sup mat_nadazdy
advertisement

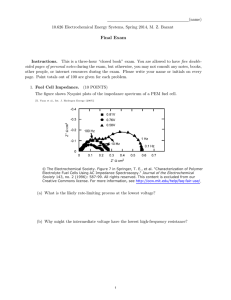
Supplementary Material For Energy resolved electrochemical impedance spectroscopy for electronic structure mapping in organic semiconductors V. Nádaždy1), F. Schauer2,3, and K. Gmucová1 1 Institute of Physics SAS, Dúbravská cesta 9, 845 11 Bratislava, Slovak Republic 2 Faculty of Education, Trnava University in Trnava, 918 43 Trnava, Slovak Republic 3 Faculty of Applied Informatics, Tomas Bata University in Zlin, 760 05 Zlin, Czech Republic A schematic experimental arrangement for the ER-EIS method is shown in Fig. S1. The three-electrode configuration of an electrochemical cell is used in the conventional manner. The electrochemical potential of the working electrode (semiconductor surface) with respect to the reference electrode (Ag/AgCl wire) is controlled by a potentiostat with a voltage source. The voltage applied to the test sample is superposition of a DC voltage U and a harmonic AC component dU = Uo sin (t) with 100 mV peak-to-peak amplitude Uo. The auxiliary electrode (Pt wire) conducts all the current needed to balance the current passing the working electrode. The magnitude of the ac component of the current at the frequency and its phase angle with respect to dU are measured as a response. These data represent electrical impedance of the semiconductor/electrolyte interface. A dependence of the real component of the impedance on the applied voltage U provides information about the electronic density of states (DOS) in organic semiconductor materials. 1 ) Author to whom correspondence should be addressed. Electronic mail: nadazdy@savba.sk Fig. S1. Block diagram of ER-EIS method The idea of the electronic structure mapping is depicted in Fig. S2. The figure shows energy band diagrams of the test sample for three different voltages applied to the working electrode (polymer film) with respect to the reference electrode (electrolyte solution). The electrochemical potentials of the semiconductor and the electrolyte solution are represented by the Fermi levels EF and EF,electrolyte, respectively. When an external voltage U is applied, the positions of the band edges at the semiconductor surface do not change with respect to the solution due to a rather low conductivity of the semiconductor. However, the position of the Fermi level of the semiconductor changes with respect to the solution potential (eU = EF EF,electrolyte). Therefore, a variation of the applied voltage U in the surface region results in a bending of the bands. If the voltage U applied to the polymer is positive (negative), the band is bent upwards (downwards) and the electronic DOS are mapped at the energy below (above) the energy of the Ag/AgCl reference electrode -4.66 eV, which is related to the zero vacuum energy level. Fig. S2. Energy band diagrams of the test sample for three different voltages U applied to the polymer film with respect to the reference electrode: (a) U > 0V, (b) U = 0V, (c) U < 0V.