Balancing Chemical Equations
advertisement

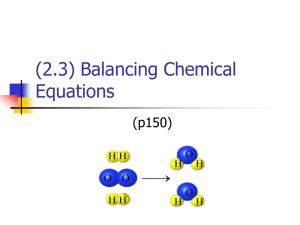
Date: Questions EQ: What are the parts of chemical equation? What is equal about equations? Why and How are the equations balanced? ENGAGE – Lego (or Skittles) activity A Chemical equations are NOT accurate in terms of QUANTITY (how much) of each reactant and product. The Law of Conservation of Mass must be met: matter can NOT be created nor destroyed in a chemical reaction. B Parts of a chemical equation: What is a “balanced” chemical equation? to yield = form=produce 4Al + 3O2 2Al2O3 (s) coefficient subscripts states of matter (# of molecules) (# of atoms) (s, l, g, aq – aqueous solution) REACTANTS What is the evidence that chemical reaction took place? When a chemical reaction is called “balanced”? PRODUCT C How have the atoms of the reactants been rearranged in the products of the reaction below? C + O2 CO2 C + O=O O=C=O BREAK FORM -Bonds between the oxygen atoms have been BROKEN -NEW BONDS have formed between the carbon atom and two O atoms. D Count the number of atoms of each element below: O2: O=2 atoms H2O: H = 2 atoms, O=1 atom Mg(NO3)2: Mg = 1, N=2, O=6 Count the number of molecules below: O2 = 1 molecule of oxygen gas 2H2O= 2 molecules of water 17HCl = 17 molecules of HCl E A balanced chemical equation is one in which each side of the equation has the same number of atoms of the same kind on both sides. Example: C (s) + O2 (g) → CO2 (g) (balanced) 1 C 2 O H2 (g) + O2 (g) → H2O (l) 2 H 2 O What are the only numbers that you can manipulate when balancing equations? F 1 C 2 O (not balanced) 2 H 1 O To balance chemical equation, you must add COEFFICIENTS (big numbers) in front of the chemical formulas in the equation. You cannot add or change subscripts!!! Why? B/c it will alter the identity of the substance otherwise. When is oxygen balanced? G Rule: Balance oxygen – O, last. If hydrogen present instead – same rule. H Balancing with shapes: _____H2 + ____O2 ____H2O You try! ____ Al2O3 ___Al + ____O2 Coefficient ratio: Balancing using T-chart: _____H2 + ____O2 ____H2O Coefficient ratio: You try! ____ Al2O3 ___Al + ____O2 R P Coefficient ratio: ___________ R P Coefficient ratio: _________ You try! (Use shapes of T-chart) ___Fe + ____O2 ____Fe2O3 R P Coefficient ratio: __________ I WE (with teacher) a. ___P4 + ____O2 _____ P4O6 Coefficient ratio: _______ b. ____AgNO3(aq) + ___Cu(s) ___Cu(NO3)2(aq) + ____Ag(s) (Hint: treat “NO3” as one element ) Coefficient ratio: _______ YOU (with a partner) c. ____C + ______H2 ____CH4 Coefficient ratio: _______ d. ____Cu2O + ____C _____ Cu +_____ CO2 Coefficient ratio: _____ I (independent practice) e. ___H2O2 ___H2 + ___ O2 f. ____Cl2 + ____KI ____KCl + ____I2 Challenge (choose your method to balance): CH4 + O2 CO2 + H2O Summary Coefficient ratio: _______ Coefficient ratio: _______