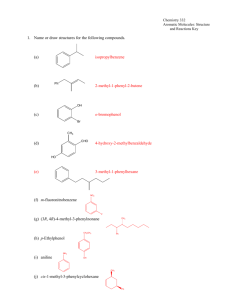
Aromatic Nucleophilic Substitution
advertisement

CHEM 73/8311 CS9 Aromatic electrophilic Substitution.docx 2012March21 1 Aromatic Electrophilc Substitution I. arenium ion mechanism - AE + DE X X E+ X X E E E E + X+ A. first step is usually rate determining B. evidence 1. usually no primary deuterium isotope effect (X = H) 2. arenium ions observed by NMR in superacid solutions 3. mechanism is consistent with distribution of products II. -complex A. probably forms prior to arenium ion B. may be rate determining step certain cases III. electron-transfer mechanism X X E+ E X E + others resonance forms X + others resonance forms oxidation of anisole and other electron rich aromatic in presence of NO 2 gave same product distribution as direct nitration (common intermediate) IV. DE + AE (SE1) E G+ V. single directing and activating groups G CHEM 73/8311 CS9 Aromatic electrophilic Substitution.docx 2012March21 2 A. rationalized using arenium ion B. electron donating groups accelerate versus H and direct toward ortho and para positions H H E G G H E E G G C. electron withdrawing groups slow down versus H and direct toward meta D. Cl, Br, and I deactivate but are ortho and para directors E. ipso attack - five possible fates: E E H E H Nu migration, ipso group loss, electrophile X X X electrophile migration, ipso group -E+ -X+ X loss, nucleophile addition E H X E 1. known groups displaced after ipso attack: stable alkyl cations, electro positive groups (SiR3 and SnR3) 2. silicon and tin are good ipso directors because of stabilization of -carbocation (hyperconjugation) SiR3 X- H VI. Multiple groups A. strongest activating group controls H NO2 NO2 Cl Cl NO2 position of substitution Cl Cl Cl2 B. position between meta goups Cl Cl NO2 NO2 NO2 H Cl disfavored Cl Cl Cl Cl CHEM 73/8311 CS9 Aromatic electrophilic Substitution.docx 2012March21 3 C. ortho effect: meta and ortho-para directors meta to each other - substitution occurs ortho to meta director. Initial attack is ipso which does not place charge at NO 2 carbon VII. structure/reactivity A. Partial rate factors – rate relative to single benzene position 1. f o k obs k benzene fraction (ortho ) 6(benzene ) 2(ortho ) 2. for example: if nitration of trichloromethylbenzene were 15 times slower than benzene and 29% of product is para then f p 1 6 0.29 0.116 15 1 B. reactive electrophiles have early transition state, rates and product distribution are less sensitive to substitutents: Partial rates factors for toluene fm fp alkylation (RBr, GaBr3) -2.4 1.4 5.0 bromination (Br2, AcOH) -13.1 5.5 2420 C. early versus late transition state E H E H E E late transition state H E H E early transition state CHEM 73/8311 CS9 Aromatic electrophilic Substitution.docx 2012March21 1. late transition state structure is like intermediate: using perturbation theory o,p substituents overlap with MO at positions with non-zero coefficients, m substituents have limited interactions due to node 0.6 LUMO coefficients 0.0 0.6 2. early transition state structure is like reactant – determined by electron density CH3 NO2 0.52 HOMO coefficents 0.51 0.31 0.53 0.23 0.50 0.24 energies 0.26 -0.48 -0.57 III. other ring systems A. multiple rings 1. more reactive than benzene 2. form intermediate with most stable resonance forms (1 substitution is kinetically preferred, 2 substitution is thermodynamically preferred) H E E+ E H + -H+ -H+ E E B. heterocyclic 1. five membered rings- heteroatom donates a pair of electrons to aromatic ring: more reactive than benzene and directs (2) substitution (pyrrole) 4 CHEM 73/8311 CS9 Aromatic electrophilic Substitution.docx 2012March21 NH NH E+ H NH H NH NH E H E H NH H E NH H E E E 5 2. six member ring: heteroatom donates only single electron to aromatic ring: less reactive than benzene and directs to 3 position (pyridine) avoiding charge on heteroatom N E+ N N N EE E H H H IX. electrophiles A. NO2+ (AcONO2 or HNO3/H2SO4), i-Pr+ (iPrCl/GaCl3), SO3 (H2SO4), NO+ (HNO2, HCl), Cl2, H+, RCO+ (RCOCl/AlCl3) X. selected reactions A. deuteration - general-acid catalyzed B. nitration 2SO 4 HNO 3 H2SO4 H2ONO 2 HSO 4 H2ONO 2 H2O NO2 H H3O HSO4 NO 2 C 6H6 product 1. reaction is zero order in aromatic compound if aromatic is very reactive indicating formation of nitronium ion is rate determining or formation of nitronium ion is slow. CHEM 73/8311 CS9 Aromatic electrophilic Substitution.docx 2012March21 6 2. Reaction is first order in aromatic if aromatic is moderately reactive. D. Freidel Crafts Acylation either goes by acylium or complexed acid halide 1.Other Lewis acids include SbF5, TiCl4, SnCl4, BF3 2. F-C acylation is more selective than alkylation indicating acylium ions are less reactive than alkyl cation toward aromatic Cl Cl O + R Cl Al Cl O Cl Cl Al Cl R Cl Cl O Al Cl Cl R Cl Cl O C Cl Cl R O Al Cl Cl Cl R H Al R H Cl Cl Cl O Al Cl Cl Cl R O Al Cl Cl Cl E. Halogenation 1. halogenation with I2 < Br2 < Cl2 (order of reactivity) occurs directly only with more reactive aromatics 2. Lewis acid catalysts are often used, for example AlCl3, FeBr3 3. Other reagents are used HOCl, HOBr, HOI, ICl, CH3CO2Cl, CF3CO2Br 4. Note that rate laws are often complex E. Kolbe-Schmitt + O Na O C O + + H O Na O O Na OH C C O O F. deacylation and dealkylation (lone pair on CO) + HCl CHEM 73/8311 CS9 Aromatic electrophilic Substitution.docx O H 7 O O H H2SO4 2012March21 C H H O + H+ C G. protonate oxygen first? Other mechanisms? Distinguish by experiment? O O N Cl O NH Cl HCl O NH - Cl Cl O O NH NH NH H Cl H Cl Cl Cl .H. Formylation with chloroform Cl Cl Cl - OH Cl Cl H C Cl Cl - O O Cl - H2O O CH - O Cl O O CH H H OH - HO Cl CH Cl - Cl - O Cl Cl Cl CH Cl C CH Cl - O CH O - O - HO Aromatic Nucleophilic Substitution I. SNAr (AN + DN), bimolecular kinetics A. electron-withdrawing groups ortho and para, ussually requires 2 or more activating groups OMe OEt + MeO O2N NO2 OMe OEt - O2N NO2 O2N NO2 CHEM 73/8311 CS9 Aromatic electrophilic Substitution.docx 2012March21 8 B. first step is usually rate determining so rate shows little dependence on L C. replacement of halogens: F > Cl > Br > I because first step is rate determining. II. SN1 (DN + AN) A. usually with diazonium group - Y + N2 + Y N2 B. first-order in diazonium, independent of nucleophile C. tight-ion pair probably occurs D. normally occurs by radical process in the presence of Cu salts (Sandmeyer) III. benzyne (AxhDH + DN + AN + AHDxh) - favored by strong base A. NH3 NH2 14 14 14 Cl Cl NH2 14 NH2 14 + 14 NH3 NH2 14 Cl NH2 14 14 + NH2 NH2 B. first or second step is rate determing depending on leaving group 1. proton loss is rate determining for Br and I 2. leaving group loss is rate determining for F and Cl C. formation of most stable carbanion intermediate determines position of nucleophile attack NH2 + 14 CHEM 73/8311 CS9 Aromatic electrophilic Substitution.docx OMe OMe NH2 OMe - Cl OMe NH2 OMe 2012March21 OMe - NH3 Cl NH2 OMe NH2 OMe NH2 9 - OMe NH3 Cl NH2 NH2 IV. SRN1 - favored by reducing agents A. ArX G ArX G ArX Ar X Ar Y ArY ArY ArX ArY ArX B. initiated by electron transfer V. leaving groups A. for SNAr: F > NO2 > OTs > SOPh > Cl, Br, I > N3 > NR3 > OR, SR, NH2 (ipso activation) B. for SRN1: N2, halide, SPh, NMe3+, OPO(OEt)2 ???? VI. selected reactions??? A. Bucherer reactions: enamine hydroylsis CHEM 73/8311 CS9 Aromatic electrophilic Substitution.docx NH2 NH2 2012March21 10 O NH2 - OH + NH3 NaHSO3 H H2O SO3- SO3- several steps H H SO3H H H H H several steps concerted? OH OH - NaHSO3 SO3H H H B. von Richter reaction: http://www.answers.com/topic/list-of-organic-reactions#wp-V - NO2 -C O N O - - N O N O C H N - - N - O N O O N O C N- C C NH O N O H2O C N-H H2O H2O - O C N N OH C O N N OH C O- O - - OH C O OH2 OH HO N NH C O C. Sommelet-Hauser rearrangement (start from more stable anion) http://books.google.com/books?id=w9lTxIaPCQIC&pg=PA438&lpg=PA438&dq=%22so mmelet+hauser%22+rearrangement+mechanism&source=web&ots=Kve7pICAT1&sig= Dt5xzAwfRzCT2B_w71yiYKcp-F0 H H N CH3 CH3 H H H H CH3 NH 2 CH3 N CH3 CH3 2 steps note most stable anion is not reactive. N CH3 H N CH3 CH2 CH2 H CH3 CH3 CH3 2 steps CH3 N CH3 CH2