Information gap - EAL Nexus
advertisement

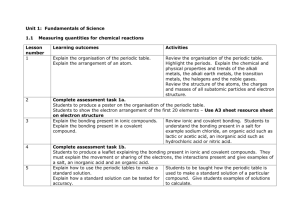
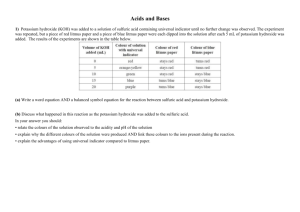
This project and its actions were made possible due to co-financing by the European Fund for the Integration of Third-Country Nationals Nexus resource Reactants and products Information gap Subject: Science Age groups: 8-11, 12-14 Topic: Chemical reactions Licence information | This resource is free to use for educational purposes. ©British Council 2014 Source | This resource was originally developed by Alison Fisher and has been adapted by EAL Nexus. Reactants and Products. Work on your own. Circle the product and answer this question: How do you know a chemical reaction has taken place? cake sugar butter flour I know a chemical reaction has taken place because………….……………….. ………………………………………………………………………………………………………. Underline the products in the sentences below. Remember that a product is what you have made after the experiment has finished. 1. Haseeb took methane and burnt it in oxygen. This produced carbon dioxide and water. 2. Iron sulphate is formed when iron is added to sulphuric acid. Hydrogen gas is also produced. 3. Pupils found that sodium chloride and water were formed when sodium hydroxide was added to hydrochloric acid. 4. If you add nitric acid to sodium carbonate they will react together and produce sodium nitrate, carbon dioxide and water. Now compare your answers with your partner. He or she should have underlined the reactants. ©British Council 2014 Now work together with your partner to write the sentences as word equations. Example: sodium hydroxide + hydrochloric acid sodium chloride + water 1. + + 2. + + 3. + + + 4. + Licence information | This resource is free to use for educational purposes. ©British Council 2014 Source | This resource was originally developed by Alison Fisher and has been adapted by EAL Nexus. + Reactants and Products. Work on your own. Circle the reactants and answer this question: How do you know a chemical reaction has taken place? cake sugar butter flour I know a chemical reaction has taken place because………….……………….. ………………………………………………………………………………………………………. Underline the reactants in the sentences below. Remember that a reactant is what you use at the beginning of an experiment. 1. Haseeb took methane and burnt it in oxygen. This produced carbon dioxide and water. 2. Iron sulphate is formed when iron is added to sulphuric acid. Hydrogen gas is also produced. 3. Pupils found that sodium chloride and water were formed when sodium hydroxide was added to hydrochloric acid. 4. If you add nitric acid to sodium carbonate they will react together and produce sodium nitrate, carbon dioxide and water. Now compare your answers with your partner. He or she should have underlined the products. Licence information | This resource is free to use for educational purposes. ©British Council 2014 Source | This resource was originally developed by Alison Fisher and has been adapted by EAL Nexus. Now work together with your partner to write the sentences as word equations. Example: sodium hydroxide + hydrochloric acid sodium chloride + water 1. + + 2. + + 3. + + + 4. + Licence information | This resource is free to use for educational purposes. ©British Council 2014 Source | This resource was originally developed by Alison Fisher and has been adapted by EAL Nexus. +