Quiz 1 Ans
advertisement

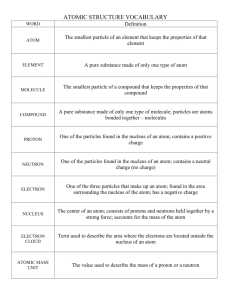
Year 9 Advanced Science Chemistry Quiz ONE 2015 [25 marks] ANSWERS Name: MR. HUNG 1. Give 2 similarities between neutron and proton. [2 marks] Neutron and proton are found in the nucleus of an atom Both have a mass of unit 1 – much heavier than electron. Both contribute to the mass of an atom These sub-atomic particles cannot leave the atom. (Any TWO) 2. Give 2 differences between proton and electron. [2] Proton is found in the middle nucleus and Electron is surrounding the nucleus. Proton has a positive charge and electron has a negative charge Proton is much heavier than electron. (Any TWO) 3. How to find the neutron number of an atom? [2] Mass number is the sum of Proton number and Neutron number So, Neutron number = Mass number – Proton number OR The difference between the Mass number and Proton number 4. State the name and find the neutron number of the following elements.[4] Helium Fluorine 4-2 = 2 neutrons 19-9 = 10 Lithium 7-3 = 4 Uranium 235 – 92 = 143 Bromine 80-35 = 45 5. What is the electronic configuration of the following element? [3] Eg. ( 2, 8, 4) (2, 8, 8) (2, 8, 3) (2, 6) 6. What is OCTET rule? Which group and elements have this octet arrangement? [5] It states that atoms of main-group elements tend to combine in such a way that each atom has eight electrons in its valence shell, giving it the same electronic configuration as a noble gas. elements gain or lose electrons to become ions. Helium, Argon and Neon. 7. a) Write an equation for the following reaction during the burning of gas in a Bunsen burner. The following are the chemicals involved: Oxygen, Carbon Dioxide, Methane, Water. [2] Methane + Oxygen --------- Water and Carbon Dioxide b) What are the other products (energy) formed during this reaction? [2] Heat and Light from the flame 8. What are the differences between ANION and CATION? Give an example of each type. [4] An ANION is a negative ion formed from Non-metal atom after gaining electron(s) Eg. Chloride ion C1 A CATION is a positive ion formed from Metal atom after losing electron(s) Eg. Sodium ion Na+