Balancing Chemical Equations
advertisement

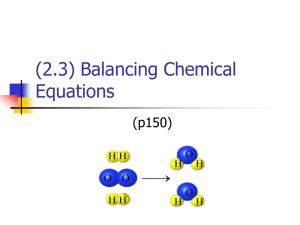
Balancing Chemical Equations Balancing chemical equations is an important skill used throughout chemistry. The Law of Conservation of Matter explains why chemical equations are balanced. It states that in a chemical reaction, matter (atoms) are not created or destroyed- the atoms are rearranged. A similar idea is the law of conservation of mass. Since matter is not created or destroyed, the total amount of mass at the beginning and the end of a reaction is the same. Let’s review a little of the material we started our study of matter with. We said that when new materials are produced, a chemical change (reaction) has taken place. A chemical equation shows this and takes into consideration the amounts of material that is started with and that are present at the end. Indications that a chemical change has occurred include bubbling, an odor, temperature changes and formation of a precipitation. A chemical equation uses symbols and formulas to show what happens during a chemical change. The materials that enter into the reaction are called the reactants. The new materials that form are called the products. Usually an arrow is between the 2 sides, pointing towards the products. The arrow can be pointed either way- so be careful on determining products and reactants. Sometimes the arrow is going both ways- showing that while new products are being made, they are breaking back into the reactants at the same time. If one arrow is bigger than the other, then that “direction” of the reaction is happening at a faster rate. There are several steps to follow to balance a chemical equation. Be careful in doing each step. A mistake early on, makes doing the rest at times impossible! You will need to start with a correctly written equation- that is all the symbols and formulas are written correctly. In the first equation you will be doing, you will be given equations with correctly written symbols and formulas. Later you will be writing your own. Second step is to count all the atoms on each side of the equation. We call this taking an inventory. I prefer the method shown. Other websites show it a slightly different way. Listing the elements down the center helps to separate the products from the reactants. Notice in this example, the polyatomic ion stays together in the inventory. In this problem it is appropriate to do that since the polyatomic ion is present on both sides of the equation. If the polyatomic ion is not present on both sides, then you cannot leave it together in the inventory. We will work on recognizing when to do this later. The third step is to add coefficients to balance the equation. A coefficient is a whole number written in front of a symbol or formula. It tells how many of the material there is. For example 2NaCl says there are 2 salt molecules. In the 2, there are 2 atoms of Na and 2 atoms of Cl. In 2Na2S, there are 4 Na’s and 2 S’s. Remember a balanced equation has the same number of each kind of atom (or polyatomic ion) on each side of the equation. Start with something that is not balanced. In the problem on the previous page, maybe you would put a coefficient 2 in front of the Al on the reactant side. Then go to the inventory and change the atom count. You will have 2 Al’s and 6 Br’s. Maybe next, maybe you would put a coefficient of 6 in front of the KBr on the product side. That would give you the 6 Br’s you need. Don’t forget to change the K’s in the inventory. You continue this process until everything is the same on both sides. The final answer for this problem would be: 2AlBr3 + 3K2SO4 6KBr + Al2(SO4)3 Keep these things in mind: *You can change a coefficient more than 1 time *Be sure to change everything in the inventory affected by the addition of a coefficient I would encourage you to watch the Khan video if want to view the process. He does these without an inventory. I would encourage you not to do this- in some problems there can be a lot to keep track off! http://www.khanacademy.org/science/chemistry/chemical-reactionsstoichiometry/e/balancing_chemical_equations Define chemical equation Define reactant Define product Define coefficient Define balanced chemical equation What is the law of conservation of mass? Why must chemical equations be balanced? List and learn the seven diatomic elements.