25). We first focused on the 64 genes in associated loci with more than one consistent 1009 line of supporting evidence. As expected, many of these genes overlap with CNS 1010 processes, including those gene sets identified by the above pathway methods. ELAVL4, 1011 GRID1, CADM2, NRXN3, NEGR1, and SCG3 are implicated in synaptic function, cell- 1012 cell adhesion, and glutamate signaling. The pathways implicated also include genes that 1013 cause monogenic obesity syndromes (MC4R, BDNF, BBS4, POMC), or function in 1014 extreme/early onset obesity in humans and mouse models (SH2B1, NEGR1)8,43,44. Other 1015 genes with multiple lines of supporting evidence are related to insulin secretion/action, 1016 energy metabolism, lipid biology, and/or adipogenesis (TCF7L2, GIPR, IRS1, FOXO3, Page 26 of 68 1017 ASB4, RPTOR, NPC1, CREB1, FAM57B, APOB48, HSD17B12), encode RNA 1018 binding/processing proteins (PTBP2, ELAVL4, CELF1, RALYL), are in the MAP kinase 1019 signaling pathway (MAP2K5, MAPK3), or regulate cell proliferation or cell survival 1020 (FAIM2, PARK2, OLFM4). 1021 1022 Other loci contain good candidates either highlighted by DEPICT only or, alternatively, 1023 fall short of the DEPICT FDR threshold but have other lines of supporting evidence 1024 (Table 1 & 2). For example, PCDH9, TAOK2, and STX1B are prioritized by DEPICT and 1025 are related to synaptic function and/or glutamate signaling. Several other associated loci 1026 contain likely relevant genes that are only supported by manual review of the literature, 1027 including several related to glucose, energy, or lipid/cholesterol metabolism (KLF7, GRP, 1028 ADPGK, APOE, HMGCR). Although we cannot be certain that any individual gene is 1029 related to the association at a given locus, the strong enrichment of pathways among 1030 genes within associated loci argues for a causal role for these pathways, prioritizes 1031 specific genes for follow-up experiments, and provides the strongest genetic evidence to 1032 date for a role of particular biological and CNS processes in the regulation of human 1033 body mass. 1034 1035 Discussion 1036 Our meta-analysis of nearly 340,000 individuals identified 97 GWS loci associated with 1037 BMI, including 56 novel loci. Together these loci account for 2.7% of the variation in BMI, 1038 and suggest that as much as 21% of BMI variation can be accounted for by common 1039 genetic variation. Our analyses now provide robust data to implicate particular genes 1040 and pathways affecting BMI, including those involved in synaptic function and glutamate 1041 receptor signaling. Synaptic plasticity and glutamate receptor activity respond to 1042 changes in feeding and fasting, are regulated by key obesity-related molecules such as Page 27 of 68 1043 BDNF and MC4R, and impinge on key hypothalamic circuits45-48. Also, these pathways 1044 overlap with one of the several proposed mechanisms of action of topiramate, which is a 1045 component of one of two weight-loss drugs approved by the United States Food and 1046 Drug Administration49,50. This observation suggests the relevant site of action for this 1047 drug may be glutamate receptor activity, which supports the idea that these genes 1048 and pathways could reveal more targets for weight-loss therapies. BMI-associated loci 1049 also overlap with genes and pathways implicated in neurodevelopmental processes 1050 (Supplementary Tables 21A & B, 22). Finally, consistent with previous work and with 1051 findings from monogenic obesity syndromes, we confirm a role for the central nervous 1052 system -- particularly genes expressed in the hypothalamus -- in regulation of body mass. 1053 1054 Examining the genes at BMI-associated loci in the context of gene expression, molecular 1055 pathways, eQTL results, mutational evidence, and positional data (i.e., the gene nearest 1056 the signal) provides multiple complementary avenues through which to prioritize genes 1057 for relevance in BMI biology. Genes, such as NPC1 and ELAVL4, are implicated by 1058 multiple lines of evidence (literature, mutational, eQTL, and DEPICT) and become strong 1059 candidate genes in their respective regions. However, in most regions, only one or a few 1060 lines of evidence prioritize a particular gene. Literature review also helps identify high 1061 quality candidate genes and processes, particularly related to metabolism. These genes 1062 highlight insulin biology, energy homeostasis, lipid biology, and adipogenesis. It is 1063 important to recognize that pathway methods and literature reviews are limited by 1064 current data sets and knowledge, and thus provide only a working model of obesity 1065 biology. For example, little is known about host genetic factors that regulate the 1066 microbiome. Variation in immune-related genes such as TLR4 could presumably exert 1067 an influence on obesity through the microbiome51. Together, our results underscore the Page 28 of 68 1068 heterogeneous etiology of obesity and its links with multiple related metabolic diseases 1069 and processes. 1070 1071 BMI variants are generally associated with related cardiometabolic traits in accord with 1072 established epidemiological relationships. This could be due to shared genetic effects or 1073 to other causes of cross-phenotypic correlations. However, some BMI-associated 1074 variants have effects on related traits counter to epidemiological expectations. Once 1075 better understood, these mechanisms may not only help to explain why not all obese 1076 individuals develop related metabolic diseases, but may also suggest possible 1077 mechanisms to prevent development of metabolic disease in those who are already 1078 obese. 1079 1080 Larger studies of common genetic variation, studies of rare variation (including those 1081 based on imputation, exome chips, and sequencing), and improved computation tools 1082 will continue to identify genetic variants associated with BMI and further refine the 1083 biology of obesity. Already, the 97 loci identified here represent an important step in 1084 understanding the physiological mechanisms leading to obesity. These findings 1085 strengthen the connection between obesity and other metabolic diseases, enhance our 1086 appreciation of the tissues, physiological processes, and molecular pathways that 1087 contribute to obesity, and will guide future research aimed at unraveling the complex 1088 biology of obesity. 1089 Page 29 of 68 1090 Methods Summary 1091 Main Analyses We conducted a two-stage of meta-analysis of genetic association 1092 studies for BMI in European-descent adults (Supplementary Fig. 1). Secondary meta- 1093 analyses included: 1) all ancestries (European + Non-European), 2) European men, 3) 1094 European women, and 4) European population-based studies. Each study performed 1095 linear regression assuming an additive genetic model on inverse normally transformed 1096 BMI (kg/m2). QC following study level analyses was conducted following procedures 1097 outlined elsewhere (T.W.W., in press). Fixed effects inverse-variance weighted meta- 1098 analyses were conducted using METAL52, with significance evaluated at P < 5×10-8. 1099 Heterogeneity was assessed between groups using methods outlined elsewhere15 and 1100 using P < 5×10-4 to assess significance. Fine-mapping Comparing meta-analyses 1101 across ethnic populations, we calculated credible sets of SNPs likely to contain the 1102 causal variant27,28. Risk, variance explained, and secondary signals We assessed the 1103 cumulative effects of the 97 GWS loci on mean BMI and to predict obesity (BMI ≥30 1104 kg/m2) using the c statistic from logistic regression. Polygene analyses and approximate, 1105 conditional analyses were performed using GCTA32,33. We performed within-family 1106 prediction analysis using full-sib pairs selected from independent families and selected 1107 SNPs were used to calculate the percent of phenotypic variance explained and risk 1108 prediction. The SNP-derived predictor was calculated using PLINK and estimation 1109 analyses were performed using GCTA32,53,54. Enrichment of Metabochip SNPs We 1110 used the 1,909 independent SNPs included for BMI replication on the Metabochip to 1111 determine the number of SNPs with directional consistency between Speliotes et al.24 1112 and the current analysis. Functional variants All variants within 500 kb and in LD (r2 > 1113 0.7) with a BMI SNP were annotated for protein-coding effects based on RefSeq 1114 transcripts. CNVs We used a list of SNPs in high LD (r2 > 0.8) with known CNVs in 1115 European populations55 to test if CNVs account for BMI associations. eQTLs We Page 30 of 68 1116 examined cis associations between BMI loci and expression of nearby genes in several 1117 tissues56-63. We report associations with a study-specific FDR of 5% (or 1% for some 1118 datasets), that are in LD (r2 > 0.7) with the BMI SNP, and with P > 0.05 for the peak 1119 expression SNP after conditioning on the BMI SNP. Pathway analyses MAGENTA was 1120 used to identify predefined gene sets enriched for association signals36. GRAIL was 1121 used to identify genes near BMI loci co-occurring in the published scientific text42. We 1122 used DEPICT to identify the most likely causal gene at a given locus, gene sets enriched 1123 in genetic associations, tissues and cell types in which nearby genes are highly 1124 expressed, and to detect significantly enriched pathways for GWS results (T. H. Pers et 1125 al., in preparation). Cross-trait lookups Association results for the 97 BMI SNPs were 1126 requested from 13 consortia with GWAS on related cardiometabolic traits. We employed 1127 the meta-regression technique to determine the joint effect of all 97 BMI-increasing loci 1128 on other cardiometabolic phenotypes64. For each cardiometabolic trait, we converted the 1129 effect estimates and standard errors (or P values) from meta-analysis to Z-scores 1130 oriented with respect to the BMI-increasing allele, then classified each BMI SNP as 1131 having a positive, negative, or non-significant effect on each of the traits, and generated 1132 a heat map using Euclidean distance and complete linkage clustering to order both loci 1133 and traits (Fig. 2). A bubble plot (Extended Data Fig. 5) was generated using all BMI 1134 loci that reached P < 5.15×10-4 (0.05/97) for each related cardiometabolic trait. NHGRI 1135 GWAS Catalog Lookups We used the NHGRI GWAS Catalog65 and manual curated 1136 genome-wide publications to identify associations within 500 kb and r2 > 0.7 with a BMI- 1137 index SNP. Regulatory variation We tested variants in LD (r2 > 0.7) with BMI SNPs for 1138 global enrichment of regulatory marks using permutation-based tests in a subset of 41 1139 markers of open chromatin, histone modification, and transcription factor binding35,66. 1140 Marks were considered significantly enriched if P < 1.2×10-3. 1141 Page 31 of 68 1142 1143 Full Methods and any associated references are available in the online version of the 1144 paper at www.nature.com/nature 1145 1146 Page 32 of 68 1147 References 1148 1149 1150 1151 1152 1153 1154 1155 1156 1157 1158 1159 1160 1161 1162 1163 1164 1165 1166 1167 1168 1169 1170 1171 1172 1173 1174 1175 1176 1177 1178 1179 1180 1181 1182 1183 1184 1185 1186 1187 1188 1189 1190 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. 11. 12. 13. 14. 15. Maes, H.H., Neale, M.C. & Eaves, L.J. Genetic and environmental factors in relative body weight and human adiposity. Behavior genetics 27, 325-51 (1997). Visscher, P.M., Brown, M.A., McCarthy, M.I. & Yang, J. Five years of GWAS discovery. American journal of human genetics 90, 7-24 (2012). Zaitlen, N. et al. Using extended genealogy to estimate components of heritability for 23 quantitative and dichotomous traits. PLoS genetics 9, e1003520 (2013). Frayling, T.M. et al. A common variant in the FTO gene is associated with body mass index and predisposes to childhood and adult obesity. Science 316, 889-94 (2007). Scuteri, A. et al. Genome-wide association scan shows genetic variants in the FTO gene are associated with obesity-related traits. PLoS genetics 3, e115 (2007). Loos, R.J. et al. Common variants near MC4R are associated with fat mass, weight and risk of obesity. Nature genetics 40, 768-75 (2008). Chambers, J.C. et al. Common genetic variation near MC4R is associated with waist circumference and insulin resistance. Nature genetics 40, 716-8 (2008). Willer, C.J. et al. Six new loci associated with body mass index highlight a neuronal influence on body weight regulation. Nature genetics 41, 25-34 (2009). Thorleifsson, G. et al. Genome-wide association yields new sequence variants at seven loci that associate with measures of obesity. Nature genetics 41, 18-24 (2009). Okada, Y. et al. Common variants at CDKAL1 and KLF9 are associated with body mass index in east Asian populations. Nature genetics 44, 3026 (2012). Monda, K.L. et al. A meta-analysis identifies new loci associated with body mass index in individuals of African ancestry. Nature genetics 45, 690-6 (2013). Lindgren, C.M. et al. Genome-wide association scan meta-analysis identifies three Loci influencing adiposity and fat distribution. PLoS genetics 5, e1000508 (2009). Heard-Costa, N.L. et al. NRXN3 is a novel locus for waist circumference: a genome-wide association study from the CHARGE Consortium. PLoS genetics 5, e1000539 (2009). Heid, I.M. et al. Meta-analysis identifies 13 new loci associated with waisthip ratio and reveals sexual dimorphism in the genetic basis of fat distribution. Nature genetics 42, 949-60 (2010). Randall, J.C. et al. Sex-stratified genome-wide association studies including 270,000 individuals show sexual dimorphism in genetic loci for anthropometric traits. PLoS genetics 9, e1003500 (2013). Page 33 of 68 1191 1192 1193 1194 1195 1196 1197 1198 1199 1200 1201 1202 1203 1204 1205 1206 1207 1208 1209 1210 1211 1212 1213 1214 1215 1216 1217 1218 1219 1220 1221 1222 1223 1224 1225 1226 1227 1228 1229 1230 1231 1232 1233 1234 1235 16. 17. 18. 19. 20. 21. 22. 23. 24. 25. 26. 27. 28. 29. 30. Fox, C.S. et al. Genome-wide association for abdominal subcutaneous and visceral adipose reveals a novel locus for visceral fat in women. PLoS genetics 8, e1002695 (2012). Liu, C.T. et al. Genome-wide association of body fat distribution in african ancestry populations suggests new Loci. PLoS genetics 9, e1003681 (2013). Meyre, D. et al. Genome-wide association study for early-onset and morbid adult obesity identifies three new risk loci in European populations. Nature genetics 41, 157-9 (2009). Scherag, A. et al. Two new Loci for body-weight regulation identified in a joint analysis of genome-wide association studies for early-onset extreme obesity in French and german study groups. PLoS genetics 6, e1000916 (2010). Berndt, S.I. et al. Genome-wide meta-analysis identifies 11 new loci for anthropometric traits and provides insights into genetic architecture. Nature genetics 45, 501-12 (2013). Wheeler, E. et al. Genome-wide SNP and CNV analysis identifies common and low-frequency variants associated with severe early-onset obesity. Nature genetics 45, 513-7 (2013). Kilpelainen, T.O. et al. Genetic variation near IRS1 associates with reduced adiposity and an impaired metabolic profile. Nature genetics 43, 753-60 (2011). Guo, Y. et al. Gene-centric meta-analyses of 108 912 individuals confirm known body mass index loci and reveal three novel signals. Human molecular genetics 22, 184-201 (2013). Speliotes, E.K. et al. Association analyses of 249,796 individuals reveal 18 new loci associated with body mass index. Nature genetics 42, 937-48 (2010). Voight, B.F. et al. The metabochip, a custom genotyping array for genetic studies of metabolic, cardiovascular, and anthropometric traits. PLoS genetics 8, e1002793 (2012). Bradfield, J.P. et al. A genome-wide association meta-analysis identifies new childhood obesity loci. Nature genetics 44, 526-31 (2012). Maller, J.B. et al. Bayesian refinement of association signals for 14 loci in 3 common diseases. Nature genetics 44, 1294-301 (2012). Wakefield, J. A Bayesian measure of the probability of false discovery in genetic epidemiology studies. American journal of human genetics 81, 208-27 (2007). Peters, U. et al. A systematic mapping approach of 16q12.2/FTO and BMI in more than 20,000 African Americans narrows in on the underlying functional variation: results from the Population Architecture using Genomics and Epidemiology (PAGE) study. PLoS genetics 9, e1003171 (2013). Juster, F.T. & Suzman, R. An Overview of the Health and Retirement Study. Journal of Human Resources 30, S7-S56 (1995). Page 34 of 68 1236 1237 1238 1239 1240 1241 1242 1243 1244 1245 1246 1247 1248 1249 1250 1251 1252 1253 1254 1255 1256 1257 1258 1259 1260 1261 1262 1263 1264 1265 1266 1267 1268 1269 1270 1271 1272 1273 1274 1275 1276 1277 1278 1279 1280 1281 31. 32. 33. 34. 35. 36. 37. 38. 39. 40. 41. 42. 43. 44. 45. 46. 47. 48. Bouchonville, M. et al. Weight loss, exercise or both and cardiometabolic risk factors in obese older adults: results of a randomized controlled trial. International journal of obesity (2013). Yang, J., Lee, S.H., Goddard, M.E. & Visscher, P.M. GCTA: a tool for genome-wide complex trait analysis. American journal of human genetics 88, 76-82 (2011). Yang, J. et al. Conditional and joint multiple-SNP analysis of GWAS summary statistics identifies additional variants influencing complex traits. Nature genetics 44, 369-75, S1-3 (2012). ENCODE_Project_Consortium et al. An integrated encyclopedia of DNA elements in the human genome. Nature 489, 57-74 (2012). Bernstein, B.E. et al. The NIH Roadmap Epigenomics Mapping Consortium. Nature biotechnology 28, 1045-8 (2010). Segre, A.V., Groop, L., Mootha, V.K., Daly, M.J. & Altshuler, D. Common inherited variation in mitochondrial genes is not enriched for associations with type 2 diabetes or related glycemic traits. PLoS genetics 6(2010). Wullschleger, S., Loewith, R. & Hall, M.N. TOR signaling in growth and metabolism. Cell 124, 471-84 (2006). Lango Allen, H. et al. Hundreds of variants clustered in genomic loci and biological pathways affect human height. Nature 467, 832-8 (2010). Teslovich, T.M. et al. Biological, clinical and population relevance of 95 loci for blood lipids. Nature 466, 707-13 (2010). Saxena, R. et al. Genetic variation in GIPR influences the glucose and insulin responses to an oral glucose challenge. Nature genetics 42, 142-8 (2010). Musunuru, K. et al. From noncoding variant to phenotype via SORT1 at the 1p13 cholesterol locus. Nature 466, 714-9 (2010). Raychaudhuri, S. et al. Identifying relationships among genomic disease regions: predicting genes at pathogenic SNP associations and rare deletions. PLoS genetics 5, e1000534 (2009). Magi, R. et al. Contribution of 32 GWAS-Identified Common Variants to Severe Obesity in European Adults Referred for Bariatric Surgery. PloS one 8, e70735 (2013). Lee, A.W. et al. Functional inactivation of the genome-wide association study obesity gene neuronal growth regulator 1 in mice causes a body mass phenotype. PloS one 7, e41537 (2012). Yang, Y., Atasoy, D., Su, H.H. & Sternson, S.M. Hunger states switch a flip-flop memory circuit via a synaptic AMPK-dependent positive feedback loop. Cell 146, 992-1003 (2011). Wu, Q., Clark, M.S. & Palmiter, R.D. Deciphering a neuronal circuit that mediates appetite. Nature 483, 594-7 (2012). Melo, C.V. et al. BDNF regulates the expression and distribution of vesicular glutamate transporters in cultured hippocampal neurons. PloS one 8, e53793 (2013). Shen, Y., Fu, W.Y., Cheng, E.Y., Fu, A.K. & Ip, N.Y. Melanocortin-4 receptor regulates hippocampal synaptic plasticity through a protein Page 35 of 68 1282 1283 1284 1285 1286 1287 1288 1289 1290 1291 1292 1293 1294 1295 1296 1297 1298 1299 1300 1301 1302 1303 1304 1305 1306 1307 1308 1309 1310 1311 1312 1313 1314 1315 1316 1317 1318 1319 1320 1321 1322 1323 1324 1325 1326 1327 49. 50. 51. 52. 53. 54. 55. 56. 57. 58. 59. 60. 61. 62. 63. 64. 65. kinase A-dependent mechanism. The Journal of neuroscience : the official journal of the Society for Neuroscience 33, 464-72 (2013). Gibbs, J.W., 3rd, Sombati, S., DeLorenzo, R.J. & Coulter, D.A. Cellular actions of topiramate: blockade of kainate-evoked inward currents in cultured hippocampal neurons. Epilepsia 41 Suppl 1, S10-6 (2000). Poulsen, C.F. et al. Modulation by topiramate of AMPA and kainate mediated calcium influx in cultured cerebral cortical, hippocampal and cerebellar neurons. Neurochemical research 29, 275-82 (2004). Henao-Mejia, J. et al. Inflammasome-mediated dysbiosis regulates progression of NAFLD and obesity. Nature 482, 179-85 (2012). Willer, C.J., Li, Y. & Abecasis, G.R. METAL: fast and efficient metaanalysis of genomewide association scans. Bioinformatics 26, 2190-1 (2010). Purcell, S. et al. PLINK: a tool set for whole-genome association and population-based linkage analyses. American journal of human genetics 81, 559-75 (2007). Frazer, K.A. et al. A second generation human haplotype map of over 3.1 million SNPs. Nature 449, 851-61 (2007). Mills, R.E. et al. Mapping copy number variation by population-scale genome sequencing. Nature 470, 59-65 (2011). Emilsson, V. et al. Genetics of gene expression and its effect on disease. Nature 452, 423-8 (2008). Zhong, H., Yang, X., Kaplan, L.M., Molony, C. & Schadt, E.E. Integrating pathway analysis and genetics of gene expression for genome-wide association studies. American journal of human genetics 86, 581-91 (2010). Grundberg, E. et al. Mapping cis- and trans-regulatory effects across multiple tissues in twins. Nature genetics 44, 1084-9 (2012). Dixon, A.L. et al. A genome-wide association study of global gene expression. Nature genetics 39, 1202-7 (2007). Fehrmann, R.S. et al. Trans-eQTLs reveal that independent genetic variants associated with a complex phenotype converge on intermediate genes, with a major role for the HLA. PLoS genetics 7, e1002197 (2011). Nelis, M. et al. Genetic structure of Europeans: a view from the North-East. PloS one 4, e5472 (2009). Myers, A.J. et al. A survey of genetic human cortical gene expression. Nature genetics 39, 1494-9 (2007). Westra, H.J. et al. Systematic identification of trans eQTLs as putative drivers of known disease associations. Nature genetics (2013). Dastani, Z. et al. Novel loci for adiponectin levels and their influence on type 2 diabetes and metabolic traits: a multi-ethnic meta-analysis of 45,891 individuals. PLoS genetics 8, e1002607 (2012). Hindorff, L.A. et al. Potential etiologic and functional implications of genome-wide association loci for human diseases and traits. Proceedings of the National Academy of Sciences of the United States of America 106, 9362-7 (2009). Page 36 of 68 1328 1329 1330 1331 1332 1333 1334 1335 1336 1337 1338 1339 1340 1341 1342 1343 1344 1345 1346 1347 1348 1349 1350 1351 1352 1353 1354 1355 1356 1357 1358 1359 1360 1361 1362 1363 1364 1365 1366 1367 1368 1369 1370 1371 66. 67. 68. 69. 70. 71. 72. 73. 74. 75. 76. 77. 78. 79. 80. 81. 82. 83. 84. Bernstein, B.E. et al. An integrated encyclopedia of DNA elements in the human genome. Nature 489, 57-74 (2012). Winkler, T.W. et al. Quality control and conduct of genome-wide association meta-analyses. Nature Protocols (Accepted Feb. 6, 2014). Devlin, B. & Roeder, K. Genomic control for association studies. Biometrics 55, 997-1004 (1999). Wen, W. et al. Meta-analysis identifies common variants associated with body mass index in east Asians. Nature genetics 44, 307-11 (2012). Wang, K., Li, M. & Hakonarson, H. ANNOVAR: functional annotation of genetic variants from high-throughput sequencing data. Nucleic acids research 38, e164 (2010). Adzhubei, I.A. et al. A method and server for predicting damaging missense mutations. Nature methods 7, 248-9 (2010). (ESP), N.G.E.S.P. Exome Variant Server. Vol. 2013 (Seattle, WA). Ng, P.C. & Henikoff, S. Predicting deleterious amino acid substitutions. Genome research 11, 863-74 (2001). Morris, A.P. et al. Large-scale association analysis provides insights into the genetic architecture and pathophysiology of type 2 diabetes. Nature genetics 44, 981-90 (2012). Deloukas, P. et al. Large-scale association analysis identifies new risk loci for coronary artery disease. Nature genetics 45, 25-33 (2013). Ehret, G.B. et al. Genetic variants in novel pathways influence blood pressure and cardiovascular disease risk. Nature 478, 103-9 (2011). Scott, R.A. et al. Large-scale association analyses identify new loci influencing glycemic traits and provide insight into the underlying biological pathways. Nature genetics 44, 991-1005 (2012). Manning, A.K. et al. A genome-wide approach accounting for body mass index identifies genetic variants influencing fasting glycemic traits and insulin resistance. Nature genetics 44, 659-69 (2012). Pattaro, C. et al. Genome-wide association and functional follow-up reveals new loci for kidney function. PLoS genetics 8, e1002584 (2012). Boger, C.A. et al. CUBN is a gene locus for albuminuria. Journal of the American Society of Nephrology : JASN 22, 555-70 (2011). Stolk, L. et al. Meta-analyses identify 13 loci associated with age at menopause and highlight DNA repair and immune pathways. Nature genetics 44, 260-8 (2012). Elks, C.E. et al. Thirty new loci for age at menarche identified by a metaanalysis of genome-wide association studies. Nature genetics 42, 1077-85 (2010). Williams, W.W. et al. Association testing of previously reported variants in a large case-control meta-analysis of diabetic nephropathy. Diabetes 61, 2187-94 (2012). Sandholm, N. et al. New susceptibility loci associated with kidney disease in type 1 diabetes. PLoS genetics 8, e1002921 (2012). Page 37 of 68 1372 1373 1374 1375 1376 1377 1378 1379 1380 1381 1382 1383 1384 1385 1386 1387 1388 1389 1390 1391 1392 1393 1394 1395 1396 1397 1398 1399 1400 1401 1402 1403 1404 1405 1406 1407 1408 1409 1410 1411 1412 1413 1414 1415 1416 1417 1418 1419 1420 1421 85. 86. 87. 88. 89. Li, Q., Brown, J.B., Huang, H. & Bickel, P.J. Measuring reproducibility of high-throughput experiments. The Annals of Applied Statistics 5, 17521779 (2011). Willer, C. et al. Discovery and Refinement of Loci Associated with Lipid Levels. Submitted (2013). Feng, J., Liu, T., Qin, B., Zhang, Y. & Liu, X.S. Identifying ChIP-seq enrichment using MACS. Nature protocols 7, 1728-40 (2012). Abecasis, G.R. et al. An integrated map of genetic variation from 1,092 human genomes. Nature 491, 56-65 (2012). Pruim, R.J. et al. LocusZoom: regional visualization of genome-wide association scan results. Bioinformatics 26, 2336-7 (2010). Supplementary Information is linked to the online version of the paper at www.nature.com/nature. Acknowledgments A full list of acknowledgments can be found in the Supplementary Information. Academy of Finland; Agence Nationale de la Recherche; Agency for Science, Technology and Research of Singapore (A*STAR); Althingi (the Icelandic Parliament); Ardix Medical; Association Diabète Risque Vasculaire; AstraZenec; Augustinus Foundation; Australian National Health and Medical Research Council; Australian Research Council; Avera Institute; Bayer Diagnostics; Becton Dickinson; Biobanking and Biomolecular Resources Research Infrastructure; Biotechnology and Biological Sciences Research Council; Boehringer Ingelheim Foundation; Boston University School of Medicine; British Heart Foundation; Bristol-Myers Squibb; Cancer Research UK; Cardionics; Canadian Institutes of Health Research; Cavadis B.V.; Celiac Disease Consortium; Center for Medical Systems Biology ; Center of Excellence in Genomics (EXCEGEN); Central Norway Health Authority; Centre for Medical Systems Biology; Centre of Excellence Baden-Wuerttemberg; Chief Scientist Office of the Scottish Government; City of Kuopio and Social Insurance Institution of Finland; Clinic Department of Dermatology, University Hospital Essen; CNAMTS ("Caisse Nationale de l’Assurance Maladie des Travailleurs Salariés"); Commission of the European Communities; CVON; Danish National Research Foundation; Danish Pharmacists’ Fund; deCODE Genetics; Deutsche Forschungsgemeinschaft; Diabetes Hilfs- und Forschungsfonds Deutschland; Diabetes UK; Directorate C - Public Health and Risk Assessment; Directorate C-Public Health; Dresden University of Technology; Dutch Brain Foundation; Dutch Diabetes Research Foundation; Dutch Digestive Disease Foundation; Dutch Government; Dutch Inter University Cardiology Institute Netherlands; Dutch Kidney Foundation; Dutch Ministry of Justice; Economic Structure Enhancing Fund (FES) of the Dutch government; Egmont Foundation; Emil Aaltonen Foundation; Erasmus University; Estonian Government; European Commission Framework Programme 6; European Community's Seventh Framework Programme; European Network for Genetic and Genomic Epidemiology; European Science Council; European Science Foundation; European Special Populations Research Network; European Union framework program 6 EUROSPAN project; European Union Ingenious HyperCare Consortium; Faculty of Biology and Medicine of Lausanne, Switzerland; Federal Ministry of Education and Research (BMBF); Federal Ministry of Education and Research (German Obesity Biomaterial Bank); Fédération Française de Cardiologie; French Ministry of Research; Finland’s Slottery Machine Association; Finnish Academy SALVE program ‘‘Pubgensense’’; Finnish Diabetes Association; Finnish Diabetes Research Page 38 of 68 1422 1423 1424 1425 1426 1427 1428 1429 1430 1431 1432 1433 1434 1435 1436 1437 1438 1439 1440 1441 1442 1443 1444 1445 1446 1447 1448 1449 1450 1451 1452 1453 1454 1455 1456 1457 1458 1459 1460 1461 1462 1463 1464 1465 1466 1467 1468 1469 1470 1471 1472 Foundation; Finnish Foundation of Cardiovascular Research and Finnish Cultural Foundation; Finnish Funding Agency for Technology and Innovation; Finnish Medical Society; Finnish National Public Health Institute; Finska Läkaresällskapet; Flemish League against Cancer; Folkhälsan Research Foundation; Fondation de France; Foundation against Cancer; Foundation for Life and Health in Finland; Foundation for Strategic Research and the Stockholm County Council; Foundation Heart and Arteries; French Research Agency; GlaxoSmithKline; G. Ph. Verhagen Foundation; Geestkracht program of the Netherlands Organization for Health Research and Development; Genetic Association Information Network; GenomEUtwin; German Diabetes Association; German Federal Ministry of Education and Research; German National Genome Research Network ; German Research Council; The Great Wine Estates of the Margaret River region of Western Australia; Healthway; Greek General Secretary of Research and Technology; Gyllenberg Foundation; Health & Consumer Protection; Health Care Centers in Vasa; Helmholtz Zentrum München - German Research Center for Environmental Health; Helsinki University Central Hospital Research Funds (EVO); Hjartavernd (the Icelandic Heart Association); hospital districts of Pirkanmaa, South Ostrobothnia, and Central Finland; HYPERGENES Consortium; Innovation-Oriented Research Program on Genomics; INSERM ("Réseaux en Santé Publique Interactions entre les determinants de la santé"); INTEROMICS (MIUR - CNR Italian Flagship Project); Interuniversity Cardiology Institute of the Netherlands; Italian Ministry of Health; Juho Vainio Foundation; Juselius Foundation; Juvenile Diabetes Research Foundation; Juvenile Diabetes Research Foundation International (JDRF); King's College London; Knut and Alice Wallenberg Foundation; Kuopio University Hospital from Ministry of Health and Social Affairs; Leo Laboratories; Medical Research Council (MRC); Lilly; Liv och Hälsa Foundation; Louis-Jeantet Foundation; Lundberg Foundation; March of Dimes Birth Defects Foundation; Medical Research Council of Canada; Medical Research Council, UK; Medical Research Foundation of Umeå University; Merck Santé; Merck Sharp and Dohme-Chibret Laboratory; Ministry for Health, Welfare and Sports; Ministry of Cultural Affairs; Ministry of Economic Affairs; Ministry of Education; Ministry of Education and Culture of Finland; Ministry of Education, Culture and Science; Ministry of Health of the Republic of Seychelles; Ministry of Science, Education and Sport of the Republic of Croatia; Ministry of Social Affairs and Health in Finland; MRC Centre for Obesity and Related Metabolic Diseases; MRC Human Genetics Unit, Arthritis Research UK; MRC-GlaxoSmithKline; Munich Center of Health Sciences; Municipal Health Care Center and Hospital in Jakobstad; Municipality of Rotterdam; Närpes and Korsholm; Närpes Health Care Foundation; National Alliance for Research on Schizophrenia and Depression; National Cancer Institute; National Center for Advancing Translational Sciences; National Center for Research Resources; National Human Genome Research Institute (NHGRI); National Heart, Lung, and Blood Institute (NHLBI); National Institute for Health Research (NIHR); National Institute of Allergy and Infectious Diseases (NIAID); National Institute on Aging; National Institute of Child Health and Human Development (NICHD); National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK); National Institutes of Health (NIH); National Institutes of Mental Health; National Institute of Neurological Disorders and Stroke (NINDS); National Research Initiative; Netherlands Consortium for Healthy Aging; Netherlands Genomics Initiative; Netherlands Organisation for Health Research and Development; Netherlands Organization for Scientific Research; Neuroscience Campus Amsterdam; NIA Intramural Research Program; NIH Roadmap for Medical Research; Nordic Center of Excellence in Disease Genetics; Nordic Centre of Excellence on Systems; Nord-Trøndelag County Council; Northern Ireland Research Development; Northern Netherlands Collaboration of Provinces (SNN); Norwegian Institute of Public Health; Norwegian Research Council; Page 39 of 68 1473 1474 1475 1476 1477 1478 1479 1480 1481 1482 1483 1484 1485 1486 1487 1488 1489 1490 1491 1492 1493 1494 1495 1496 1497 1498 1499 1500 1501 1502 1503 1504 1505 1506 1507 1508 1509 1510 1511 1512 1513 1514 1515 1516 1517 1518 1519 1520 1521 1522 1523 Norwegian University of Science and Technology; Novartis Pharma; Novo Nordisk; Novo Nordisk Foundation; Ollqvist Foundation; Onivins; Orion-Farmos Research Foundation; Paavo Nurmi Foundation; Päivikki and Sakari Sohlberg Foundation; Paul Michael Donovan Charitable Foundation; Perklén Foundation; Pfizer; Pierre Fabre; Province of Groningen, University Medical Center Groningen, the University of Groningen; Radboud University Medical Centre, Nijmegen; Research Institute for Diseases in the Elderly; Reynold's Foundation; Roche; Royal Swedish Academy of Science; sanofi-aventis; Science for Life Laboratory – Uppsala and the Swedish Society for Medical Research; Science Foundation Ireland; Servier Research Group; Siemens Healthcare, Erlangen; Signe and Ane Gyllenberg Foundation; Sigrid Juselius Foundation; 6th Framework Program of the European Union; Social Insurance Institution of Finland; Social Ministry of the Federal State of Mecklenburg-West Pomerania; South Tyrolean Sparkasse Foundation; Stanford University; State of Bavaria; Stockholm County Council; Strategic Cardiovascular Program of Karolinska Institutet and Stockholm County Council; Strategic Cardiovascular Programme of Karolinska Institutet; Stroke Association; Susan G. Komen Breast Cancer Foundation; Société Francophone du Diabète; Swedish Cancer Society; Swedish Cultural Foundation in Finland; Swedish Diabetes Association; Swedish Foundation for Strategic Research; Swedish Heart-Lung Foundation; Swedish Research Council; Swedish Medical Research Council; Swedish Society of Medicine; Swiss National Science Foundation; SYSDIET; Tampere and Turku University Hospital; Tampere Tuberculosis Foundation; Tekes; The Andrea and Charles Bronfman Philanthropies; The Finnish Diabetes Research Foundation; Topcon; Torsten and Ragnar Söderberg Foundation; UK Medical Research Council; United Kingdom NIHR Cambridge Biomedical Research Centre; University and Research of the Autonomous Province of Bolzano; University Hospital Oulu, Biocenter, University of Oulu; University of Geneva; University of Maryland General Clinical Research Center; University of Tartu; University of Ulm; USDA National Institute of Food and Agriculture; VU University’s Institute for Health and Care Research; Wilhelm and Else Stockmann Foundation; Yrjö Jahnsson Foundation; Zorg Onderzoek Nederland-Medische Wetenschappen; Wellcome Trust. Author Contributions Steering Committee Overseeing the Consortium Gonçalo R. Abecasis, Themistocles Assimes, Ines Barroso, Sonja I. Berndt, Michael Boehnke, Ingrid Borecki, Panagiotis Deloukas, Caroline S. Fox, Timothy M. Frayling, Leif Groop, Iris M. Heid, Joel N. Hirschhorn, David Hunter, Erik Ingelsson, Robert Kaplan, Ruth J.F. Loos, Mark I. McCarthy, Karen L. Mohlke, Kari E. North, Jeffrey R. O'Connell, David Schlessinger, David Strachan, Unnur Thorsteinsdottir, Cornelia M. van Duijn Writing Group Ines Barroso, Jaques S. Beckmann, Sonja I. Berndt, Martin L. Buchkovich, Damien C. Croteau-Chonka, Felix R. Day, Stefan Gustafsson, Joel N. Hirschhorn, Erik Ingelsson, Anne E. Justice, Bratati Kahali, Cecilia M. Lindgren, Adam E. Locke, Ruth J.F. Loos, Karen L. Mohlke, Kari E. North, Tune H. Pers, Corey Powell, André Scherag, Elizabeth K. Speliotes, Sailaja Vedantam, Cristen J. Willer Data Cleaning and Preparation Damien C. Croteau-Chonka, Felix R. Day, Tonu Esko, Tove Fall, Teresa Ferreira, Stefan Gustafsson, Zoltán Kutalik, Adam E. Locke, Jian'an Luan, Reedik Mägi, Joshua C. Randall, André Scherag, Sailaja Vedantam, Thomas W. Winkler, Andrew R. Wood, Tsegaselassie Workalemahu Page 40 of 68 1524 1525 1526 1527 1528 1529 1530 1531 1532 1533 1534 1535 1536 1537 1538 1539 1540 1541 1542 1543 1544 1545 1546 1547 1548 1549 1550 1551 1552 1553 1554 1555 1556 1557 1558 1559 1560 1561 1562 1563 1564 1565 1566 1567 1568 1569 1570 1571 1572 1573 1574 GWAS Look-ups in Other Consortia (ADIPOGen Consortium) Zari Dastani, ADIPOGen Consortium; (CARDIOGRAMplusC4D) CARDIOGRAMplusC4D, Panos Deloukas, Stavroula Kanoni, Sekar Kathiresan; (ENDOMETRIOSIS GWAS) Grant W. Montgomery, Dale R. Nyholt, Krina T. Zondervan, The International Endogene Consortium; (FinnDiane/GENIE) Niina Sandholm; (GENIE) Eoin P. Brennan, Amy Jayne McKnight, Rany M. Salem; (GENIE look up) The GENIE Consortium; (GLOBAL LIPIDS look up) The GLGC, (ICBP look up) The IBPC; (CNV) Robert E. Handsaker, Steven A. McCarroll; (IgA Nephropathy) Krzysztof Kiryluk, Richard P. Lifton; (MAGIC look up) Robert A. Scott, MAGIC (MetaAnalyses of Glucose and Insulin-Related Traits Consortium) investigators; (ReproGen) Joanne M. Murabito, John R.B. Perry, Lisette Stolk, The ReproGen Consortium; (CKDGen) CKDGen Consortium Gene Expression (eQTL) Analyses (Brain Eqtl) Ruth J.F. Loos, Jing Hua Zhao; (EGCUT) Tonu Esko, Andres Metspalu, Eva Reinmaa; (eQTL Liver/Omental/Subq eSNPs) Eric E. Schadt; (MolOBB) Alexander Werner Drong, Fredrik Karpe, Josine L. Min, George Nicholson; (MuTHER) Åsa K. Hedman, Sarah Keildson, MuTHER Consortium Other Analyses and Contributions (Health and Retirement Study) Wei Zhao, Jennifer A. Smith, Jessica D. Faul, David R. Weir; (DEPICT) Rudolf Fehrmann, Lude Franke, Joel N. Hirschhorn; Juha Karjalainen, Tune H. Pers; (ENCODE) Martin L. Buchkovich, Jin Chen, Ellen M. Schmidt, Cristen J. Willer; (QIMR cohort) Michael E. Goddard, Anna A.E. Vinkhuyzen, Peter M. Visscher, Jian Yang Project Design, Management and Coordination of Contributing Studies METABOCHIP STUDIES (ADVANCE) Themistocles L. Assimes, Joshua W. Knowles, Thomas Quertermous; (AMCPAS) John Kastelein, Panos Deloukas; (ARIC Metabochip) Eric Boerwinkle, Kari E. North;(B1958C) Elina Hypponen, Chris Power; (BHS MC) John Beilby, Jennie Hui; (CARDIOGENICS) Panos Deloukas; (CLHNS) Linda S. Adair, Karen L. Mohlke; (DESIR) Stéphane Cauchi, Philippe Froguel; (DIAGEN) Stefan R. Bornstein, Peter E.H. Schwarz; (DILGOM) Pekka Jousilahti, Antti M. Jula, Satu Männistö, Markus Perola, Veikko Salomaa;(DPS) Matti Uusitupa; (DR's EXTRA) Timo A. Lakka, Rainer Rauramaa; (Dundee – GoDarts) Colin Neil Alexander Palmer; (EAS) Jackie F. Price; (EGCUT) Andres Metspalu; (ELY) Nita G. Forouhi, Claudia Langenberg, Ruth J.F. Loos, Ken K. Ong, Robert A. Scott, Nicholas J. Wareham; (EMIL (SWABIA)) Bernhard O. Boehm; (EPIC-Norfolk) Nita G. Forouhi, Claudia Langenberg, Ruth J.F. Loos, Ken K. Ong, Robert A Scott, Nicholas J Wareham;(FBPP) Aravinda Chakravarti, Richard Cooper, Steven C. Hunt;(Fenland) Nita G. Forouhi, Claudia Langenberg, Ruth J.F. Loos, Ken K. Ong, Robert A. Scott, Nicholas J. Wareham; (FIN-D2D 2007) Sirkka M. KeinanenKiukaanniemi, Timo E. Saaristo; (FUSION stage 2) Francis S. Collins, Jouko Saramies, Jaakko Tuomilehto;(GLACIER) Paul W. Franks; (GxE) Richard S. Cooper, Joel N. Hirschhorn, Colin A. McKenzie; (HNR) Raimund Erbel, Karl-Heinz Jöckel, Susanne Moebus; (HUNT 2) Kristian Hveem; (IMPROVE) Ulf de Faire, Anders Hamsten, Steve Humphries, Elena Tremoli; (KORA S3 (MetaboChip)) Iris M. Heid, Annette Peters, Konstantin Strauch, H.-Erich Wichmann; (Leipzig adults) Michael Stumvoll; (LURIC), Winfried März; (MEC Metabochip) Christopher Haiman, Loic Le Marchand; (METSIM) Johanna Kuusisto, Markku Laakso; (MORGAM) Philippe Amouyel, Dominique Arveiler, Page 41 of 68 1575 1576 1577 1578 1579 1580 1581 1582 1583 1584 1585 1586 1587 1588 1589 1590 1591 1592 1593 1594 1595 1596 1597 1598 1599 1600 1601 1602 1603 1604 1605 1606 1607 1608 1609 1610 1611 1612 1613 1614 1615 1616 1617 1618 1619 1620 1621 1622 1623 1624 Giancarlo Cesana, Jean Ferrières, David-Alexandre Trégouët, Jarmo Virtamo; (MRC NSHD) Diana Kuh; (PIVUS) Erik Ingelsson; (PROMIS) John Danesh, Panos Deloukas, Danish Saleheen; (SardiNIA) Gonçalo R. Abecasis, David Schlessinger; (ScarfSheep) Ulf de Faire, Anders Hamsten; (SPT) Richard S. Cooper, Joel N. Hirschhorn, Colin A. McKenzie; (STR) Erik Ingelsson; (Tandem) Murielle Bochud, Pascal Bovet; (THISEAS) George Dedoussis, Panos Deloukas; (Tromsø) Inger Njølstad; (ULSAM) Erik Ingelsson; (WHI Metabochip) Charles Kooperberg, Ulrike Peters; (Whitehall) Aroon D. Hingorani, Mika Kivimaki, Nick Wareham; (WTCCC-T2D) Mark I. McCarthy, Cecilia M. Lindgren; (DietGeneExpression (DGE)) Berit Johansen NEW GWAS (All LOLIPOP Studies) John C. Chambers, Jaspal S. Kooner; (ASCOT) Mark J. Caulfield, Peter Sever; (Athero-Express Biobank Studies) Folkert W. Asselbergs, Hester M. de Ruijter, Frans L. Moll, Gerard Pasterkamp; (Busselton Health Study) John Beilby, Jennie Hui; (COROGENE) Markus Perola, Juha Sinisalo; (DESIR) Stéphane Cauchi, Philippe Froguel; (DNBC) Mads Melbye, Jeffrey C. Murray; (EGCUT) Andres Metspalu; (Emerge) M. Geoffrey Hayes; (ERF) Ben A. Oostra, Cornelia M. van Duijn; (FamHS) Ingrid B. Borecki; (FINGESTURE) John D. Rioux; (GOOD) Claes Ohlsson; (HBCS) Johan G Eriksson; (Health ABC) Tamara B. Harris, Yongmei Liu; (HERITAGE Family Study) Claude Bouchard, D.C. Rao, Mark A. Sarzynski; (HYPERGENES) Daniele Cusi; (IPM BioMe) Erwin P. Bottinger, Ruth J.F. Loos; (LifeLines) The Lifelines Cohort Study; (LLS) P. Eline Slagboom; (MGS) Pablo V. Gejman; (NELSON) Paul I.W. de Bakker, Pieter Zanen; (PLCO2) Sonja I. Berndt, Stephen J. Chanock; (PREVEND) Pim van der Harst; (PROCARDIS) Martin Farrall, Hugh Watkins; (PROSPER/PHASE) Ian Ford, J. Wouter Jukema, Naveed Sattar; (QFS) Claude Bouchard, André Marette, Louis Pérusse, Angelo Tremblay, Marie-Claude Vohl; (QIMR Polygene) Heath C. Andrew, Nicholas G. Martin, Madden A.F. Pamela; (RISC) Timothy M. Frayling, Mark Walker; (RSII) Oscar H. Franco, Albert Hofman, Fernando Rivadeneira, André G. Uitterlinden, Cornelia M. van Duijn, Jacqueline C. Witteman, M. Carola Zillikens; (RSIII) Oscar H. Franco, Albert Hofman, Fernando Rivadeneira, André G. Uitterlinden, Cornelia M. van Duijn, Jacqueline C. Witteman, M. Carola Zillikens; (SHIP-TREND) Henri Wallaschofski; (Sorbs) Anke Tönjes; (TRAILS) Albertine J. Oldehinkel, Harold Snieder; (TWINGENE) Erik Ingelsson; (TwinsUK) Tim D. Spector; (WGHS) Paul M. Ridker PREVIOUS GWAS (AGES) Vilmundur Gudnasson, Tamara B. Harris; (Amish) Alan R. Shudiner; (ARIC GWAS) Kari E. North; (B58C T1D CONTROLS) David P. Strachan; (B58C WTCCC) David P. Strachan; (BRIGHT) Anna F. Dominiczak, Martin Farrall; (CAPS) Erik Ingelsson; (COLAUS) Gérard Waeber, Dawn Waterworth; (CROATIA-Vis) Igor Rudan; (deCODE) Kari Stefansson, Unnur Thorsteinsdottir; (DGI) Leif C. Groop; (EGCUT) Andres Metspalu; (EPIC-Norfolk) Jing Hua Zhao; (Fenland) Nicholas J. Wareham; (Finnish Twin Cohort) Jaakko Kaprio; (FRAM) L. Adrienne Cupples; (FUSION (GWAS)) Richard N. Bergman, Michael Boehnke; (GerMIFS I) Jeanette Erdmann, Christian Hengstenberg, Heribert Schunkert; (Health 2000) Paul Knekt; (HPFS) David Hunter; (KORA S4 (GWA)) Christian Gieger; (MICROS) Andrew A. Hicks, Peter P. Pramstaller; (NFBC66) Marjo-Riitta Jarvelin; (NHS) David Hunter; (NSPHS) Ulf Gyllensten; (ORCADES) Harry Campbell; (PLCO) Sonja I. Berndt, Stephen J. Chanock; (RSI) Oscar H. Franco, Albert Hofman, Fernando Rivadeneira, André G. Uitterlinden, Cornelia M. van Duijn, Jacqueline C. Witteman, M. Carola Zillikens; (RUNMC) Lambertus A. Kiemeney; (SASBAC) Erik Ingelsson; (SHIP) Henri Wallaschofski; (WTCCC-CAD) Alistair S. Hall, Page 42 of 68 1625 1626 1627 1628 1629 1630 1631 1632 1633 1634 1635 1636 1637 1638 1639 1640 1641 1642 1643 1644 1645 1646 1647 1648 1649 1650 1651 1652 1653 1654 1655 1656 1657 1658 1659 1660 1661 1662 1663 1664 1665 1666 1667 1668 1669 1670 1671 1672 1673 1674 Nilesh J. Samani; (WTCCC-T2D) Mark I. McCarthy, Cecilia Lindgren; (Young Finns Study (YFS)) Terho Lehtimäki, Olli T. Raitakari Genotyping of Contributing Studies METABOCHIP STUDIES (ADVANCE) Devin Absher, Themistocles L. Assimes, Joshua W. Knowles, Thomas Quertermous; (AMCPAS) Kathleen Stirrups; (ARIC Metabochip) Eric Boerwinkle, Kari E. North; (B1958C) Neil R. Robertson, Christopher J. Groves, Thorhildur Juliusdottir; (BHS MC) Gillian M. Arscott, Jennie Hui; (CARDIOGENICS) Kathleen Stirrups; (CLHNS) Damien C. Croteau-Chonka; (DESIR) Elodie Eury, Stéphane LOBBENS; (DIAGEN) Amy J. Swift; (Dundee – GoDarts) Nigel William Rayner, Amanda J. Bennett, Colin Neil Alexander Palmer; (EAS) James F. Wilson; (EGCUT) Tõnu Esko, Lili Milani; (ELY) Claudia Langenberg, Ruth J.F. Loos, Ken K. Ong, Nicholas J. Wareham; (EMIL (SWABIA)) Bernhard O. Boehm; (EPIC-Norfolk) Claudia Langenberg, Ruth J.F. Loos, Ken K. Ong, Nicholas J. Wareham; (FBPP) Aravinda Chakravarti; (Fenland) Claudia Langenberg, Ruth J.F. Loos, Ken K. Ong, Nicholas J. Wareham; (FIN-D2D 2007) Peter S. Chines; (FUSION stage 2) Leena Kinnunen; (GLACIER) Ines Barroso; (HNR) Markus M. Noethen; (HUNT 2) Mario A. Morken; (KORA S4 (MetaboChip)), Harald Grallert,Peter Lichtner; (Leipzig adults) Yvonne Böttcher, Peter Kovacs; (LURIC) Marcus E. Kleber; (MEC Metabochip), Christopher Haiman; (MRC NSHD) Diana Kuh, Ken K. Ong, Andrew Wong; (PIVUS) Christian Berne, Erik Ingelsson, Lars Lind, Johan Sundström, Kathleen Stirrups; (SardiNIA) Ramaiah Nagaraja, Serena Sanna; (ScarfSheep) Bruna Gigante; (STR) Nancy L. Pedersen; (Tandem) Georg B. EHRET, François Mach; (THISEAS) Kathleen Stirrups; (Tromsø) Lori L Bonnycastle; (ULSAM) Johan Ärnlöv, Erik Ingelsson, Ann-Christine Syvänen; (WHI Metabochip), Charles Kooperberg, Ulrike Peters; (Whitehall) Claudia Langenberg; (WTCCC-T2D) Mark I. McCarthy, Andrew Tym Hattersley; (DietGeneExpression (DGE)) Berit Johansen NEW GWAS (All LOLIPOP Studies) John C. Chambers Jaspal S. Kooner; (ASCOT) Patricia B. Munroe; (Athero-Express Biobank Study) Sander W. van der Laan; (Busselton Health Study) John Beilby, Jennie Hui; (DESIR) Elodie EURY, Stéphane LOBBENS; (EGCUT) Tõnu Esko, Lili Milani; (Emerge) Dana C. Crawford, M. Geoffrey Hayes; (ERF) Aaron Isaacs, Ben A. Oostra, Cornelia M. van Duijn; (FamHS) Ingrid B. Borecki, Warwick E. Daw, Mary F. Feitosa, Aldi T. Kraja, Mary K. Wojczynski, Qunyuan Zhang; (GOOD) Claes Ohlsson; (Health ABC) Yongmei Liu; (HERITAGE Family Study) Mark A. Sarzynski; (IPM BioMe) Erwin P. Bottinger; (LifeLines) Morris A. Swertz, The LifeLines Cohort Study; (LLS) Quinta Helmer; (MGS) Pablo V. Gejman; (NELSON) Joanna Smolonska; (PLCO2) Stephen J. Chanock, Kevin B. Jacobs, Zhaoming Wang; (PREVEND) Folkert W. Asselbergs, Irene Mateo Leach, Pim van der Harst; (PROCARDIS) John F. Peden; (PROSPER/PHASE) J. Wouter Jukema, P. Eline Slagboom, Stella Trompet; (QFS) Claire Bellis, John Blangero; (RSII) Karol Estrada, Fernando Rivadeneira, André G. Uitterlinden; (RSIII) Karol Estrada, Fernando Rivadeneira, André G. Uitterlinden; (SHIP-TREND) Georg Homuth, Uwe Völker; (TRAILS) Marcel Bruinenberg, Catharina A. Hartman; (TWINGENE) Anders Hamsten, Nancy L. Pedersen; (TwinsUK) Massimo Mangino, Alireza Moayyeri; (WGHS) Daniel I. Chasman, Lynda M. Rose; PREVIOUS GWAS Page 43 of 68 1675 1676 1677 1678 1679 1680 1681 1682 1683 1684 1685 1686 1687 1688 1689 1690 1691 1692 1693 1694 1695 1696 1697 1698 1699 1700 1701 1702 1703 1704 1705 1706 1707 1708 1709 1710 1711 1712 1713 1714 1715 1716 1717 1718 1719 1720 1721 1722 1723 1724 1725 (AGES) Albert Vernon Smith; (Amish) Jeffrey R. O'Connell; (B58C T1D CONTROLS) Wendy L. McArdle; (B58C WTCCC) Wendy L. McArdle; (BRIGHT) Martin Farrall; (CAPS) Henrik Grönberg; (COLAUS) Dawn Waterworth; (CROATIA-Vis) Caroline Hayward; (EGCUT) Mari Nelis; (Fenland) Nicholas J. Wareham; (Finnish Twin Cohort) Jaakko Kaprio; (KORA S3 (GWA)) Thomas Illig; (KORA S4 (GWA)) Martina MüllerNurasid; (MICROS) Andrew A. Hicks; (NFBC66) Marjo-Riitta Jarvelin; (ORCADES) Alan F. Wright; (PLCO) Stephen J. Chanock; (RSI) Karol Estrada, Fernando Rivadeneira, André G. Uitterlinden; (SASBAC) Per Hall; (SHIP) Georg Homuth, Uwe Völker; (WTCCC-CAD) Alistair S. Hall, Nilesh J. Samani; (WTCCC-T2D) Mark I. McCarthy, Andrew Tym Hattersley; (Young Finns Study (YFS)) Terho Lehtimäki, Olli T. Raitakari Phenotype Coordination of Contributing Studies METABOCHIP STUDIES (ADVANCE) Alan S Go, Thomas Quertermous; (AMC-PAS) Kees G Hovingh; (ARIC Metabochip) Eric Boerwinkle; (B1958C) Elina Hypponen, Chris Power; (BHS MC) Alan L James, Arthur Willian (Bill) Musk; (CARDIOGENICS) Alison H Goodall, Christian Hengstenberg; (CLHNS) Isabelita N Bas, Nanette R Lee; (DESIR) Gaëlle Gusto; (DIAGEN) Jürgen Gräßler, Gabriele Müller; (DPS) Jaana Lindström; (DR's EXTRA) Maija Hassinen; (Dundee – GoDarts) Andrew David Morris, Colin Neil Alexander Palmer, Alex Surendra Fleetwood DoneyEAS, Stela McLachlan; (EGCUT) Tõnu Esko, Andres Metspalu; (ELY) Nita G Forouhi, Nicholas J Wareham; (EMIL (SWABIA)), Roza Blagieva,Bernhard O Boehm,Wolfgang Kratzer, Sigrun Merger, Thomas Seufferlein, Koenig Wolfgang; (EPIC-Norfolk) Nita G Forouhi, Nicholas J Wareham (FBPP), Richard Cooper, Steven C Hunt; (Fenland) Nita G Forouhi, Nicholas J Wareham; (GLACIER) Goran Hallmans; (GxE) Terrence Forrester, Bamidele O Tayo; (HNR) Raimund Erbel, Karl-Heinz Jöckel, Susanne Moebus; (HUNT 2) Oddgeir Holmen; (KORA S3 (MetaboChip)) Wolfgang Koenig, Barbara Thorand, Annette Peters, H.-Erich Wichmann; (Leipzig adults) Matthias Blüher; (MEC Metabochip) Lynne Wilkens; (METSIM) Heather M Stringham; (MRC NSHD) Diana Kuh; (PIVUS) Christian Berne, Erik Ingelsson, Lars Lind, Johan Sundström; (PROMIS) Danish Saleheen; (SardinNIA) Antonella Mulas; (ScarfSheep) Karin Leander; (SPT) Terrence Forrester, Bamidele O Tayo, Nancy L Pedersen; (Tandem) Murielle Bochud, Pascal Bovet; (THISEAS) Maria Dimitriou; (Tromsø) Tom Wilsgaard; (ULSAM) Johan Ärnlöv, Vilmantas Giedraitis, Erik Ingelsson; (WHI Metabochip) Charles, Ulrike Peters; (Whitehall) Meena Kumari; (WTCCC-T2D) Andrew Tym Hattersley; (DietGeneExpression (DGE)) Ida H Caspersen, Berit Johansen NEW GWAS (All LOLIPOP Studies) John C Chambers, Jaspal S Kooner, William R Scott, Sian-Tsung Tan; (ASCOT) Mark J Caulfield, Peter Sever, Alice V Stanton; (Athero-Express Biobank Study) Frans L. Moll; (Busselton Health Study) John Beilby, Jennie Hui; (DESIR) Gaëlle Gusto; (DNBC) Heather Allison Boyd, Bjarke Feenstra, Frank Geller; (EGCUT) Tõnu Esko, Andres Metspalu; (Emerge) Josh C. Denny, Abel N. Kho; (ERF) Ben A Oostra, Cornelia M van Duijn; (FamHS) Ingrid B Borecki, Mary F Feitosa; (GOOD) Claes Ohlsson, Liesbeth Vandenput; (Health ABC) Melissa E Garcia, Tamara B Harris, Michael A Nalls; (HBCS) Johan G Eriksson; (HERITAGE Family Study) Claude Bouchard; (HYPERGENES) Daniele Cusi; (IPM BioMe) Omri Gottesman; (LifeLines) Salome Scholtens, Morris A Swertz, Judith M Vonk, The LifeLines Cohort Study; (LLS) Anton JM de Craen; (MGS) Pablo V. Gejman; (NELSON) Dirkje S. Postma; (PLCO2) Sonja I Berndt; (PREVEND) Stephan JL Bakker, Ron T Gansevoort; (PROCARDIS) Robert Clarke, Anders Hamsten; (PROSPER/PHASE) Anton JM de Craen, Ian Ford, J Wouter Jukema, Naveed Sattar; (QFS) Claude Bouchard, Angelo Trembay; (QIMR Page 44 of 68 1726 1727 1728 1729 1730 1731 1732 1733 1734 1735 1736 1737 1738 1739 1740 1741 1742 1743 1744 1745 1746 1747 1748 1749 1750 1751 1752 1753 1754 1755 1756 1757 1758 1759 1760 1761 1762 1763 1764 1765 1766 1767 1768 1769 1770 1771 1772 1773 1774 1775 1776 Polygene) Heath C. Andrew, Nicholas G. Martin, Madden A.F. Pamela; (RSII) Oscar H. Franco; Albert Hofman, Fernando Rivadeneira, André G. Uitterlinden, Cornelia M. van Duijn, Jacqueline C. Witteman; (RSIII) Oscar H. Franco, Albert Hofman, Fernando Rivadeneira, André G. Uitterlinden, Cornelia M. van Duijn, Jacqueline C. Witteman; (SHIP-TREND) Stephan B. Felix, Hans-Jörgen Grabe, Roberto Lorbeer, Rainer Rettig; (Sorbs) Anke Tönjes; (TRAILS) Catharina A Hartman, Ronald P Stolk, Floor V Van Oort; (TWINGENE) Patrik KE Magnusson, Nancy L Pedersen; (TwinsUK) Massimo Mangino, Cristina Menni; (WGHS) Daniel I. Chasman, Lynda M. Rose PREVIOUS GWAS (Amish) Alan R Shudiner; (B58C T1D CONTROLS) David P Strachan; (B58C WTCCC) David P Strachan; (BRIGHT) Anna F Dominiczak; (CAPS) Henrik Grönberg; (CHS) YiiDer Ida Chen; (COLAUS) Gérard Waeber, Dawn Waterworth; (CROATIA-Vis) Igor Rudan; (DGI) Valeriya Lyssenko; (EGCUT) Andres Metspalu; (Fenland) Nicholas J Wareham; (Finnish Twin Cohort) Jaakko Kaprio, Markku Koskenvuo; (NFBC66) MarjoRiitta Jarvelin, Jaana Laitinen; (NTRNESDA) Gonneke Willemsen; (ORCADES) Alan F Wright; (PLCO) Sonja I Berndt; (RSI) Oscar H. Franco, Albert Hofman, Fernando Rivadeneira, André G. Uitterlinden, Cornelia M. van Duijn, Jacqueline C. Witteman; (SASBAC) Per Hall; (SHIP) Stephan B. Felix, Hans-Jörgen Grabe, Roberto Lorbeer, Rainer Rettig; (UKBS-CC) Jennifer Jolley; (WTCCC-CAD) Alistair S Hall, Nilesh J Samani; (WTCCC-T2D) Andrew Tym Hattersley; (Young Finns Study (YFS)) Terho Lehtimäki, Olli T Raitakari Data Analysis METABOCHIP STUDIES (ADVANCE) Devin Absher, Themistocles L. Assimes, Lindsay L. Waite; (AMCPAS) Stavroula Kanoni; (ARIC Metabochip) Steven Buyske, Anne E. Justice, Kari E. North; (B1958C) Teresa Ferreira; (BHS MC) Denise Anderson; (CARDIOGENICS) Stavroula Kanoni; (CLHNS) Damien C. Croteau-Chonka; (DESIR) Stéphane Cauchi, Loïc YENGO; (DGE DietGeneExpression) Ida H. Caspersen; (DIAGEN) Anne U. Jackson, Gabriele Müller; (DILGOM) Kati Kristiansson; (Dundee – GoDarts) Teresa Ferreira; (EAS) Jennifer L. Bolton, Ross M. Fraser; (EGCUT) Tõnu Esko, Krista Fischer, Evelin Mihailov; (ELY) Jian'an Luan; (EMIL (SWABIA)) Bernhard O. Boehm, Wolfgang Kratzer; (EPICNorfolk) Jian'an Luan; (FBPP) Aravinda Chakravarti, Georg B. Ehret; (Fenland) Jian'an Luan; (GLACIER) Frida Renstrom, Dmitry Shungin; (GxE) Cameron D Palmer; (HNR) Sonali Pechlivanis, André Scherag; (IMPROVE) Lasse Folkersen, Rona J. Strawbridge; (KORA S3 (MetaboChip)), Mathias Gorski, Janina S. Ried, Thomas W. Winkler; (KORA S4 (MetaboChip)) Eva Albrecht; (Leipzig adults) Anubha Mahajan, Inga Prokopenko; (LURIC) Graciela Delgado de Moissl, Tanja B. Grammer, Marcus E. Kleber, Stefan Pilz, Hubert Scharnagl; (MEC Metabochip) Unhee Lim, Fred Schumacher; (METSIM) Alena Stančáková; (MRC NSHD), Jian'an Luan, Andrew Wong; (PIVUS) Stefan Gustafsson, Erik Ingelsson; (PROMIS) Stavroula KanoniSardi; (SardiNIA) Jennifer L. BraggGresham; (ScarfSheep) Lasse Folkersen, Rona J Strawbridge; (SPT) Cameron D Palmer, Stefan Gustafsson, Erik Ingelsson; (Tandem) Georg B. EHRET, François Mach; (THISEAS) Stavroula Kanoni; (ULSAM) Stefan Gustafsson, Erik Ingelsson; (WHI Metabochip) Jian Gong, Jeffrey Haessler; (Whitehall) Jian'An Luan; (WTCCC-T2D) Andrew P. Morris, Teresa Ferreira, Anubha Mahajan, Reedik Mägi; NEW GWAS (Athero-Express Biobank Studies) Sander W. van der Laan; (DESIR) Stéphane Cauchi, Loïc YENGO; (DNBC) Bjarke Feenstra, Frank Geller; (EGCUT) Tõnu Esko, Krista Page 45 of 68 1777 1778 1779 1780 1781 1782 1783 1784 1785 1786 1787 1788 1789 1790 1791 1792 1793 1794 1795 1796 1797 1798 1799 1800 1801 1802 1803 1804 1805 1806 1807 1808 1809 1810 1811 Fischer, Toomas Haller, Reedik Mägi; (Emerge) M. Geoffrey Hayes; (ERF) Najaf Amin, Ayse Demirkan; (FamHS) Mary F. Feitosa; (FINGESTURE) Ken Sin Lo; (GOOD) Claes Ohlsson, Liesbeth Vandenput; (HBCS) Niina Eklund; (Health ABC) Michael A. Nalls; (HERITAGE Family Study) Claude Bouchard, Tuomo Rankinen, D.C. Rao, Treva Rice, Mark A. Sarzynski, Yun Ju Sung; (HYPERGENES) Daniele Cusi, Zoltán Kutalik; (InCHIANTI) Andrew R. Wood, Dorota Pasko; (IPM BioMe) Janina Jeff, Vaneet Lotay, Yingchang Lu; (LifeLines) Ilja M. Nolte, Jana V. Van Vliet-Ostaptchouk; (LLS) Marian Beekman, Stefan Böhringer, HaeWon Uh; (LOLIPOP) Guohong Deng, Weihua Zhang; (MGS) Jianxin Shi; (NELSON) Stephan Ripke, Jessica van Setten; (PLCO2) Sonja I. Berndt, Zhaoming Wang; (PREVEND) Irene Mateo Leach, Pim van der Harst, Niek Verweij; (PROCARDIS) Anuj Goel, John F. Peden; (PROSPER/PHASE) Anton J.M. de Craen, Ian Ford, Stella Trompet; (QFS) John Blangero, Louis Pérusse; (QIMR Polygene) Scott D. Gordon, Sarah E. Medland, Dale R. Nyholt; (RISC) Dorota Pasko, Andrew R. Wood; (RSII) Karol Estrada, Carolina Medina-Gomez, Marjolein Peters, Fernando Rivadeneira, André G. Uitterlinden; (RSIII) Karol Estrada, Carolina Medina-Gomez, Marjolein Peters, Fernando Rivadeneira, André G. Uitterlinden; (SHIP-TREND) Alexander Teumer; (Sorbs) Reedik Mägi; (TRAILS) Harold Snieder; (TWINGENE) Stefan Gustafsson, Erik Ingelsson; (TwinsUK) Massimo Mangino; (WGHS) Daniel I. Chasman, Lynda M. Rose 1812 Author Information 1813 G.T., V.S., U.T., and K.S. are employed by deCODE Genetics/Amgen, Inc. 1814 I.B. and spouse own stock in GlaxoSmithKline and Incyte, Ltd. C.B. is a 1815 consultant for Weight Watchers, Pathway Genomics, NIKE, and Gatorade 1816 PepsiCo. PREVIOUS GWAS (AGES) Albert Vernon Smith; (Amish) Jeffrey R. O'Connell; (ARIC GWAS) Keri L. Monda, Kari E. North; (B58C T1D CONTROLS) David P. Strachan; (B58C WTCCC) David P. Strachan; (CAPS) Erik Ingelsson; (CHS) Yii-Der Ida Chen, Barbara McKnight; (CROATIA-Vis) Caroline Hayward; (deCODE) Valgerdur Steinthorsdottir, Gudmar Thorleifsson; (EGCUT) Mari Nelis; (Fenland) Jian'an Luan; (FRAM) L. Adrienne Cupples, Nancy L. Heard-Costa; (GerMIFS II) Christina Willenborg; (Health 2000) Niina Eklund; (HPFS) Lu Qi; (KORA S3 (GWA)) Claudia Lamina; (NHS) Lu Qi; (NSPHS) Åsa Johansson; (NTRNESDA) Jouke-Jan Hottenga; (PLCO) Sonja I. Berndt; (RSI) Karol Estrada, Carolina Medina-Gomez, Marjolein Peters, Fernando Rivadeneira, André G. Uitterlinden; (RUNMC) Sita H. Vermeulen; (SASBAC) Erik Ingelsson; (SEARCH) Jonathan P. Tyrer; (SHIP) Alexander Teumer; (UKBS-CC) Antony Paul Attwood; (WTCCC-T2D) Andrew P. Morris, Teresa Ferreira, Anubha Mahajan, Reedik Mägi 1817 1818 Page 46 of 68 Table 1 | Loci reaching genome-wide significance (P < 5 x 10-8) for BMI in European sexcombined analysis Alleles SNP Chr:position (bp) Nearest gene *Notable gene(s) Effect/other Effect allele frequency β SE Variance explained N P value Novel loci rs657452 1:49,362,434 AGBL4 - A/G 0.394 0.023 0.003 0.025% 313,651 5.48E-13 rs12286929 11:114,527,614 CADM1 - G/A 0.523 0.022 0.003 0.023% 321,903 1.31E-12 rs7903146 10:114,748,339 TCF7L2 TCF7L2(B) C/T 0.713 0.023 0.003 0.022% 322,130 1.11E-11 rs10132280 14:24,998,019 STXBP6 - C/A 0.682 0.023 0.003 0.023% 321,797 1.14E-11 rs17094222 10:102,385,430 HIF1AN - C/T 0.211 0.025 0.004 0.021% 321,770 5.94E-11 rs7599312 2:213,121,476 ERBB4 ERBB4(D) G/A 0.724 0.022 0.003 0.019% 322,024 1.17E-10 rs2365389 3:61,211,502 FHIT - C/T 0.582 0.020 0.003 0.019% 316,768 1.63E-10 rs2820292 1:200,050,910 NAV1 - C/A 0.555 0.020 0.003 0.019% 321,707 1.83E-10 rs12885454 14:28,806,589 PRKD1 - C/A 0.642 0.021 0.003 0.020% 320,823 1.94E-10 rs16851483 3:142,758,126 RASA2 - T/G 0.066 0.048 0.008 0.029% 233,929 3.55E-10 rs1167827 7:75,001,105 HIP1 HIP1(B); PMS2L3(B,Q); PMS2P5(Q); WBSCR16(Q) G/A 0.553 0.020 0.003 0.020% 306,238 6.33E-10 rs758747 16:3,567,359 NLRC3 - T/C 0.265 0.023 0.004 0.020% 308,688 7.47E-10 rs1928295 9:119,418,304 TLR4 TLR4(B) T/C 0.548 0.019 0.003 0.018% 321,979 7.91E-10 A/G 0.620 0.019 0.003 0.017% 318,385 8.11E-10 rs9925964 16:31,037,396 KAT8 ZNF646(M,Q); VKORC1(Q); ZNF668(Q); STX1B(D); FBXL19(D) rs11126666 2:26,782,315 KCNK3 KCNK3(D) A/G 0.283 0.021 0.003 0.017% 321,979 1.33E-09 rs2650492 16:28,240,912 SBK1 SBK1(D); APOB48R(B) A/G 0.303 0.021 0.004 0.018% 319,464 1.92E-09 rs6804842 3:25,081,441 RARB RARB(B) G/A 0.575 0.019 0.003 0.017% 321,463 2.48E-09 rs4740619 9:15,624,326 C9orf93 C9orf93(C,M) T/C 0.542 0.018 0.003 0.016% 321,887 4.56E-09 rs13191362 6:162,953,340 PARK2 PARK2(B,D) A/G 0.879 0.028 0.005 0.016% 321,902 7.34E-09 rs3736485 15:49,535,902 DMXL2 SCG3(B,D); DMXL2(M) A/G 0.454 0.018 0.003 0.015% 321,398 7.41E-09 G/C 0.153 0.031 0.005 0.024% 233,722 7.76E-09 C/T 0.089 0.031 0.005 0.015% 321,893 8.45E-09 - T/C 0.631 0.018 0.003 0.015% 321,924 1.20E-08 SCARB2 NUP54(M); SCARB2(Q) CYP17A1(B); SFXN2(Q) rs17001654 4:77,348,592 rs11191560 10:104,859,028 NT5C2 rs1528435 2:181,259,207 UBE2E3 rs1000940 17:5,223,976 RABEP1 - G/A 0.320 0.019 0.003 0.016% 321,836 1.28E-08 rs2033529 6:40,456,631 TDRG1 LRFN2(D) G/A 0.293 0.019 0.003 0.015% 321,917 1.39E-08 rs11583200 1:50,332,407 ELAVL4 ELAVL4(B,Q,D) C/T 0.396 0.018 0.003 0.015% 322,095 1.48E-08 rs9400239 6:109,084,356 FOXO3 FOXO3(B); AI057453(unspliced EST)(Q) C/T 0.688 0.019 0.003 0.015% 321,988 1.61E-08 rs10733682 9:128,500,735 LMX1B LMX1B(B) A/G 0.478 0.017 0.003 0.015% 320,727 1.83E-08 rs11688816 2:62,906,552 EHBP1 EHBP1(B) G/A 0.525 0.017 0.003 0.015% 322,051 1.89E-08 rs11057405 12:121,347,850 CLIP1 - G/A 0.901 0.031 0.006 0.017% 314,111 2.02E-08 rs11727676 4:145,878,514 HHIP HHIP(B) T/C 0.910 0.036 0.006 0.021% 296,401 2.55E-08 rs3849570 3:81,874,802 GBE1 GBE1(B,M) A/C 0.359 0.019 0.003 0.016% 284,339 2.60E-08 rs6477694 9:110,972,163 EPB41L4B C9orf4(D) C/T 0.365 0.017 0.003 0.014% 322,048 2.67E-08 rs7899106 10:87,400,884 GRID1 GRID1(B) G/A 0.052 0.040 0.007 0.015% 321,770 2.96E-08 rs2176598 11:43,820,854 HSD17B12 HSD17B12(B,M) T/C 0.251 0.020 0.004 0.015% 316,848 2.97E-08 rs2245368 7:76,446,079 PMS2L11 - C/T 0.180 0.032 0.006 0.030% 205,675 3.19E-08 rs17724992 19:18,315,825 PGPEP1 GDF15(B); PGPEP1(Q) A/G 0.746 0.019 0.004 0.014% 319,588 3.42E-08 rs7243357 18:55,034,299 GRP GRP(B,G) T/G 0.812 0.022 0.004 0.014% 322,107 3.86E-08 rs2033732 8:85,242,264 RALYL RALYL(D) C/T 0.747 0.019 0.004 0.014% 321,406 4.89E-08 Page 47 of 68 Previously identified loci rs1558902 16:52,361,075 FTO FTO(B) A/T 0.415 0.082 0.003 0.325% 320,073 7.51E-153 rs6567160 18:55,980,115 MC4R MC4R(B) C/T 0.236 0.056 0.004 0.111% 321,958 3.93E-53 2:622,348 TMEM18 - G/A 0.828 0.060 0.004 0.103% 318,287 1.11E-50 rs13021737 rs10938397 4:44,877,284 GNPDA2 GABRG1(B) G/A 0.434 0.040 0.003 0.079% 320,955 3.21E-38 rs543874 1:176,156,103 SEC16B - G/A 0.193 0.048 0.004 0.072% 322,008 2.62E-35 rs2207139 6:50,953,449 TFAP2B TFAP2B(B) G/A 0.177 0.045 0.004 0.058% 322,019 4.13E-29 rs11030104 11:27,641,093 BDNF BDNF(B,M) A/G 0.792 0.041 0.004 0.056% 322,103 5.56E-28 rs3101336 1:72,523,773 NEGR1 NEGR1(B,C,D) C/T 0.613 0.033 0.003 0.053% 316,872 2.66E-26 rs7138803 12:48,533,735 BCDIN3D FAIM2(D) A/G 0.384 0.032 0.003 0.047% 322,092 8.15E-24 G/A 0.462 0.031 0.003 0.047% 321,759 8.78E-24 A/C 0.403 0.031 0.003 0.046% 321,930 3.14E-23 rs10182181 2:25,003,800 ADCY3 ADCY3(B,M,Q); POMC(B,G); NCOA1(B) rs3888190 16:28,796,987 ATP2A1 SH2B1(B,M,Q); APOB48R(M,Q); ATXN2L(Q); SBK1(Q,D); SULT1A2(Q); TUFM(Q) rs1516725 3:187,306,698 ETV5 - C/T 0.872 0.045 0.005 0.045% 320,644 1.89E-22 rs12446632 16:19,842,890 GPRC5B GPRC5B(C); IQCK(Q) G/A 0.865 0.040 0.005 0.038% 316,758 1.48E-18 rs2287019 19:50,894,012 QPCTL GIPR(B,M) C/T 0.804 0.036 0.004 0.041% 300,921 4.59E-18 rs16951275 15:65,864,222 MAP2K5 MAP2K5(B,D); LBXCOR1(M) T/C 0.784 0.031 0.004 0.033% 322,098 1.91E-17 rs3817334 11:47,607,569 MTCH2 MTCH2(M,Q); C1QTNF4(Q,I); SPI1(Q); CELF1(D) T/C 0.407 0.026 0.003 0.033% 321,959 5.15E-17 rs2112347 5:75,050,998 POC5 POC5(M); HMGCR(B); COL4A3BP(B) T/G 0.629 0.026 0.003 0.032% 322,019 6.19E-17 rs12566985 1:74,774,781 FPGT-TNNI3K - G/A 0.446 0.024 0.003 0.029% 319,282 3.28E-15 rs3810291 19:52,260,843 ZC3H4 ZC3H4(Q,D) A/G 0.666 0.028 0.004 0.036% 296,261 4.81E-15 rs7141420 14:78,969,207 NRXN3 NRXN3(D) T/C 0.527 0.024 0.003 0.028% 321,970 1.23E-14 rs13078960 3:85,890,280 CADM2 CADM2(D) G/T 0.196 0.030 0.004 0.028% 322,135 1.74E-14 rs10968576 9:28,404,339 LINGO2 LINGO2(D) G/A 0.320 0.025 0.003 0.027% 322,061 6.61E-14 rs17024393 1:109,956,211 GNAT2 AMPD2(D) C/T 0.040 0.066 0.009 0.033% 297,874 7.03E-14 rs12429545 13:53,000,207 OLFM4 OLFM4(B) A/G 0.133 0.033 0.005 0.026% 312,934 1.09E-12 rs13107325 4:103,407,732 SLC39A8 SLC39A8(M,Q) T/C 0.072 0.048 0.007 0.030% 321,461 1.83E-12 rs11165643 1:96,696,685 PTBP2 PTBP2(D) T/C 0.583 0.022 0.003 0.023% 320,730 2.07E-12 rs17405819 8:76,969,139 HNF4G HNF4G(B) T/C 0.700 0.022 0.003 0.021% 322,085 2.07E-11 rs1016287 2:59,159,129 FLJ30838 - T/C 0.287 0.023 0.003 0.021% 321,969 2.25E-11 rs4256980 11:8,630,515 TRIM66 TRIM66(M,D); TUB(B) G/C 0.646 0.021 0.003 0.020% 320,028 2.90E-11 rs12401738 1:78,219,349 FUBP1 USP33(D) A/G 0.352 0.021 0.003 0.020% 322,070 1.15E-10 rs205262 6:34,671,142 C6orf106 SNRPC(Q) G/A 0.273 0.022 0.004 0.019% 315,542 1.75E-10 rs12016871 13:26,915,782 MTIF3 GTF3A(Q) T/C 0.203 0.030 0.005 0.029% 233,803 2.29E-10 rs12940622 17:76,230,166 RPTOR RPTOR(B) G/A 0.575 0.018 0.003 0.016% 322,032 2.49E-09 rs11847697 14:29,584,863 PRKD1 - T/C 0.042 0.049 0.008 0.019% 306,243 3.99E-09 rs2075650 19:50,087,459 TOMM40 TOMM40(B); APOE(B); APOC1(B) A/G 0.848 0.026 0.005 0.017% 308,408 1.25E-08 rs2121279 2:142,759,755 LRP1B - T/C 0.152 0.025 0.004 0.015% 322,065 2.31E-08 rs29941 19:39,001,372 KCTD15 - G/A 0.669 0.018 0.003 0.015% 321,970 2.41E-08 rs1808579 18:19,358,886 C18orf8 NPC1(B,G,M,Q); C18orf8(Q) C/T 0.534 0.017 0.003 0.014% 322,032 4.17E-08 Page 48 of 68 SNP positions are reported according to Build 36 and their alleles are coded based on the positive strand. Effect alleles, allele frequencies, betas (β), standard errors (SE), sample sizes (N), and P values are based on the meta-analysis of GWAS I+II+Metabochip association data from the European All dataset. *Notable genes from biological relevance to obesity (B); GRAIL results (G); BMI-associated variant is in strong LD (r2 ≥ 0.7) with a missense variant in the indicated gene (M); association and eQTL data converge to affect gene expression (Q); DEPICT analyses (D); copy number variation (C). Page 49 of 68 Table 2 | Loci reaching genome-wide significance from analyses other than European sexcombined SNP Chr:position (bp) Nearest gene *Notable gene(s) CALCR CALCR(B); hsamiR-653(Q) Alleles Effect (Effect/ allele Other) frequency β SE N P value Most significant analysis Variance P Value explained (European (European All) All) Novel Loci C/G 0.430 0.029 0.005 161,756 European 2.08E-10 Population Based 5.00E-07 0.018% LOC100287559 BBS4(B,M,Q) T/C 0.671 0.019 0.003 338,384 3.92E-09 All Ancestries 6.83E-08 0.014% PLCD4(B,Q); CYP27A1(B); TTLL4(M,Q); STK36(B,M); ZNF142(M); RQCD1(Q) C/T 0.424 0.024 0.004 152,153 6.78E-09 European Men 4.17E-07 0.012% - C/A 0.413 0.017 0.003 339,166 8.60E-09 All Ancestries 6.55E-08 0.014% RIT2(B,D) G/A 0.391 0.023 0.004 171,837 1.51E-08 1.63E-07 0.013% - C/T 0.599 0.017 0.003 337,300 1.61E-08 All Ancestries 4.16E-07 0.013% G/C 0.229 0.020 0.004 338,177 2.07E-08 All Ancestries 8.99E-08 0.013% T/G 0.403 0.017 0.003 339,152 2.18E-08 All Ancestries 8.65E-08 0.013% 9.67E-08 0.013% rs9641123 7:93,035,668 rs7164727 15:70,881,044 rs492400 2:219,057,996 rs2080454 16:47,620,091 CBLN1 rs7239883 18:38,401,669 LOC284260 rs2836754 21:39,213,610 ETS2 USP37 rs9914578 17:1,951,886 SMG6 SMG6(D); N29617(unsplic ed EST)(Q) rs977747 1:47,457,264 TAL1 - rs9374842 6:120,227,364 European Women LOC285762 - T/C 0.744 0.023 0.004 209,392 European 2.67E-08 Population Based MAPK3(D); KCTD13(D); TAOK2(D); YPEL3(D); DOC2A(D); FAM57B(D) G/A 0.510 0.022 0.004 179,613 European 2.70E-08 Population Based 2.24E-06 0.013% rs4787491 16:29,922,838 INO80E rs1441264 13:78,478,920 MIR548A2 - A/G 0.613 0.017 0.003 326,858 2.96E-08 All Ancestries 6.04E-08 0.015% rs17203016 2:207,963,763 CREB1 CREB1(B); KLF7(B) G/A 0.195 0.021 0.004 333,383 3.41E-08 All Ancestries 8.15E-08 0.014% rs16907751 8:81,538,012 ZBTB10 - C/T 0.913 0.047 0.009 145,842 3.89E-08 European Men 1.26E-07 0.019% rs13201877 6:137,717,234 IFNGR1 OLIG3(G) G/A 0.140 0.024 0.004 339,026 4.29E-08 All Ancestries 2.35E-07 0.013% 1.42E-07 0.015% rs9540493 13:65,103,705 MIR548X2 rs1460676 2:164,275,935 FIGN rs6465468 7:95,007,450 ASB4 A/G 0.452 0.021 0.004 206,327 European 4.97E-08 Population Based - C/T 0.179 0.021 0.004 339,157 4.98E-08 All Ancestries 8.98E-07 0.011% ASB4(B) T/G 0.306 0.025 0.005 166,136 4.98E-08 European Women 2.32E-06 0.012% European Women PCDH9(D) Previously identified loci rs6091540 20:50,521,269 ZFP64 - C/T 0.721 0.030 0.004 171,875 2.15E-11 8.02E-08 0.014% rs7715256 5:153,518,086 GALNT10 - G/T 0.422 0.017 0.003 339,153 8.85E-09 All Ancestries 1.70E-07 0.013% rs2176040 2:226,801,046 LOC646736 IRS1(B,Q) A/G 0.365 0.024 0.004 152,818 9.99E-09 European Men 6.06E-06 0.009% SNP positions are reported according to Build 36 and their alleles are coded based on the positive strand. Effect alleles, allele frequencies, betas (β), standard errors (SE), sample sizes (N), and P values are based on the meta-analysis of GWAS I+II+Metabochip association data from the European All dataset. *Notable genes from biological relevance to obesity (B); GRAIL results (G); BMI-associated variant is in strong LD (r2 ≥ 0.7) with a missense variant in the indicated gene (M); association and eQTL data converge to affect gene expression (Q); DEPICT analyses (D); copy number variation (C). Page 50 of 68 Table 3 | Secondary signals reaching genome-wide significance by joint conditional association analysis Alleles Effect/Other Estimated effect allele frequency FLJ30838 T/C 0.294 0.023 0.003 0.021% 356,608 2.62E-11 Lead FLJ30838 T/G 0.457 0.021 0.004 0.021% 248,759 2.73E-08 Second signal 16:3567359 NLRC3 T/C 0.241 0.022 0.004 0.018% 316,090 2.00E-09 Lead 16:3955730 ADCY9 T/C 0.620 0.024 0.004 0.027% 222,610 2.17E-09 Second signal rs12446632 16:19842890 GPRC5B G/A 0.860 0.036 0.005 0.031% 341,146 1.06E-14 Lead rs11074446 16:20162624 GP2 T/C 0.867 0.029 0.005 0.019% 338,145 1.71E-10 Second signal rs6567160 18:55980115 MC4R C/T 0.233 0.048 0.004 0.084% 360,901 3.52E-38 Lead rs17066842 18:56191604 MC4R G/A 0.960 0.051 0.008 0.020% 312,679 6.99E-10 Second signal rs9944545 18:56109224 MC4R T/C 0.296 0.020 0.004 0.017% 349,842 1.01E-08 Second signal rs11030104 11:27641093 BDNF A/G 0.791 0.051 0.004 0.087% 354,703 1.26E-34 Lead rs10835210 11:27652486 BDNF C/A 0.570 0.020 0.004 0.020% 334,430 1.25E-08 Second signal Chr:position (bp) Nearest gene rs1016287 2:59159129 rs4671328 2:58788786 rs758747 rs879620 SNP Estimated β Estimated SE Estimated variance explained Estimated N Estimated P value Lead SNP or second signal? SNP positions are reported according to Build 36 and their alleles are coded based on the positive strand. Effect allele frequencies, betas (β), standard errors (SE), variances explained, sample sizes (N), and P values from GCTA. Page 51 of 68 1819 METHODS 1820 Main Analyses 1821 Study design 1822 We conducted a two-stage meta-analysis to identify BMI-associated loci in 1823 European adults (Extended Data Fig. 1, Extended Data Table 1). In stage 1 1824 we performed meta-analysis of 80 GWAS (N=234,069); and stage 2 incorporated 1825 data from 34 additional studies (N=88,137) genotyped using the Metabochip25 1826 (Supplementary Tables 1-3). Secondary meta-analyses were also conducted 1827 for: 1) all ancestries, 2) European men, 3) European women, and 4) European 1828 population-based studies. The total number of subjects and SNPs included in 1829 each stage for all analyses is shown in Extended Data Table 1. 1830 1831 Phenotype 1832 BMI, measured or self-reported weight in kg / height in meters squared 1833 (Supplementary Tables 1 & 3) was adjusted for age, age2, and any necessary 1834 study-specific covariates (e.g., genotype-derived principal components) in a 1835 linear regression model. The resulting residuals were transformed to approximate 1836 normality using inverse normal scores. For studies with no known related 1837 individuals, residuals were calculated separately by sex and case/control status. 1838 For family-based studies, residuals were calculated with men and women 1839 together, adding sex as an additional covariate in the linear regression model. 1840 Relatedness was accounted for in a study-specific manner (Supplementary 1841 Table 2). Page 52 of 68 1842 1843 Sample QC, imputation, and association 1844 Following study-specific quality control (QC) measures (Supplementary Table 1845 2), all contributing GWAS common SNPs were imputed using the HapMap Phase 1846 II 1847 CEU+YRI+CHB+JPT HapMap Release 22 for the African American and Hispanic 1848 GWAS. Directly genotyped (GWAS and Metabochip) and imputed variants 1849 (GWAS only) were then tested for association with the inverse normally 1850 transformed BMI residuals using linear regression assuming an additive genetic 1851 model. QC following study level analyses was conducted following procedures 1852 outlined elsewhere67. CEU reference panel for European-descent studies54, and 1853 1854 Meta-analysis 1855 Fixed effects meta-analyses were conducted using the inverse variance- 1856 weighted method implemented in METAL52. Study-specific GWAS results as well 1857 as GWAS meta-analysis results were corrected for genomic control (GC) using 1858 all SNPs68. Study-specific Metabochip results as well as Metabochip meta- 1859 analysis results were GC-corrected using 4,425 SNPs included on the 1860 Metabochip for replication of associations with QT-interval, a phenotype not 1861 correlated with BMI, after pruning of SNPs within 500 kb of an anthropometry 1862 replication SNP. The final meta-analysis combined the GC-corrected GWAS and 1863 Metabochip meta-analysis results. 1864 Page 53 of 68 1865 Identification of novel loci 1866 We used a distance criterion of ±500 kb surrounding each GWS peak (P < 5×10-8) 1867 to define independent loci and to place our results in the context of previous 1868 studies, including our previous GIANT meta-analyses. Of several locus models 1869 tested, this definition most closely reflected the loci defined by approximate 1870 conditional analysis using GCTA. (Tables 1 & 2, respectively). Current index 1871 SNPs falling within 500 kb of a SNP previously associated with BMI, weight, 1872 extreme obesity, or body fat percentage11,20,22,24,26 were considered previously 1873 identified. 1874 1875 Characterization of BMI associated SNP effects 1876 Heterogeneity among studies 1877 To investigate potential sources of heterogeneity between groups we compared 1878 the effect estimates of our 97 GWS SNPs for men vs. women of European 1879 ancestry and Europeans vs. non-Europeans. To address the effects of studies 1880 ascertained on a specific disease or phenotype on our results we also compare 1881 effect estimates of European ancestry studies of population-based studies with 1882 the following European-descent subsets of studies: 1883 studies (i.e., those ascertained on a specific disease or phenotype); 2) T2D 1884 cases; 3) T2D controls; 4) combined T2D cases and controls; 5) CAD cases; 6) 1885 CAD controls; and 7) combined CAD cases and controls (Supplementary 1886 Tables 10 & 11). We also tested for heterogeneity of effect estimates between 1887 our European sex-combined meta-analysis and results from recent GWAS meta- Page 54 of 68 1) non-population-based 1888 analyses for BMI in individuals of African or East Asian ancestry11,69 1889 (Supplementary Table 9). Heterogeneity was assessed as described in Randall 1890 et al.15. A Bonferroni-corrected P value < 5×10-4 (corrected for 97 tests) was used 1891 to assess significance. 1892 1893 Fine-mapping 1894 We compared the meta-analysis results and credible sets of SNPs likely to 1895 contain the causal variant, based on the method of Maller et al. 27, across the 1896 European-only, non-European, and all ancestries sex-combined meta-analyses. 1897 For each index SNP falling within a Metabochip fine-mapping region (27 for BMI), 1898 all SNPs available within 500 kb on either side of the index SNP were selected. 1899 Effect size estimates and standard errors for each SNP were converted to 1900 approximate Bayes’ Factors (ABFs) according to the method described by 1901 Wakefield28. All ABFs were then summed across the 1 MB region and the 1902 proportion of the posterior odds of being the causal variant was calculated for 1903 each variant (ABF for SNPi/Sum of ABFs for the region). The set of SNPs that 1904 accounts for 99% of posterior odds of association in the region denotes the set 1905 most likely to contain the causal variant for that association region 1906 (Supplementary Table 12). 1907 1908 Cumulative effects, risk prediction, and variance explained 1909 We assessed the cumulative effects of the 97 GWS loci on mean BMI and on 1910 their ability to predict obesity (BMI ≥30 kg/m2) using the c statistic from logistic Page 55 of 68 1911 regression models in the Health and Retirement Study30, a longitudinal study of 1912 26,000 European Americans 50 years or older. The variance explained by each 1913 SNP was calculated using the effect allele frequency (f) and beta (β) from the 1914 meta-analyses using the formula VarExp = β2(1-f)2f. 1915 1916 For polygene analyses, the approximate conditional analysis from GCTA32,33, 1917 was used to select SNPs using a range of P value thresholds (i.e., 5×10-8, 5×10-7, 1918 …, 5×10-3) based on summary data from the European sex-combined meta- 1919 analysis excluding TwinGene and QIMR studies. We performed a within-family 1920 prediction analysis using full-sib pairs selected from independent families (1,622 1921 pairs from the QIMR cohort and 2,758 pairs from the TwinGene cohort) and then 1922 SNPs at each threshold were used to calculate the percent of phenotypic 1923 variance explained and predict risk (Extended Data Figs. 2 & 3). We then 1924 confirmed the results from population-based prediction and estimation analyses 1925 in independent sample of unrelated individuals from the TwinGene (N = 5,668) 1926 and QIMR (N = 3,953) studies ((Extended Data Fig. 3, Fig. 1C). The SNP- 1927 derived predictor was calculated using the profile scoring approach implemented 1928 in PLINK and estimation analyses were performed using the all-SNP estimation 1929 approach implemented in GCTA. 1930 1931 Enrichment analysis of Metabochip SNPs selected for replication 1932 The 5,055 SNPs that were included for BMI replication on the Metabochip 1933 included 1,909 independent SNPs (r2 < 0.1 and >500 kb apart), of which 1,458 Page 56 of 68 1934 displayed directionally consistent effect estimates with those reported in 1935 Speliotes et al.24. To estimate the number of Metabochip SNPs truly associated 1936 with BMI, we counted the number of SNPs with directional consistency (DC) 1937 between Speliotes et al.24 and a meta-analysis of non-overlapping samples for 1938 these 1,909 SNPs. We then calculated DC in the presence of a mixture of 1939 associated and non-associated SNPs assuming P(DC | associated) = 1 and 1940 P(DC | not associated) = 0.5. In this formulation, DC = R/2 + S, meaning that S = 1941 2DC – T, where T equals the total number of SNPs, R equals the number of 1942 SNPs not associated with BMI, and S equals the number of SNPs associated 1943 with BMI. With DC = 1,458 and T = 1,909, we estimate S to be 2DC – T = 2 × 1944 1,458 – 1,909 = 1,007. 1945 1946 Joint and conditional multiple SNP association analysis 1947 In order to identify additional signals in regions of association, we used GCTA 32, 1948 an approach that uses meta-analysis summary statistics and an LD matrix 1949 derived from a reference sample, to perform approximate joint and conditional 1950 SNP association analysis. We used 6,654 unrelated individuals of European 1951 ancestry from the ARIC cohort as the reference sample to approximate 1952 conditional P values. 1953 Page 57 of 68 1954 Identifying potential biological pathways, genes, tissues, and functional 1955 variants at associated loci 1956 Manual gene annotation and biological description 1957 All genes within 500 kb of an index SNP were annotated for molecular function, 1958 cellular function, and for evidence of association with BMI-related traits in human 1959 or animal model experiments (Supplementary Table 22). We used several 1960 avenues 1961 (http://csg.sph.umich.edu/boehnke/spotter/), 1962 (http://www.ncbi.nlm.nih.gov/pubmed/), 1963 UNIPROT (http://www.uniprot.org/). When no genes mapped to this interval the 1964 nearest gene on each side of the index SNP was annotated. In examining 1965 possible functions of genes in the region, we excluded any references to GWAS 1966 or other genetic association studies. We analyzed 405 genes in the 97 GWS loci 1967 and manually curated them into 25 biological categories containing more than 1968 three genes. for annotation, OMIM including Spotter PubMed (http:/www.omim.org), and 1969 1970 Functional variants 1971 All variants within 500 kb (HapMap release 22/1000 Genomes CEU) and in LD 1972 (r2 > 0.7) with an index SNP were annotated for functional effects based on 1973 RefSeq transcripts using Annovar70 (http://www.openbioinformatics.org/annovar/). 1974 PhastCon, Grantham, GERP, and PolyPhen71 predictions were accessed via the 1975 Exome Variant Server72 (http://evs.gs.washington.edu/EVS), and from SIFT73 1976 (http://sift.jcvi.org/) (Extended Data Table 3). Page 58 of 68 1977 1978 CNVs correlated with BMI index SNPs 1979 To study common copy number variations (CNVs), we used a list of CNV-tagging 1980 SNPs in high LD (r2 > 0.8) with deletions in European populations from Phase 1 1981 release of the 1000 Genomes55. 1982 1983 eQTLs 1984 We examined the cis associations between the 97 GWS SNPs and expression of 1985 nearby genes in whole blood, lymphocytes, skin, liver, omental fat, subcutaneous 1986 fat, and brain tissue56-63 (Supplementary Table 23). Conditional analyses were 1987 performed by including both the BMI-associated SNP and the most significant 1988 cis-associated SNP for the given transcript. Conditional analyses were conducted 1989 for all datasets, except the brain tissue dataset due to limited power. To minimize 1990 the potential for false-positives, only cis associations below a study-specific FDR 1991 of 5% (or 1% for some datasets), in LD with the peak SNP (r2 > 0.7) for the 1992 transcript, and with conditional P value >0.05 for the peak SNP, are reported 1993 (Extended Data Table 2). 1994 1995 Pathway analyses 1996 MAGENTA 1997 We used the MAGENTA method (Meta-Analysis Gene-set Enrichment of variaNT 1998 Associations) to test predefined gene sets for enrichment at BMI-associated 1999 loci36. We used the GWAS+Metabochip data as input and applied default settings. Page 59 of 68 2000 2001 GRAIL 2002 We used GRAIL, Gene Relationships Across Implicated Loci42, to identify genes 2003 near BMI-associated loci having similarities in the published scientific text using 2004 PubMed abstracts as of December 2006. The BMI loci were queried against 2005 HapMap release 22 for the European panel, and we controlled for gene size. 2006 2007 Data-driven Expression-Prioritized Integration for Complex Traits (DEPICT) 2008 We used DEPICT, Data-driven Expression-Prioritized Integration for Complex Traits, to 2009 identify the most likely causal gene at a given associated locus, reconstituted gene sets 2010 enriched for BMI associations, and tissues and cell types in which genes from 2011 associated loci are highly expressed (T. H. Pers et al., in preparation). To accomplish 2012 this, the method relies on publicly available gene sets (including molecular pathways) 2013 and uses gene expression data from 77,840 gene expression arrays to predict which 2014 other genes are likely to be part of these gene sets, thus combining known annotations 2015 with predicted annotations. For details and negative control analyses please refer to 2016 Supplementary Online Materials (T.H. Pers et al., in preparation). 2017 2018 We first clumped the European-only GWAS-based meta-analysis summary statistics 2019 using 500 kb flanking regions, LD r2 > 0.1 and excluded SNPs with P ≥ 5×10-4; which 2020 resulted in a list of 590 independent SNPs. HapMap Project Phase II CEU genotype 2021 data54 was used to compute LD and genomic coordinates were defined by genome build 2022 GRCh37. Because the GWAS meta-analysis was based on both GWAS and Metabochip 2023 studies, there were discrepancies in the index SNPs that are referenced in Table 1 of 2024 the paper and the ones used in DEPICT, which was run on the GWAS data only. Page 60 of 68 2025 Therefore we forced in GWS index SNPs from the GWAS plus Metabochip GWA meta- 2026 analysis into the DEPICT GWAS-only based analysis. This enabled a more 2027 straightforward comparison of genes in DEPICT loci and genes in GWS loci highlighted 2028 by manual lookups, and did not lead to any significant bias towards SNPs on the 2029 Metabochip (data not shown). We forced in 62 of the GWS loci in Table 1, so all of 97 2030 SNPs were among the 590 SNPs. The 590 SNPs were further merged into 511 non- 2031 overlapping regions (FDR < 0.05) used in DEPICT analysis. For additional information 2032 on the analysis please refer to Supplementary Online Materials. 2033 2034 Cross-trait analyses 2035 Cross-trait lookups 2036 To carefully explore 2037 cardiometabolic traits and diseases, association results for the 97 BMI index 2038 SNPs were requested from 13 GWAS meta-analysis consortia: 2039 (T2D)74, CARDIoGRAM-C4D (CAD)75, ICBP (systolic and diastolic blood 2040 pressure (SBP, DBP))76, GIANT (waist-to-hip ratio, hip circumference, and waist 2041 circumference, each unadjusted and adjusted for BMI) (A.R.W. et al. and D. 2042 Shungin et al., submitted), GLGC (high density lipoprotein cholesterol (HDL), low 2043 density lipoprotein cholesterol (LDL), triglycerides (TG), and total cholesterol 2044 (TC)) [manuscript in preparation], MAGIC (fasting glucose, fasting insulin, fasting 2045 insulin adjusted for BMI, and two-hour glucose)40,77,78, ADIPOGen (BMI-adjusted 2046 adiponectin)64, CKDgen (urine albumin-to-creatinine ratio (UACR), estimated 2047 glomerular filtration rate (eGFR), and overall CKD)79,80, ReproGen (age at the relationship between BMI and an array of Page 61 of 68 DIAGRAM 2048 menarche, age at menopause)81,82, GENIE (diabetic nephropathy)83,84. Proxies 2049 (r2 > 0.8 in CEU) were used when an index SNP was unavailable. 2050 2051 Enrichment of Concordant Effects 2052 We compared the effects for the 97 BMI index SNP across these related traits 2053 using a one-sided binomial test of the number of concordant effects versus a null 2054 expectation of P = 0.5. Concordant and nominally significant (P < 0.05) SNP 2055 effects were similarly tested using a one-sided binomial test with a null 2056 expectation of P = 0.05. We evaluated significance in either test with a 2057 Bonferroni-corrected threshold of P = 0.002 (0.05/23 traits tested). 2058 2059 Joint effects of cross-trait associations 2060 In order to determine the joint effect of all 97 BMI loci on other cardiometabolic 2061 phenotypes, we employed the meta-regression technique from Dastani et al.64 to 2062 correlate the effect estimates of the BMI increasing alleles with effect estimates 2063 from meta-analyses for each of the metabolic traits from other consortia 2064 (DIAGRAM, MAGIC, ICBP, GLGC, ADIPOgen, ReproGEN and CARDIoGRAM). 2065 2066 Cross-Traits Heatmap 2067 To explore observed concordance in effects of BMI loci on other cardiometabolic 2068 and anthropometric traits, we converted the effect estimates and standard errors 2069 (or P values) from meta-analysis to Z-scores oriented with respect to the BMI- 2070 increasing allele, for each of the 97 BMI index SNPs in the twenty-three traits. Page 62 of 68 2071 We then classified each Z-score as follows to generate a vector of the Z-score of 2072 each trait at each locus: 2073 2074 0 (not significant) if -2 <= Z <= 2 2075 1 (significant positive) if Z > 2 2076 -1 (significant negative) if Z < -2, 2077 2078 Fig. 2 displays these locus-trait relationships in a heat map using Euclidean 2079 distance and complete linkage clustering to order both loci and traits. 2080 2081 Cross-Traits Bubble Plot (Extended Data Fig. 5) 2082 We also represent the genetic overlap between other cardiometabolic traits and 2083 BMI susceptibility loci with a bubble plot in which the size of each bubble is 2084 proportional to the fraction of BMI-associated loci for which there was a 2085 significant association (P < 5×10-4). Each pair of bubbles is connected by a line 2086 proportional to the number significant BMI-increasing loci overlapping between 2087 the traits. 2088 2089 NHGRI GWAS Catalog Lookups 2090 We extracted previously reported GWAS association within 500 kb of and r2 > 0.7 2091 with any BMI-index SNP from the National Human Genome Research Institute 2092 (NHGRI) GWAS Catalog65 (www.genome.gov/gwastudies; Supplementary 2093 Tables 17A & B). For studies reporting greater than 30 significant hits, additional Page 63 of 68 2094 SNP-trait associations were pulled from the literature and compared to BMI index 2095 SNPs the same as with other GWAS Catalog studies. 2096 2097 Regulatory variation 2098 ENCODE/Roadmap 2099 To identify global enrichment of datasets at the BMI-associated loci we 2100 performed permutation-based tests in a subset of 41 open chromatin (DNase- 2101 seq), histone modification (H3K27ac, H3K4me1, H3K4me3, H3K9ac), and 2102 transcription factor binding datasets from the ENCODE Consortium66, Roadmap 2103 Epigenomics Project35, and when available the ENCODE Integrative Analysis85,86 2104 (Supplementary Table 19A, B, & C). We processed Roadmap Epigenomics 2105 sequencing data with multiple biological replicates using MACS2 87 and then 2106 applied same Irreproducible Discovery Rate pipeline used in the ENCODE 2107 Integrative Analysis85,86. Roadmap Epigenomics data with only a single replicate 2108 were analyzed using MACS2 alone. We examined variants in LD with 97 BMI 2109 index SNPs based on r2 > 0.7 from the 1000 Genomes Phase 1 version 2 EUR 2110 samples88. We matched the index SNP at each locus with 500 variants having no 2111 evidence of association (P > 0.5, ~1.2 million total variants) with a similar 2112 distance to the nearest gene (± 11,655 bp), number of variants in LD (± 2113 8 variants), and minor allele frequency. Using these pools, we created 10,000 2114 sets of control variants for each of the 97 loci and identified variants in LD (r2 > 2115 0.7) and within 1 Mb. For each SNP set, we calculated the number of loci with at 2116 least one variant located in a regulatory region under the assumption that one Page 64 of 68 2117 regulatory variant is responsible for each association signal. We estimated the P 2118 value assuming a sum of binomial distributions to represent the number of index 2119 SNPs (or their LD proxies; r2 > 0.7) that overlap a regulatory dataset compared to 2120 the expectation observed in the 500 matched control sets. Datasets were 2121 considered significantly enriched if the P value was below a Bonferroni-corrected 2122 threshold of 1.2×10-3, adjusting for 41 tests. 2123 Page 65 of 68 2124 Figure Legends 2125 Fig. 1. Genetic characterization of BMI-associated variants. A. Plot of the cumulative 2126 phenotypic variance explained by each locus ordered by decreasing effect size. B. The 2127 relationship between effect size and allele frequency. Previously identified loci are blue 2128 circles and novel loci are red triangles. C. The estimated variance in BMI explained by 2129 SNPs selected at a range of P values using unrelated individuals from the QIMR (N = 2130 3,924; purple) and TwinGene (N = 5,668; gold) studies, their weighted average (cyan), 2131 and the variance explained inferred from within-family prediction (red; Extended Data 2132 Fig. 2). In orange is the estimated variance explained by all HapMap Phase III SNPs in 2133 16,275 unrelated individuals from the QIMR, TwinGene, and ARIC studies. D. QQ plot of 2134 meta-analysis P values for all 1,909 BMI-replication SNPs (blue) and after removing 2135 SNPs near the 97 associated loci (green). E. Histogram of cumulative effect of BMI risk 2136 alleles. Mean BMI for each bin is shown by the black dots (with standard deviation) and 2137 corresponds to the right hand y-axis. F. Regional association plot of the region upstream 2138 of and including MC4R89. SNP associations from the European sex-combined meta- 2139 analysis are plotted with joint conditional P values indicated for each of the three GWS 2140 signals. SNPs are shaded and shaped based on the index SNP with which they are in 2141 strongest LD (rs6567160 in blue, rs994545 in yellow, and rs17066842 in green). 2142 Page 66 of 68 2143 Fig 2. Effects of BMI-associated loci on related metabolic traits. Unsupervised 2144 hierarchical clustering of the 97 BMI-associated loci (y-axis) on 23 related metabolic 2145 traits (x-axis). The top row shows the a priori expected relationship with BMI (green is 2146 concordant effect direction, purple is opposite). Loci with statistically significant 2147 concordant direction of effect are highlighted in green, and significant but opposing 2148 effects are in purple. Grey indicates a non-significant relationship and those with no 2149 information are in white. The key in the upper left corner also shows the count of gene- 2150 phenotype pairs in each category (cyan bars). 2151 Page 67 of 68 2152 Fig 3. Tissues and reconstituted gene sets significantly enriched for genes within 2153 BMI-associated loci. A. Genes within BMI-associated loci (P < 5×10-4) are enriched for 2154 expression in the central nervous system and brain tissues as predicted by DEPICT. 2155 Tissues are sorted by physiological system and significantly enriched tissues are in 2156 black. B. The gene sets most significantly enriched for BMI-associated loci as identified 2157 by DEPICT (P < 10-6, FDR < 4×10-4). Nodes represent reconstituted gene sets and are 2158 color-coded by P value. Edge thickness between nodes is proportional to degree of gene 2159 overlap as measured by the Jaccard index. Nodes with gene overlap greater than 25% 2160 were collapsed into a single “meta-node” (denoted with a blue border). C. The nodes 2161 contained within the most enriched gene set meta-node, “Clathrin-Coated Vesicle,” 2162 which shares genes with other gene sets relevant to glutamate signaling and synapse 2163 biology. D. The “Generation of a Signal Involved in Cell-Cell Signaling” meta-node 2164 represents several overlapping gene sets relevant to obesity and energy metabolism 2165 (gene sets with P < 4×10-3, FDR < 0.05 shown). For the complete list of enriched gene 2166 sets refer to Supplementary Table 21A. Page 68 of 68