Supplementary Material Post-transplant immune suppression and
advertisement

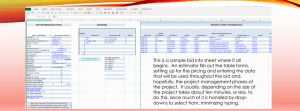
Supplementary Material Post-transplant immune suppression and anti-infective prophylaxis Tacrolimus (FK) was administered to achieve a 12-hour serum trough level of 8-10 ng/mL for the first six months post-LT, 5-8 ng/mL from 6-12 months post-transplant and 3-5 ng/mL for > 12 months post-LT. LT recipients with kidney disease received FK with lower goal trough levels (5-8 ng/mL for the first six months post-LT followed by 3-5 ng/mL beyond six months) and mycophenolate mofetil at target dose of 1.5 to 2.0 g daily (or equivalent dosage of mycophenolic acid). All recipients received opportunistic infection (OI) prophylaxis with valganciclovir, trimethoprim-sulfamethoxazole, and nystatin suspension with additional prophylaxis as indicated by plasma CD4+ T lymphocyte cell count/mm3 (CD4) or prior OI. DAA doses Standard doses of SOF (400 mg daily), SMV (150 mg daily), and RBV (400 mg daily AM, 600 mg daily PM for weight ≤ 75 kg) were utilized. Laboratory Monitoring Plasma hepatitis C virus RNA levels were quantified using COBAS® AmpliPrep/COBAS® TaqMan® HCV Quantitative Test, v2.0 (Roche Diagnostics, Indianapolis, IN; lower limit of detection (LLD) of 15 IU/mL with the exception of one EOT HCV RNA quantification performed at an outside laboratory (LLD 43 IU/mL). HCV genotype (GT) was determined by the VERSANT® HCV Genotype 2.0 Assay (LiPA; Siemens Medical Solutions Diagnostics, Tarrytown, NY). Supplementary Table 1: Specific patient characteristics prior to and during DAA therapy Age Gender Race HCV GT Prior IFN-Tx #38 54 Male Hispanic 2b Yes #43 56 Male White 1a Yes #44 39 Male Hispanic 1aa No #89 44 Male Black 1b Yes #93 48 Male White 1a No Co-morbidities CKD CKD, HCCb Obesity CKD CKD, DM HIV Diagnosis Risk for HIV/HCV 1980 IVDU PJP, candidiasis 184 (17) < 20 ABC + 3TC + RAL 1989 Hemophilia 2004 IVDU, MSM 2007 MSM None None 464 (35) <20 TDF + FTC + EFV TDF + FTC + RAL 6/7/08 193 (35) 26 TDF + FTC + RAL Prior OI CD4 (%)d HIV RNAe Initial cART Regimen cART at DAA Start LT Date LT Characteristic/ Complication Post-LT Infections No change 7/29/12 Hepatic artery stenosis CDI, Influenza, PNA, MRSA SSTI 1993 MSM #94 39 Male White 1a No CKD, FTT, MSOFc 1985 Hemophilia #101 60 Male White 1a No CKD, CAD, HCCb 1985 IVDU #110 67 Male Hispanic 1a No CKD, DM, HCCb 1994 MSM PJP No MAC None None 405 (36) <20 TDF + FTC + RAL 44 (25) <20 ABC + 3TC + RAL No change No change 2/4/13 324 (35) <20 ABC + 3TC + EFV ABC + 3TC + DTG 11/26/13 12/3/11 5/29/13 360 (27) <20 TDF + FTC + EFV TDF + FTC + RAL 7/23/09 150 (28) <20 TDF + 3TC + ATV/r TDF + FTC + RAL 4/24/07 None HCV+ donor None DCD graft Living donor graft Re-do LT for HAT None None None None Thrush, CDI E.coli peritonitis w/ bacteremia SSTI None Moderate cellular rejection 2008 7/25/13: F0, A1 1,725,138 1.53 2.19 Prior Graft Rejection None Mild cellular rejection 6/24/08 Latest Liver Biopsyf HCV RNAg APRI FIB-4 3/6/13: F0, A1 63,607,896 2.09 4.27 6/1/09: F0, A1 3,547,068 0.73 2.62 No change None – (moderate cellular rejection, post-DAA) 5/23/13: F0-F1, A1 2,077,031 0.65 2.22 Mild cellular rejection 12/4/13 None None Moderate cellular rejection 8/14/09 12/4/13: Mild ACR 8,679,828 0.41 1.29 1/22/13: F0 2,212,930 0.85 2.02 12/23/13: NRH 37,508,565 8.60 16.12 4/25/12: F2, A1 552,941 1.09 3.44 MELD CP Score 10 N/A 11 N/A 9 N/A 12 N/A Initial IS Regimen FK 1 mg AM, 0.5 mg PM FK 1 mg BID FK 0.5mg BID FK 5mg BID, MMF 250 mg BID IS at DAA Start FK 0.5 mg BID FK 0.5 mg BID Same 27 11C 14 N/A 7 N/A Pred 2.5mg QD, FK 0.5mg BID FK 1.5mg BID FK 0.5mg q 10 days Same Same Same FK 0.5 mg BID No change No change 3/26/14 SOF + SMV 1/17/14 SOF + SMV Unable to assess patient intubated DAA Start Date DAA Regimen FK 1 mg AM, 0.5 mg PM 4/15/14 SOF + RBV 3/18/14 SOF + SMV Reported Side Effect Congestion, Mild Fever Mental fogginess Back pain Headache Fatigue N/A Heroin relapse. Graft rejection (after DAAs) N/A N/A IS Change During DAA Other Adverse Event N/A None None increased after DAA 5/21/14 SOF + SMV FK 3 mg BID, MMF 250 BID FK 1 mg BID, MMF 250 BID 9/25/14 SOF + SMV 11 N/A FK 0.5mg qAM, 1mg qPM; MPA 350 BID Death None increased after DAA 6/1/14 SOF + SMV FK 1 mg BID 8/7/14 SOF + SMV Fatigue, peripheral edema None reported N/A N/A Abbreviations: DAA, direct-acting antiviral; HCV, hepatitis C virus; GT, genotype; IFN-Tx, interferon-based treatment; CKD, chronic kidney disease (GFR < 60 mL/min/1.73 m2 for ≥ 3 months); HCC, hepatocellular carcinoma; DM, type 2 diabetes mellitus; FTT, failure-to-thrive; MSOF, multi-system organ failure; CAD, coronary artery disease; HIV, human immunodeficiency virus; IVDU, intravenous drug use; MSM, men who have sex with men; OI, opportunistic infection; PJP, pneumocystis jirovecii pneumonia; MAC, mycobacterium avium complex; CD4, CD4+ T-lymphocyte count; cART, combined antiretroviral therapy; ABC, abacavir; 3TC, lamivudine; TDF, tenofovir; FTC, emtricitabine; RAL, raltegravir; DTG, dolutegravir; ATV/r, ritonavir-boosted atazanavir; LT, liver transplant; DCD, donation after cardiac death; HAT, hepatic artery thrombosis; CDI, clostridium difficile infection; PNA, pneumonia; MRSA, methicillin-resistant staphylococcus aureus; SSTI, skin or soft tissue infection; ACR, acute cellular rejection; NRH, nodular regenerative hyperplasia, APRI, AST-toplatelet ratio index; FIB-4, fibrosis-4 score; MELD, model for end-stage liver disease; CP, Child-Pugh; IS, immunosuppression; FK, tacrolimus; MMP, mycophenolate mofetil; MPA, mycophenolic acid; pred, prednisone; SOF, sofosbuvir; RBV, ribavirin; SMV, simeprevir a Pre-transplant GT 3, however after LT with HCV+ liver GT 1a confirmed b HCC present prior to LT c Patient with respiratory failure requiring mechanical ventilation, liver failure, recurrent gastrointestinal bleeding, hypotension requiring vasopressor support, protein-calorie malnutrition, extra-vascular volume overload with pleural effusions, ascites and peripheral edema d Measured in cells/mm3 e Measured in copies/mL f Most recent liver biopsy prior to DAA initiation g Measured in IU/mL