pmic7840-sup-0010-test
advertisement

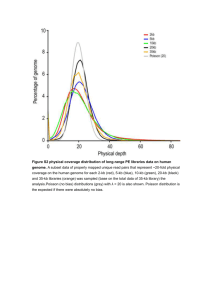
Materials and Methods: Data: Mass spectral dataset for ME-AM1 was downloaded from PRIDE database [1] and was part of the study by Schneider, K et. al.[2]. Briefly, Tandem mass spectrometry data was generated from ME-AM1 grown on acetate and on methanol (Three biological replicates for each substrate). Proteins from ME-AM1 cells were separated by 1D SDS/PAGE and were followed after trypsin digestion on reverse phase HPLC coupled to a LTQ-orbitrap high accuracy mass spectrometer [2]. Six spectral files with accessions 17691-17696 were downloaded and converted to common search format MGF(Mascot generic format) using PRIDEinspector tool [3]. In total 538,820 tandem mass spectra were present in six files. Genomic similarity/diversity estimation: dnadiff[4] utility from MUMmer suite was used to compare genome sequences with default parameters. Genome identity percentages for reference and query genes are the percentage of aligned bases reported by dnadiff. While reporting average base pair identity between genomes, M to M alignments were considered. The total non-redundant gene pool or Pan genome of the Methylobacteriacea family was constructed by clustering all protein sequences from individual methylobacterial genomes using CD-HIT[5] at 70% identity over 70% of query sequence criterion. The cluster thus generated was processed using in-house Perl scripts to generate a Phylogenetic Profile Matrix, where presence or absence of a protein coding feature in any genome was represented by 1 or 0. Ultimately this matrix was used to determine categorical existence of proteins. WebMGA[6] server was used to annotate COG functional classes for a set of gene sequences. Proteogenomic analysis: Raw mass spectra were searched against the reference genome of ME-AM1 (NC_012808). Genome translation, peptide identification by multiple search algorithms, FDR estimation and novel peptide identification were carried out in an automated fashion by GenoSuite framework recently developed by our group[7]. All four search algorithms, supported by GenoSuite, namely OMSSA[8], X!Tandem[9], InsPecT[10] and MassWiz[11] were deployed for peptide identification. Search parameters were 20 ppm precursor ion tolerance, 0.1 Da product ion tolerance, trypsin as proteoase enzyme with one missed cleavage allowed, carbamydomethylation of cysteine as fixed modification and oxidation of methionine was used as the variable modification. Target database was composed of a six frame translated genome, proteins from the other four replicons and common contaminants from CRAP (ftp://ftp.thegpm.org/fasta/cRAP) database. All 115 protein sequences from CRAP database were included in the search database. All the sequences were reversed to create the decoy database. A concatenated target-decoy search approach was used and in total database had 118,664 protein entries. FDR (in %)for individual algorithmsis calculated using following formula. 𝐹𝐷𝑅 = 𝐷 × 100 𝑇 D= count of decoy PSMs passing the threshold T= count of target PSMs passing the threshold Results from multiple algorithms were integrated using FDRscore[12] method. All these steps are implemented in the GenoSuite pipeline. To infer proteins, identified peptides were mapped back to the search database having 118,664 proteins. Only proteins with ≥2 peptides or single peptide hits with ≥5 significant PSMs were considered in the final report.At this criterion protein identification FDR was calculated as follows 𝑃𝑟𝑜𝑡𝑒𝑖𝑛 𝐹𝐷𝑅(%) = 𝐼𝑑𝑒𝑛𝑡𝑖𝑓𝑖𝑒𝑑 𝑑𝑒𝑐𝑜𝑦 𝑝𝑟𝑜𝑡𝑒𝑖𝑛 𝑐𝑜𝑢𝑛𝑡 × 100 𝐼𝑑𝑒𝑛𝑡𝑖𝑓𝑖𝑒𝑑 𝑡𝑎𝑟𝑔𝑒𝑡 𝑝𝑟𝑜𝑡𝑒𝑖𝑛 𝑐𝑜𝑢𝑛𝑡 Data visualisation: To analyze the conservation of novel proteins across closely related genomes we used BRIG[13] tool with blastx option to search the genomes against proteins. To visualize peptide identifications against the whole genome of MEAM1 a peptide co-ordinate file was created using in-house developed scripts and Circos[14] was used for visualization of this data. To mark methylotrophy islands we used methylotrophy gene information from Vuilleumier, S et. al. [15]. To visualize genomic context of individual proteins identified from mass spectrometry data, we used ORFmapper utility provided in GenoSuite. Peptide spectrum matches (PSMs) were visualized for manual inspection using pLabel utility of pFind[16] studio with 0.5 Da tolerance. ORF prediction and function prediction: To impart additional confidence to novel proteins abinitio predictions by GENEMARK, Prodigal, Glimmer and FgeneSB were compared. This was done by comparing co-ordinates of gene predictions with that of novel genesusing in-house scripts. The following steps describe how each of these algorithms were employed for gene prediction GeneMark – GeneMarkS (v4.6b) was used to create the training model and prokaryotic GeneMark.hmm (v2.8) was employed to predict genes in ME-AM1 using the model obtained from GeneMarkS, while considering RBS for gene start prediction. Prodigal – version 2.60 was used with standard parameters to obtained gene prediction. Glimmer – The current version 3.02 of the software suite was obtained for analysis. Using the “g3-from-scratch.csh” script of the glimmer suite, gene prediction was performed on the nucleotide sequences for the genome of MEAM1. The program “g3-from-scratch.csh” runs a series of programs to sequentially predict putative orfs to be used as a training set and to build model, which is eventually used by the main program glimmer3 for the final stage of gene prediction. Fgenesb – the online implementation of fgenesb was used to predict genes in ME-AM1 with BradyrhizobiumjaponicumUSDA110 as a close organism. For functional prediction of proteins, PFAM (http://pfam.xfam.org/search#tabview=tab1) and Goanna (http://www.agbase.msstate.edu/cgi-bin/tools/GOanna.cgi) web tools were used with their default settings. For PFAM default settings include an e-value threshold of 1.0 for pfam-A database and 0.001 for pfam-B search. Goanna parameters were Database: UniProt, Expect: 10e-1, Matrix: BLOSUM62 and word size:3. Normalized spectral abundance factor (NSAF): In-house scripts were used to calculate NSAF for identified proteins in all six samples independently. For comparisons between growth conditions, average NSAF was calculated for every protein. To calculate NSAF we followed the following formula proposed by Zybailov, B et. al. [17] and considered only spectra which identified unique peptides at <1% FDR. (𝑁𝑆𝐴𝐹)𝑘 = (𝑆𝑝𝐶/𝐿)𝑘 𝑁 ∑𝑖=1(𝑆𝑝𝐶/𝐿)𝑖 Where, SpC= Total number of the qualified spectra assigned to protein L= Length of the protein N= Total number of proteins identified in the study Reference List 1. Martens, L., Hermjakob, H., Jones, P., Adamski, M. etal., PRIDE: the proteomics identifications database. Proteomics. 2005, 5, 3537-3545. 2. Schneider, K., Peyraud, R., Kiefer, P., Christen, P. etal., The ethylmalonyl-CoA pathway is used in place of the glyoxylate cycle by Methylobacterium extorquens AM1 during growth on acetate. J.Biol.Chem. 2012, 287, 757-766. 3. Wang, R., Fabregat, A., Rios, D., Ovelleiro, D. etal., PRIDE Inspector: a tool to visualize and validate MS proteomics data. Nat.Biotechnol. 2012, 30, 135-137. 4. Kurtz, S., Phillippy, A., Delcher, A. L., Smoot, M. etal., Versatile and open software for comparing large genomes. Genome Biol. 2004, 5, R12. 5. Li, W. and Godzik, A., Cd-hit: a fast program for clustering and comparing large sets of protein or nucleotide sequences. Bioinformatics. 2006, 22, 1658-1659. 6. Wu, S., Zhu, Z., Fu, L., Niu, B., and Li, W., WebMGA: a customizable web server for fast metagenomic sequence analysis. BMC.Genomics 2011, 12, 444. 7. Kumar, D., Yadav, A. K., Kadimi, P. K., Nagaraj, S. H. etal., Proteogenomic analysis of Bradyrhizobium japonicum USDA110 using GenoSuite, an automated multialgorithmic pipeline. Mol.Cell Proteomics. 2013, 12, 3388-3397. 8. Geer, L. Y., Markey, S. P., Kowalak, J. A., Wagner, L. etal., Open mass spectrometry search algorithm. J.Proteome.Res. 2004, 3, 958-964. 9. Craig, R. and Beavis, R. C., TANDEM: matching proteins with tandem mass spectra. Bioinformatics. 2004, 20, 1466-1467. 10. Tanner, S., Shu, H., Frank, A., Wang, L. C. etal., InsPecT: identification of posttranslationally modified peptides from tandem mass spectra. Anal.Chem. 2005, 77, 4626-4639. 11. Yadav, A. K., Kumar, D., and Dash, D., MassWiz: a novel scoring algorithm with target-decoy based analysis pipeline for tandem mass spectrometry. J.Proteome.Res. 2011, 10, 2154-2160. 12. Jones, A. R., Siepen, J. A., Hubbard, S. J., and Paton, N. W., Improving sensitivity in proteome studies by analysis of false discovery rates for multiple search engines. Proteomics. 2009, 9, 1220-1229. 13. Alikhan, N. F., Petty, N. K., Ben Zakour, N. L., and Beatson, S. A., BLAST Ring Image Generator (BRIG): simple prokaryote genome comparisons. BMC.Genomics 2011, 12, 402. 14. Krzywinski, M., Schein, J., Birol, I., Connors, J. etal., Circos: an information aesthetic for comparative genomics. Genome Res. 2009, 19, 1639-1645. 15. Vuilleumier, S., Chistoserdova, L., Lee, M. C., Bringel, F. etal., Methylobacterium genome sequences: a reference blueprint to investigate microbial metabolism of C1 compounds from natural and industrial sources. PLoS.One. 2009, 4, e5584. 16. Wang, L. H., Li, D. Q., Fu, Y., Wang, H. P. etal., pFind 2.0: a software package for peptide and protein identification via tandem mass spectrometry. Rapid Commun.Mass Spectrom. 2007, 21, 2985-2991. 17. Zybailov, B., Mosley, A. L., Sardiu, M. E., Coleman, M. K. etal., Statistical analysis of membrane proteome expression changes in Saccharomyces cerevisiae. J.Proteome.Res. 2006, 5, 2339-2347.