DSR S1W16 Day 2
advertisement

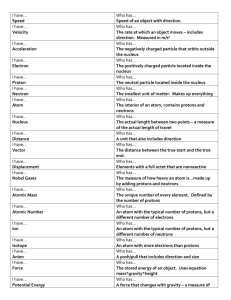
DSR S1W16 Day 2 Name: ______________________________________________ 1. The joining of two or more atoms is called a. chemical holding. b. chemical bonding. c. atom bonding. d. chemical interaction. 2. Potassium has a mass number of 39 and 19 electrons therefore a. a potassium atom has 20 neutrons and 19 protons. b. a potassium atom has 35 neutrons and 17 protons. c. a potassium atom has 19 neutrons and 20 protons. d. Not enough information given. 3. Negatively charged particle found outside the nucleus a. proton b. neutron c. electron d. nucleus 4. Except for Hydrogen and Helium, the outermost energy level of an atom is full when a. it contains 6 electrons. b. it contains 8 electrons. c. it contains 2 electrons. d. it can never be full. 5. Use the diagram below to answer the following questions. e. Number of protons f. Number of neutrons g. Number of electrons h. What is the atomic number for this atom? i. What is the charge of this atom?