Re-Teach
advertisement

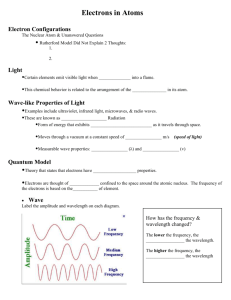
AP Chemistry August 29 to September 2 Goals: 1. 2. 3. 4. Light, Waves, and Electrons Electronic Structure Quantum Numbers Naming Binary Molecular Compounds and Acids Objectives: When finished the student will be able to: 1. calculate the wavelength, frequency, and energy of a photon of light. 2. compare and contrast the wave, particle duality of light. 3. calculate the energy level transition of an electron within a hydrogen atom given the energy, frequency, or wavelength of light emitted or absorbed during the transition. 4. use the de Broglie equation to determine the wavelength of a moving electron. 5. explain why there is an uncertainty in the location of an electron in an atom. 6. use quantum numbers to define the region of space an electron occupies. 7. write the name or formula of a binary molecular compound or an acid. Lessons: Monday: Opener: What do light, waves and electrons have in common? Activity: o Begin Lecture 2 and complete Lecture 4 Assessment: o Question and answer Homework: o Review Worksheets 2 and 4 Tuesday: Opener: Where are those electrons? Activity: o Complete Lecture 2 Assessment: o Question and answers Homework: o Be sure that worksheets 2 and 4 are completed. o Complete Prelab questions for experiment 1 Wednesday: Opener: Let’s take a breather. Activity: o Give quiz over Lecture 1 and Nomenclature of ionic compounds o Group review of worksheets 2 and 4 Assessment: o Question and answer o Collect Prelab questions for Lab 1 Homework: o Read and Prepare for Lab 1 Thursday: Opener: Prepare for Lab Activity: o Lab 1 – Formula of Magnesium Oxide Assessment: o Question and answer o Lab observations Homework: o Review lecture notes 3 o Catch Up Time Friday: Opener: Work on Lab Activity: o Finish Lab 1 – Formula of Magnesium Oxide o Begin Lecture 3 Assessment: o o Question and answer Observe lab techniques Homework: o Begin work on lab report o Review Lecture Notes 3 Re-Teach: Individual Student(s) One on one teacher to student One on one peer tutoring Small group and teacher interaction Entire Class Review using different approach and/or method