pat3701-sup-0001-SI
advertisement
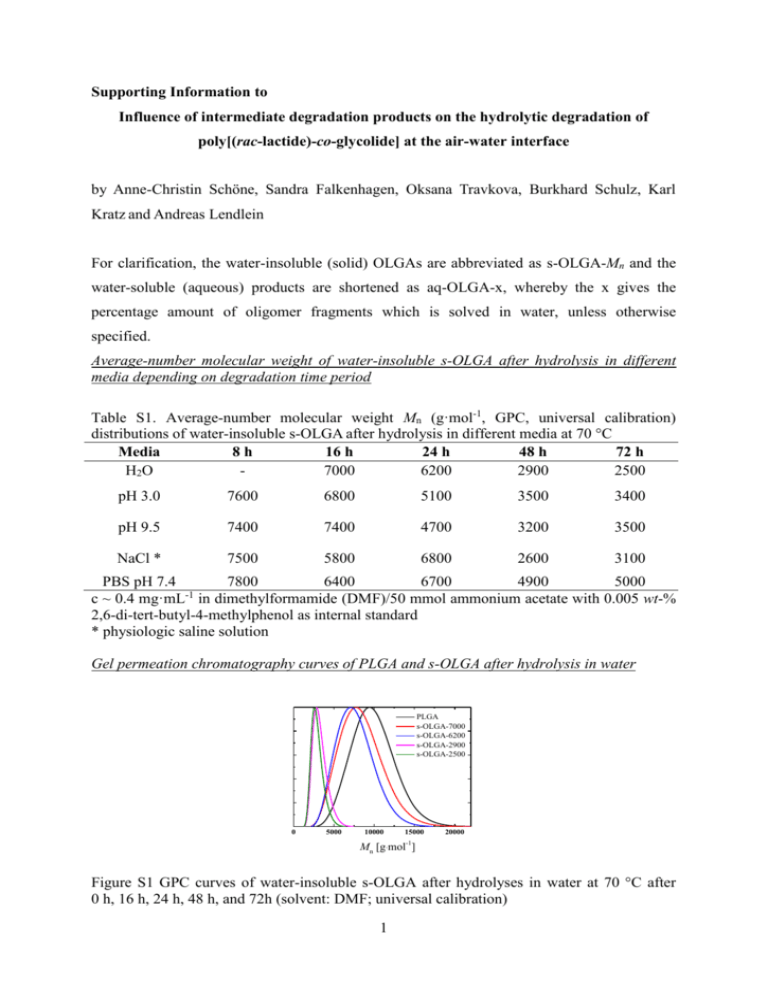
Supporting Information to Influence of intermediate degradation products on the hydrolytic degradation of poly[(rac-lactide)-co-glycolide] at the air-water interface by Anne-Christin Schöne, Sandra Falkenhagen, Oksana Travkova, Burkhard Schulz, Karl Kratz and Andreas Lendlein For clarification, the water-insoluble (solid) OLGAs are abbreviated as s-OLGA-Mn and the water-soluble (aqueous) products are shortened as aq-OLGA-x, whereby the x gives the percentage amount of oligomer fragments which is solved in water, unless otherwise specified. Average-number molecular weight of water-insoluble s-OLGA after hydrolysis in different media depending on degradation time period Table S1. Average-number molecular weight Mn (g·mol-1, GPC, universal calibration) distributions of water-insoluble s-OLGA after hydrolysis in different media at 70 °C Media 8h 16 h 24 h 48 h 72 h H2O 7000 6200 2900 2500 pH 3.0 7600 6800 5100 3500 3400 pH 9.5 7400 7400 4700 3200 3500 NaCl * 7500 5800 6800 2600 3100 PBS pH 7.4 7800 6400 6700 4900 5000 c ~ 0.4 mg·mL-1 in dimethylformamide (DMF)/50 mmol ammonium acetate with 0.005 wt-% 2,6-di-tert-butyl-4-methylphenol as internal standard * physiologic saline solution Gel permeation chromatography curves of PLGA and s-OLGA after hydrolysis in water PLGA s-OLGA-7000 s-OLGA-6200 s-OLGA-2900 s-OLGA-2500 0 5000 10000 15000 20000 -1 Mn [g·mol ] Figure S1 GPC curves of water-insoluble s-OLGA after hydrolyses in water at 70 °C after 0 h, 16 h, 24 h, 48 h, and 72h (solvent: DMF; universal calibration) 1 1 H-NMR spectra of water-insoluble s-OLGA after hydrolysis in water 1 H-NMR: δ = 1.4 (C-COOH (f), lactic acid), δ = 1.56 (-CH3-, lactic acid), δ = 3.48, δ = 4.25 (-O-CH2-, initiator), δ = 4.8 (C-COOH (e), glycolic), δ = 4.90-5.05 (-CH2-, glycolic acid), δ = 5.1-5.3 (-CH-, lactic acid) a) c DMF a e b f d s-OLGA-7500 s-OLGA-7000 s-OLGA-6200 s-OLGA-2900 s-OLGA-2500 b) s-OLGA-6200 s-OLGA-7500 s-OLGA-8300 Figure S2 NMR spectra of water-insoluble s-OLGA after hydrolysis in water at 70 °C a) 1HNMR after 8 h, 16 h, 24 h, 48 h, and 72 h and b) 13C- NMR after 8 h and 24 h and PLGA FTIR-spectroscopic measurement results of PLGA and water-insoluble s-OLGA The IR spectra (Figure S3) illustrate all characteristic groups for PLGA: a broad peak around 3500 cm-1 is attributed to the OH stretch vibration, a triplet band between 2995 and 2881 cm -1 corresponding to CH3 and CH2 symmetric and asymmeric stretching with the additional band 2 at 1423 cm-1 for C-H stretching vibration; the main ester bands of the C=O stretching vibration near 1750 cm-1, and the C-O-C asymmetric and symmetric stretching vibrations in the range of 1184 – 1090cm-1, which have been identified on the IR spectra of all investigated polymer samples of water-insoluble fraction. With the increasing hydrolysis degree the split and the red-shift in C=O stretching vibration occurs showing the appearance of the carboxylic groups and their coexistence with ester groups. The increase in the O-C=O deformation vibration at 974 – 870cm-1 attributed to carboxylic group was detected indicating a gradual hydrolysis of ester bonds with simultaneous generation of carboxyl end-groups. The split and the blue-shift of C-O-C asymmetric stretching vibrations (1169 → 1184 cm-1) can be a sign of particular cyclization of hydrolysis products in form of lactones.[55] PLGA s-OLGA-7500 s-OLGA-6200 s-OLGA-2900 2000 1800 1600 1400 1200 1000 800 600 -1 wavenumber (cm ) Figure S3: IR spectra of PLGA and three different s-OLGAs with different ratio of the number of functional end-groups to the molecular weight High-performance liquid chromatography measurements for water-soluble aq-OLGA hydrolysis in water The characterization of the two degradation products lactic acid and glycolic acid was analyzed by Yoo et al.[16] under different conditions by liquid chromatography showing a faster release of the glycolide compared to lactide. But it is still challenging to characterize the intermediate degradation products – namely the oligomers of lactic acid and glycolic acid.[56] Therefore, also other degradation fragments next to lactic acid and glycolic acid cannot to be excluded from the degradation process. The amount of lactic acid in the water-soluble fraction increased about 50 times from a degradation time of 24 h to 72 h (Table S2), which nevertheless just suggests that at the beginning of the degradation the amount of produced acid is negligible comparing to further 3 hydrolysis products, and while increasing in the absolute mass the relative content remains unchanged. Table S2: Lactic acid area, lactic acid rel. area and amount of lactic acid of partly degraded sOLGA samples determined by high performance liquid chromatography Area of lactic acid Rel. area of Lactic Amount lactic acid Sample ID [µRIU·min] acid [%] [g·L-1] s-OLGA-7 0.04 21.3 0.04 s-OLGA-32 1.01 27.6 0.92 s-OLGA-57 2.19 28.6 2.00 a) The area and rel. area of lactic acid, respectively, refer to the integral of the peak corresponding to the lactic acid Molecular weight of PLGA and water-insoluble s-OLGA after hydrolysis in different media determined by GPC and OH-group titration Gel permeations chromatography (GPC) The determination of the number molecular weight (Mn) by GPC investigations was performed using 50 mmol ammonium acetate in N,Ndimethylformamide at 35 °C as eluent with a flow rate of 0.25 mL·min-1 and 0.005 wt-% 2,6di-tert-butyl-4-methylphenol as internal standard for all samples. The system was equipped with 250 mm x 4.6 mm GRAM gel columns, 3 and 3 x 102 nm porosity, 10 µm particle size (Polymer Standard-Service GmbH, Mainz, Germany, PSS), a degasser (ERC-3315, Riemerling, Germany), a gradient pump PU 980 and an automatic injector AS-851 (both Jasco, Tokyo, Japan). Three detectors were used: a multi wavelength detector MD-910 (270 nm), a RI-930 (both Jasco) and the viscosimeter n-1001 (WGE, Dr. Bures, Dallgow, Germany), which were combined by a split. All molecular weights were determined using a universal calibration with polystyrene standards (PSS). End-group titration The content of carboxyl groups (COOH-value) and or hydroxyl groups (OH-value) were determined as the acid number (mgKOH/gpolymer) or respectively the hydroxyl number (mgKOH/gpolymer) by potentiometric titration of the copolymers dissolved in dimethylformamide (DMF, Merck), with 0.1 N-tetrabutylammonium hydroxide (Merck). The titrations were performed with the titrator DMS Titrino 716 (Metrohm, Schwizerland) and a Solvotrode with LiCl (sat.) in ethanol. The carboxyl groups were directly titrated but the determination of the hydroxyl groups is based on the esterification of the OH-groups with 4 acetic anhydride (abundantly, Sigma-Aldrich) in presence of N-methylimidazole (SigmaAldrich) as catalyst at ambient temperature and by back titration. Table S3 Average-number molecular weight Mn (g·mol-1) determined by GPC (universal calibration) and OH-group titration of PLGA and water-insoluble s-OLGA after hydrolysis in water at 70 °C for different time periods. Sample ID are abbreviated as s-OLGA-degradation time in h OH-value [g∙mol-1] GPC [g∙mol-1] 10000 4200 s-OLGA-8h 7200 4500 s-OLGA-24h 4400* 3800 Sample ID PLGA # s-OLGA-48h 2300* 3100 * Determined by taking into account the COOH-value determined for s-OLGA-24h (28.6 mg∙g-1) and s-OLGA-48h (35.0 mg∙g-1), respectively # The time refers to the correlated degradation time Brewster angle microscopy images of PLGA and s-OLGA a) PLGA b) s-OLGA-7500 c) s-OLGA-2900 100 µm Figure S4: BAM images in dependence on the grade of PLGA hydrolysis recorded at room temperature on a pure water subphase at a) = 11.0 mNm-1, b) = 12.8 mNm-1, c) = 9.8 mNm-1 (s-OLGA-7500 and s-OLGA-2900 are degraded for 8 h and 48 h, respectively) 5