Chapter 1 - Solutions
advertisement

CHAPTER 6 Problems: 1, 6, 8, 16, 18, 30, 34, 41, 48, 51, 54, 58, 66, 68, 69, 76, 86, 91,
96, 108, 130
1) What is a wave? Using a diagram, define the following terms associated with waves:
wavelength, frequency, amplitude.
wavelength - The distance between successive peaks (or troughs) in a wave. MKS unit is
meters.
frequency - The number of wavelengths passing a fixed point per unit time. MKS unit is
s-1, but the term Hertz is also used for frequency (1 Hz = 1 cycle/s = 1 s-1).
amplitude - The height of a wave, which is related to the intensity.
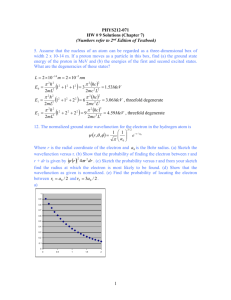
6)
a) What is the frequency of light having a wavelength of 456. Nm?
c = , and so = c/
So
= 2.998 x 108 m/s = 6.57 x 1014 s-1
456. x 10-9 m
b) What is the wavelength (in nm) of radiation having a frequency of 2.45 x 109
Hz? (This is the type of radiation used in microwave ovens.)
c = , and so = c/
So
= 2.998 x 108 m/s = 0.122 m = 1.22 x 108 nm
2.45 x 109 s-1
1
8) How many minutes would it take a radio wave to travel from the planet Venus to
Earth, when they are separated by their average distance of 28. million miles? How long
would it take an infrared wave to travel the same distance?
d (in km) = 28. x 106 mile 1.602 km = 44.9 x 106 km = 44.9 x 109 m
mile
t = d = 44.9 x 109 m = 149.6 s = 2.49 minutes
v
2.998 x 108 m/s
Since the speed of light is independent of wavelength, it would take the same time for an
infrared wave to travel the same distance.
16) The blue color of the sky results from the scattering of sunlight from molecules of air.
The blue light has a frequency of about 7.5 x 1014 Hz.
a) Calculate the wavelength (in nm) associated with this radiation.
c = , and so = c/
So
= 2.998 x 108 m/s = 4.00 x 10-7 m = 400. nm
7.5 x 1014 s-1
b) Calculate the energy (in Joule) of a single photon associated with this
frequency of light.
E = h = (6.626 x 10-34 Js)(7.5 x 1014 s-1) = 4.97 x 10-19 J
18) What is the wavelength (in nm) of radiation that has an energy of 2.13 x 10 3 kJ/mol?
In what region of the electromagnetic spectrum is this radiation found?
E(per photon) = 2.13 x 106 J
1 mol
= 3.54 x 10-18 J/photon
mol
6.022 x 1023 photon
E = hc/, and so = hc/E
So
= (6.626 x 10-34 Js)(2.998 x 108 m/s) = 5.61 x 10-8 m = 56.1 nm
3.54 x 10-18 J
This is in the UV region of the spectrum.
30) Calculate the wavelength (in nm) of a photon emitted by a hydrogen atom when its
electron drops from the n = 7 state to the n = 2 state.
(1/) = RH { (1/nf2) - (1/ni2) } = (0.01097 nm-1) { (1/22) - (1/72) } = 0.002509 nm-1
So
= 1/(0.002509 nm-1) = 399. nm
2
34) Consider the following energy levels of a hypothetical atom:
E4 = - 1.0 x 10-19 J
E3 = - 5.0 x 10-19 J
E2 = - 10. x 10-19 J
E1 = - 15. x 10-19 J
An energy level diagram is given below to help picture the processes taking place
a) What is the wavelength of the photon needed to excite an electron from E1 to
E4?
E = hc/ = E4 - E1 = ( - 1.0 x 10-19 J) - (- 15. x 10-19 J) = 14. x 10-19 J
= hc = (6.626 x 10-34 Js)(2.998 x 108 m/s) = 1.42 x 10-7 m = 142. nm
E
14. x 10-19 J
b) What is the energy (in Joule) a photon must have to excite an electron from E2
to E3?
E = E3 - E2 = (- 5.0 x 10-19 J) - (- 10. x 10-19 J) = 5 x 10-19 J
c) When an electron drops from the E3 level to the E1 level, the atom is said to
undergo emission of light. Calculate the wavelength (in nm) of the photon emitted in this
process.
E = hc/ = E3 - E1 = ( - 5.0 x 10-19 J) - (- 15. x 10-19 J) = 10. x 10-19 J
= hc = (6.626 x 10-34 Js)(2.998 x 108 m/s) = 1.99 x 10-7 m = 199. nm
E
10. x 10-19 J
41) Thermal neutrons are neutrons that move at speeds comparable to those of air
molecules at room temperature. These neutrons are most effective in initiating a nuclear
chain reaction in 235U. Calculate the wavelength (in nm) associated with a beam of
neutrons moving at 7.00 x 102 m/s. The mass of a neutron is 1.675 x 10-27 kg.
DB = h =
(6.626 x 10-34 Js)
= 5.65 x 10-10 m = 0.565 nm
-27
2
mv
(1.675 x 10 kg)(7.00 x 10 m/s)
3
48) How is the concept of electron density used to describe the position of an electron in
the quantum mechanical treatment of the atom?
According to quantum mechanics we cannot assign a specific location for an
electron in an atom (or ion or molecule). Instead we use the concept of electron denisty,
which represents the probability of finding the electron in a particular location in space.
51) The speed of a thermal neutron (see problem 6.41) is known to within 2.0 km/s.
What is the minimum uncertainty in the position of the thermal neutron?
(x)(p) h/4 , and so (x)
h
4(p)
p = m(v) = (1.675 x 10-27 kg)(2.0 x 103 m/s) = 3.35 x 10-24 kgm/s
(x)
(6.626 x 10-34 Js) = 1.57 x 10-11 m = 15.7 pm
4(3.35 x 10-24 kgm/s)
54) Describe the four quantum numbers used to characterize an electron in an atom.
The four quantum numbers (and their restrictions) are as follows:
n (principal quantum number). n = 1, 2, 3, … For the hydrogen atom this determines the
energy of the electron. In addition, the larger the value for n the greater the average
distance between the electron and the nucleus, and the larger the orbital.
(angular momentum quantum number). = 0, 1, 2, …, (n-1). Determines the shape of
the orbitals. For multielectron atoms the energy of the electron is determined by both n
and .
m (magnetic quantum number). m = 0, 1, 2, …, . Determines the orientation of
the orbital. Note that for a particular value of there are 2 + 1 distinct orbitals.
ms (spin quantum number). ms = 1/2. Determines the orientation of the electron spin,
either “spin up” (ms = 1/2) or “spin down” (ms = - 1/2).
58) An electron in an atom is in the n = 3 quantum level. List the possible values of and
m that it can have.
If n = 3, the possible values for and m are:
= 2; m = -2, -1, 0, 1, or 2
= 1; m = -1, 0, or 1
= 0; m = 0
4
66) Give the values for the four quantum numbers of an electron in the following orbitals:
a) 3s
n = 3, = 0, m = 0, ms = +1/2 or - 1/2
b) 4p
n = 4, = 1, m = -1, 0, or 1, ms = +1/2 or - 1/2
c) 3d
n = 3, = 2, m = -2, -1, 0, 1, or 2, ms = +1/2 or - 1/2
68) What is the difference between a 2px and a 2py orbital?
A 2px orbital is aligned with the x axis of the atom, and a 2py orbital is aligned
with the y axis of the atom.
69) Why do the 3s, 3p, and 3d orbitals have the same energy in a hydrogen atom but
different energies in a many-electron atom?
In a hydrogen atom there is only one electron, and so no interaction of electrons
with other electrons in the atom. In that case the energy of the electron only depends on
the value for n.
In a multielectron atom different electrons in different orbitals push each other
away, raising the energy. For a particular value for n, the smaller the value for the
lower the energy for the orbital.
76) State the Aufbau principle, and explain the role it plays in classifying the elements in
the periodic table.
The Aufbau principle applies to the ground (lowest energy) state of an atom. It
says that electrons will add to an atom by going into the lowest energy orbital that has
space to accommodate them. In adding to the atom the electrons must also obey the
Pauli principle.
86) Portions of the orbital filling diagrams representing ground state electron
configurations of certain elements are shown here. Which of them violate the Pauli
exclusion principle? Which violate Hund’s rule?
a) Violates the Pauli principle (two electrons have the same spin in the same
orbital.
b) Violates Hund’s rule. We could put three of the four electrons “spin up” and
get a larger value for total electron spin.
c) Okay as is.
5
d) Violates Hund’s rule. We could move the spin down electron into the empty
box, and make it spin up, and have all five electrons spin up.
e) Violates Hund’s rule. We could take the electron in the 4th box, which is spin
down, and make it spin up, and then have 5 of the 6 electrons spin up.
f) Violates the Pauli principle. Both electrons in the third box are spin down.
91) Explain why the ground-state electron configuration for Cr and Cu are different than
what we might expect.
Cr
Cu
predicted [Ar] 4s2 3d4
predicted [Ar] 4s2 3d9
actual [Ar] 4s1 3d5
actual [Ar] 4s1 3d10
In both cases the difference is because there is a small lowering of energy if a single
electron can be transferred from an s orbital to a d orbital to give a d orbital that is either
half-filled (5 electrons) or completely filled (10 electrons).
96) Write the ground state electron configurations for the following elements (write the
configurations using both the long and the shorthand method):
a) B 1s2 2s2 2p1 = [He] 2s2 2p1
b) V
1s2 2s2 2p6 3s2 3p6 4s2 3d3 = [Ar] 4s2 3d3
c) C
1s2 2s2 2p2 = [He] 2s2 2p2
d) As 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p3
= [Ar] 4s2 3d10 4p3
e) I
1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s2 4d10 5p5
= [Kr] 5s2 4d10 5p5
f) Au 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s2 4d10 5p5 6s1 4f14 5d10
= [Xe] 6s1 4f14 5d10
Notice that in Au an electron moves from 6s to 5d to fill the d orbital.
The advantage of the shorthand notation for atoms with a large number of
electrons is clear.
108) Ionization energy is the minimum energy required to remove an electron from an
atom. It is usually expressed in units of kJ/mol, that is, the energy in kilojoules required
to remove one mole of electrons from one mole of atoms.
6
a) Calculate the ionization energy for a hydrogen atom.
When an electron and proton are bound together in a hydrogen atom the energy of
the system is negative. A positive energy corresponds to a free electron that is no longer
bound to the proton. Therefore the energy required to remove one electron in the n = 1
state from one hydrogen atom is just the energy that must be added to an electron in the n
= 1 state to make its final energy greater than zero. The minimum energy to do this is
E = 0 - En=1 = - ( - 2.18 x 10-18 J) = 2.18 x 10-18 J
12
The ionization energy, EI, is the energy required to ionize one mole (Avogadro's number)
of hydrogen atoms with electrons initially in the n = 1 state.
EI = NA E = (6.022 x 1023 mol-1)(2.18 x 10-18 J) = 1.31 x 106 J/mol = 1310 kJ/mol
b) Repeat the calculation, assuming that the electrons are being removed from the
n = 2 state instead of from the ground (n = 1) state.
If the hydrogen atoms are initially in the n = 2 state, then for one atom
E = 0 - En=2 = - ( - 2.18 x 10-18 J) = 5.45 x 10-19 J
22
and to ionize one mole of atoms requires an energy
EI = NA E = (6.022 x 1023 mol-1)(5.45 x 10-19 J) = 3.28 x 105 J/mol = 328. kJ/mol
130) A ruby laser produces radiation of wavelength 633. nm in pulses whose duration is
1.00 x 10-9 s.
a) If the laser produces 0.376 J of energy per pulse, how many photons are
produced in each pulse?
The energy for one photon is
Ephoton = hc = (6.626 x 10-34 Js)(2.998 x 108 m/s) = 3.14 x 10-19 J
633. x 10-9 nm
The number of photons produced in one pulse is
1 photon
= 1.20 x 1018 photons per pulse
3.14 x 10-19 J
b) Calculate the power (in watts) delivered by the laser per pulse. (1 W = 1 J/s).
# photons = 0.376 J
power = energy = 0.376 J
= 3.76 x 108 J/s = 376. MJ/s
-9
time
1.0 x 10 s
7