Solutions Prelab
advertisement

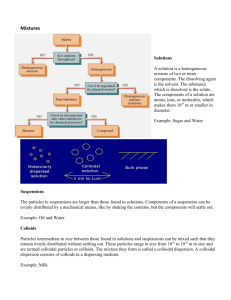
Name: ________________________ Solutions Pre Lab True Solution, Suspension and Colloidal Solution- Based on distinct properties, solutions can be classified into True Solution, Suspension and Colloid. This classification is necessary to understand concepts of colloidal solutions and distinguish it from rest of the types. Types of Solutions True Solution In a solution you have a solute (sugar, salt, etc.) that dissolves into a solvent (water, alcohol). Particle sizes distinguish one homogeneous mixture from another. Solutions are mixtures with particle sizes at the molecule or ion level. The particles have dimensions between 0.1 to 2 nanometers. Typically solutions are transparent. Light can usually pass through the solution. If the solute is able to absorb visible light then the solution will have a color. A blue liquid transmits blue light and absorbs the other colors of the spectrum. A mixture of water H2O and ethanol CH3CH2OH is homogeneous. The particles are individual molecules of H2O and CH3CH2OH. The two molecules are spread uniformly throughout the solution. A mixture of water and sodium chloride is homogeneous by chemistry standards. The particles in the mixture are molecules of H2O and hydrated sodium cations, Na+, and chloride anions, Cl1-. Solutions are transparent. You can see through them. The mixture remains stable and does not separate after standing for any period of time. The particles are so small they cannot be separated by normal filtration. A solution may have a "color" but it will still be transparent. Colloids Colloids are mixtures with particle sizes that consist of clumps of molecules. The particles have dimensions between 2 to 1000 nanometers. The colloid looks homogeneous to the naked eye. Fog and milk are examples of colloids. Colloids frequently appear "murky" or "opaque". The particles are large enough to scatter light (Tyndall Effect) from a lazer. You have experience with the way fog interacts with the light from car headlights. Colloids generally do not separate on standing. They are not separated by filtration. Suspensions Suspensions are homogeneous mixtures with particles that have diameters greater than 1000 nm, 0.000001 meter. The size of the particles is great enough so they are visible to the naked eye. Blood and aerosol sprays are examples of suspensions. Suspensions are "murky" or "opaque". They do not transmit light. Suspensions separate on standing. The mixture of particles can be separated by filtration. Property True Solution Colloidal Solutions Suspension Size of the particles < 1nm 1– 1000nm >1000nm Nature Homogeneous Heterogeneous Heterogeneous Filterability(Diffusion through parchment paper) Particles of true Solution diffuse Colloidal particles pass through rapidly through filter paper as filter paper but not through well as parchment paper. parchment paper. Suspension particles do not pass through filter paper and parchment paper. Colloidal particles are not seen to Suspension particles are big naked eye but can be studied enough to be seen by naked through ultra microscope. eye. Visibility Particles of True Solution are not visible to naked eye. Tyndall effect True Solution does not show Tyndall effect. Colloids shows Tyndall effect. Suspension may or may not show Tyndall effect. Appearance Transparent Translucent Opaque Pre-Lab Questions: 1. What is a true solution? What size characteristics does it have? 2. What are some of the characteristics that a true solution have? 3. Describe the relationship between solutes and solvents. Give an example of each. 4. What is the Tyndall effect, and how can you use that information to determine what type of solution (true, colloid, suspension) you have? 5. How can you use filters to determine what type of solution you have? 6. What are some examples of suspension? 7. How could you use a laser pointer to figure out what type of solution you have? 8. Since solution type is based on the particle size, why can’t we just measure the particles size to determine what type of solution it is? 9. How is a mixture different from a pure substance?