procedure for calculating power plant emissions
advertisement

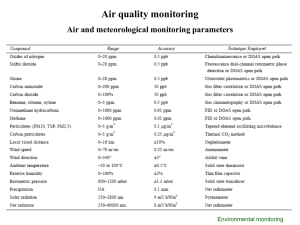
PROCEDURE FOR CALCULATING POWER PLANT EMISSIONS By: Marcial T. Ocampo September 17, 2009 Calculating power plant emissions (mg SOX / Nm3, mg NOX / Nm3, mg CO / Nm3 or as ppmv SOX, ppmv NOX, ppmv CO) is sometimes difficult for ordinary power plant engineers. Carbon dioxide emissions are usually given in % by volume dry or as kg CO2 per kWh generation. Well, your favorite energy technology expert has developed a spreadsheet model (in MS Excel) for calculating in a breeze the common power plant emissions. SO2 and CO2 emissions are readily calculated from fuel analysis (by mass for solid and liquid fuels, by volume for gaseous fuels) and typically measured by Orsat or other gravimetric methods. On the other hand, NOX and CO are result of thermal dissociation of N2 into NOx at high combustion temparature while CO is the result of incomplete combustion due to insufficient O2 or inadequate fuel and air mixing. Hence, NOX and CO are measured using instruments and later computed as mg NOX / Nm3 or mg CO / Nm3). (Nm3 means normal cubic meter of gas). The basic steps followed by my model are as follows: Basic steps: 1) Input natural gas (fuel) analysis: % volume (same as % mol), molecular weights: e.g. H2, CH4, C2H6, C3H8 ... CO2, S, O2, N2, H2O moisture, ash. 2) Convert % volume to ultimate analysis % mass or weight (%C, %H2, % S, % O2, %N2, %H2O moisture, ash) 3) From the combustion equations; C + O2 = CO2 S + O2 = S02 H2 + 1/2 O2 = H20 calculate the stoichiometric O2 in mols and lbs and that of N2 from air analysis. 4) Using the excess %O2 wet basis measurement in the flue gas, the level of excess air ratio is computed. Use this excess air ratio to compute actual excess mass of O2 and N2 to compute the total flue gas (fuel + air). 5) Convert the mass to mols using their molecular weights and convert to % volume. The computed %O2 must tally with the measured %O2 wet basis. 6) Compute dry flue gas (total flue gas - H2O moisture) and calculate %O2 dry basis. 7) Input the generator output MWe (gross), fuel firing rate (m3/s), fuel density (kg/m3), fuel lower heating value (LHV in kJ/kg). Compute firing rate in kg/s, gross heat rate of power plant in kJ/kWh and thermal efficiency in % LHV basis. 8) Using 100 lbs of fuel as basis, convert the mass of each flue gas component into kg/s and kg/MWh. 9) Input the emission measurements in mg/Nm3 and kg/h of CO, NO2, SO2 and dust. Convert to kg/s and kg/MWh. 10) Due to partial combustion to CO and formation of NO2, the previous results for CO2, O2 and N2 has to be corrected for C, O and N that should have gone to CO2, O2 and N2 had there been complete combustion to CO2 and no dissociation of O2 and N2: C + 1/2 O2 = CO 1/2 N2 + O2 = NO2 11) After correcting the kg/MWh of CO2, O2 and N2, we will have finally the corrected flue gas composition consisting of CO2, CO, NO2, SO2, O2, N2, H2O and dust. Using their molecular weights, the kg/MWh is converted to mol/MWh and % mol or % volume. Compute the resulting analysis in wet basis and % O2 measured dry basis. 12) Finally, correct the measured mg/Nm3 or ppmv to 6% O2 reference from the %O2 measured dry basis. (X, O2 ref) = (X, meas) * (21 - O2 ref) / (21 - O2 meas) where: X = CO, NO2, SO2, dust, etc. The relationship between mg/Nm3 and ppmv (ppm volume) is defined by the molecular weight of the component X and the standard molal gas volume of 0.022413 m3/mol: (mg/Nm3 of X) = (ppmv of X) / 1000 x (M.W. of X) / (0.022413 m3/mol) or (ppmv of X) = (mg/Nm3 of X) * 1000 * (0.022413 m3/mol) / (M.W. of X) where: ppmv = parts per million (volume) 1000 = (1,000,000 / million) / (1,000 mg/g) M.W. of X = molecular weight of X, g/mol Nm3 = normal cubic meter (at STP - 0 deg C, 1 atm) 13) The corrected emissions at 6% O2 is then compared with the Clean Air Act standards and with other world standards for comparison. It can also be compared with the supplier/contractor's guaranteed emission performance. 14) Compute the year-to-date or annual power generation in MWh and multiply with the average emission in kg/MWh of CO2, CO, NO2, SO2 and particulates (dust). Convert the annual emissions to metric tons per year. Compare with allowable emission levels for the particular area. I would like to invite you and your company to continue supporting this blog thru the DONATE button. You may order my power generation technology articles and project financial models thru the ENERGY DATA page. Thanks! Marcial T. Ocampo (Friendly note: All content written by Engr. Marcial T. Ocampo are copyrighted and may not be redistributed in any way or form.) A spreadsheet for calculating solid fuel, liquid fuel and gaseous fuels power plant emission has been prepared and my be obtained thru the ENERGY DATA page of this website. It includes the following basic steps: A) SELECT TYPE OF FUEL Select type of fuel by entering 1 as received 1 0 Coal 0 Carbon 0 0 0 26.10 32.41 37.47 41.51 44.74 47.33 Hydrogen 1.80 2.24 2.58 2.86 3.09 3.26 Sulfur 0.65 0.81 0.94 1.04 1.12 1.19 NOX 0.00 0.00 0.00 0.00 0.00 0.00 CO 0.00 0.00 0.00 0.00 0.00 0.00 Oxygen 11.25 13.97 16.15 17.89 19.29 20.40 Nitrogen Water Vapor 0.23 0.29 0.33 0.37 0.39 0.42 54.75 43.80 35.04 28.03 22.42 17.94 Ash Total Btu / lb 5.22 6.48 7.49 8.30 8.95 9.47 100.00 4794 100.00 5966 100.00 6903 100.00 7653 100.00 8253 100.00 8733 B) CALCULATE THEORETICAL OXYGEN REQUIREMENT From % wt No. Substance Formula Table 2 - Calculation of Combustion Products and Theoretical Oxygen Requirements - Molar Basis (Babcock & Wilcox, p. 9-4 Moles Gross Net Molecular Analysis per Analysis HV HV Combustion Molecular Oxygen Theoritical Air 100 lb Weight % wt fuel % vol btu/lb btu/lb Products Weight Required O2 (m 1 Carbon C 12.0110 26.10 2.1730 33.51 14093 14093 CO2 44.0098 1.00 2 Hydrogen H2 2.0159 1.80 0.8929 13.77 61095 51625 H2O 18.0153 0.50 29 Sulfur S 32.0660 0.65 0.0204 0.31 NOX NO2 46.0055 0.00 0.0000 0.00 CO CO 28.0104 0.00 0.0000 0.00 3 Oxygen O2 31.9988 11.25 0.3516 4 Nitrogen N2 28.0134 0.23 0.0082 Water Vapor H20 18.0153 54.75 3.0389 46.86 32 Ash TOTAL 3980 3980 SO2 64.0648 4347 4347 NO2 46.0055 1.00 CO 28.0104 -0.50 5.42 O2 31.9988 -1.00 0.13 N2 (fuel) 28.0134 0.00 H2O 18.0153 0.00 N2 (air) 28.1610 5.22 15.4203 100.00 6.4850 GIVEN 100.00 4804 4634 4,000 4634 1.00 per lb fuel C) CALCULATE FLUE GAS ANALYSIS USING ASSUMED EXCESS AIR RATIO (OR CALCULATE FROM MEASURED EXCESS O2 - this requires stoichiometric calculations using fuel analysis, molecular weights and ambient air analysis) D) CONVERT PPMV TO MG PER Nm3 USING MOLECULAR WEIGHT AND STANDARD MOLAL VOLUME OF GAS AT STANDARD TEMPERATURE AND PRESSURE (1 ATM, 0 DEG CELSIUS) As Measured Flug Gas Emission vs DENR CO2 H2O SO2 (COMPUTED) NO2 (MEASURED) CO (MEASURED) O2 N2 (fuel) H2O N2 (air) wet gas dry gas 6 Reference DENR lb mole/ % vol % vol ppm V mg / Nm3 ppm V mg / Nm3 mg / Nm3 100 lb (WET) (DRY) (DRY) (DRY) (DRY) (DRY) (DRY) 2.173 0.893 12.81842 5.26721 16.68917 0.020 0.12034 0.15668 245 700 210 601 700 0.000 0.00000 0.00000 487 1,000 418 858 1000 400 500 343 429 500 0.458 0.008 3.039 10.361 16.952 13.020 2.69969 0.04843 17.92599 61.11992 100.00000 3.51491 0.06306 79.57619 100.00000 84.38% % Sulfur Removal ppmv = (% vol / 100) * 10^6 = % vol * 10^4 X, O2 ref = X * (21 - O2 ref) / (21 - O2 meas) where O2 ref = 6% ppmv X = (mg/Nm3 X) * 10^3 * (0.0224 m3/mol) / (MW X) mg/Nm3 X = (ppmv X) / 10^3 * (MW X) / (0.0224 m3/mol)