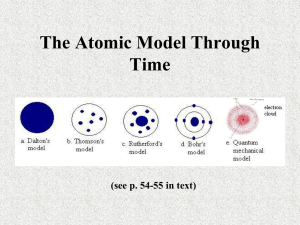
MISE - Physical Basis of Chemistry
advertisement

Redding Homework 5 - Page 1 MISE - Physical Basis of Chemistry Fifth Set of Problems - Due February 12, 2006 Submit electronically (digital drop box) by Sunday, February 12 – by 6 pm. Note: When submitting to digital Drop Box label your files with your name first and then the label - HmwkFive. Please put you name in the ‘Header’ along with the already-inserted page #. The following information may be useful when solving the problems: Ideal gas law: PV = nRT ; where R = 0.0821 L•atm•mole-1•K-1. 1 nm = 10-9 m ; 1 Ångstrom (Å) = 10-10 m ; 1 calorie (cal) = 4.184 joules (J) density of (liquid) water (H2O) = 1.00 g/mL ; 1000 mL = 1 L specific heat of (liquid) water (H2O) = 1.00 cal/gºC. q = C∆T = mc∆T = nCmolar∆T watt (W) = joule/s ; total energy delivered = watts • time (in s). Speed of light = 3.00 x 108 m/s = (wavelength)•(frequency) = •f Planck’s constant = 6.63 x 10-34 J•s ; Unit of frequency (f) = hertz (Hz) = 1/s Bohr radius constant = ao = 0.0529 nm = 5.29 x 10-11 meters. A H-like atom has one (1) electron & Z protons , Z is the atomic #. [For H, Z =1 and is electrically neutral ; other H-like atoms are ions with + charge.] Radius of electron orbit for H-like atom = n2•ao/Z ; n = 1, 2, 3, … [For H, Z = 1]. Rydberg energy constant = Ry = 2.18 x 10-18 joules (J). Energy of electron in H-like atom (E) = - Z2•Ry/n2 ; n = 1, 2, 3, … [For H, Z = 1]. Change in energy of electron in H-like atom (∆Eelectron): 1 1 ∆Eelectron = Efinal - Einitial = Z2•Ry• ; [For H, Z = 1]. 2 2 n n initial final Energy balance for energy change of electron in H-like atom emitting/absorbing light: ∆Eelectron = Ephtoton where Ephtoton = h•f = h•c/ total energy from light source = Nphtotons•Ephtoton 1 mole = 6.022 x 1023 “things” (atoms, molecules, photons) Redding Homework 5 - Page 2 1. The power output of a laser is measured by its wattage (W), the number of joules it radiates per second ( 1 W = 1 J/s). A 10.00 W laser produces a beam of green light with a wavelength of 520.0 nm. (a) Calculate the number of photons emitted in one minute by the laser. (520nm)(10-9m/1nm)= 5.20x10-7m Watts * seconds= (10.0 W) (60s) = Nphotons * (6.63x10-34)(3.0x108) 5.20x10-7m -19 600J= Nphotons * 3.825 x 10 Nphotons= 1.57x1021 (b) If the laser output is converted completely to heat and is used to heat 100.0 grams of water, initially at 25ºC, for a period of one minute, calculate the temperature change ∆T (in ºC) and the final temperature (in ºC) of the water. The idea is that the photons provide all of the energy to heat the sample. In other words, presume that heat transfer occurs in a insulated system - such that the sum of the total (heat) energy released by the photons plus the heat absorbed by the 100.0 grams of water equals zero (0). (600J)(1cal/4.184J) = 143 cal 143 cal = (100g)(1cal/g0C)(Tf-25oC) 143cal=100Tf -2500goC 2643.4=100Tf Tf= 26.4oC ΔT=1.4oC 2. Consider the hydrogen atom (Z = 1) when answering the following. [For this problem it may be useful to construct an energy level diagram like we did in class to “follow the electron” as it “jumps” between energy levels. You do not have to submit the diagram - only the reasoning and calculations used to answer any questions.] (a) Calculate the minimum (i.e., lowest) frequency (in Hz) that can be observed in the emission spectrum for the Lyman series. [Recall, the Lyman emission series results from transitions in which the electron in initially in any energy level in which n > 1 and the final energy level is n = 1.] ΔEelectron=(2.18x10-18J) (1/4 – 1/1) 6.63x10-34J*s f=(2.18x10-18J) (1/4 – 1/1) 6.63x10-34J*s f=(2.18x10-18J) (.25 – 1) 6.63x10-34J*s f=(2.18x10-18J) (-.75) 6.63x10-34J*s f=(-1.635x10-18J) f=-2.47x1015htz Redding Homework 5 - Page 3 (b) Calculate the maximum (i.e., longest) wavelength (in nm) that can be observed in the absorption spectrum for the Balmer series. [An absorption spectrum is one in which the electron absorbs a photon from an external white light source and undergoes a transition from a lower energy level to a higher energy level. Thus, the Balmer absorption series results from transitions in which the electron in initially in an energy level in which n = 2 and the final energy level could be any energy level in which n ≥ 3.] (6.63x10-34J*s)(3.0x108m/s)=(2.18x10-18J) (1/4 – 1/9) (6.63x10-34J*s)(3.0x108m/s)=(2.18x10-18J) (.13) (1.989x10-25J*m)=(2.834x10-19J) -25 (1.989x10 J*m)=(2.834x10-19J) (1.989x10-25J*m)= (2.834x10-19J) 7.02x10-7m= (7.02x10-7m)(1nm/10-9m)=701.8nm =701.8nm (c) Please refer to the pictures of the emission and absorption spectrum on page 7 of handout #4 - “The Emission of Light by Matter...the Revolution Begins...” Note how the Balmer absorption spectrum “looks” compared to the Balmer emission spectrum. The “lines” in the absorption spectrum appear as absences of color in an otherwise continuous background of colors - the exact “opposite” of the emission spectrum. Absorption line spectra are thus sometimes referred to as “dark line” spectra. The dark lines are exactly at the same locations as the colored lines in the emission spectrum. Given the description in (b) of how an absorption spectrum is set up, please explan why the hydrogen Balmer absorption spectrum “looks” the way it does - compared to the hydrogen Balmer emission spectrum. Base your explanation on the “Bohr model” of quantized energy levels, electron transitions, and their relationship to photons. The hydrogen Balmer absorption spectrum “looks” the way it does due to the electrons transitioning from level to level. Firstly, according to Bohr, there are restrictions placed on microscopic behavior of the electrons. The radii, or energy levels (n), may only be whole numbers. He hypothesized that the levels looked similar to planets rotating around the sun, thus it is sometimes referred to as the planetary model. The levels must be energetically stable; otherwise the electrons could spiral into the nucleus. The radius may be represented using the formula r=n2 (0.0529nm). The levels are represented by whole numbers such as 1, 2, 3, etc. Redding Homework 5 - Page 4 It must also be understood absorption and emission of light only occur when the electrons are transitioning. The energy flow is positive or negative depending upon the “direction” in which the electron is transitioning. For example, if the electron is in a higher level, due to electricity, it could then makes its way back to a lower energy level by transitioning from level four and ending at level two. That energy loss is negative and seen as light. Bohr represented this mathematically as follows: ΔE= Efinal – Einitial or more specifically ΔEelectron = (2.18x10-18J) (1/ni2 - 1/nf2). The energy changes are known as a photons and are equal to the energy change of the electron. As stated in part b, in the absorption spectrum, the “electron absorbs a photon from external white light source.” The electron is transitioning from a lower energy level to a higher energy level. Instead of emitting light, the light is now absorbed and is seen as dark lines. 3. For this question, imagine the hydrogen-like atom (ion) that can be created from elemental beryllium (Be) for which Z = 4. [For this problem it may be useful for some of the questions below to construct an energy level diagram like we did in class to “follow the electron” as it “jumps” between energy levels. You do not have to submit the diagram - only the reasoning and calculations used to answer any related questions.] (a) What must be the net charge (+1, +2, …) on the Be atom for it to become hydrogen-like? How do you know? The Be atom must lose 3 electrons to become hydrogen like because hydrogen only has 1 electron. If Be has 4 protons, it must have 4 electrons (assuming it is not an ion). In order for it to become like hydrogen, it would have to lose 3 electrons, giving it an overall charge of +3 because it would have 3 more protons than electrons. It would no longer be neutral. (b) Determine the wavelength (in nm) and the frequency (in Hz) of the first spectral line in the Balmer emission series. [The first spectral line, is the one of longest possible wavelength (i.e., least photon energy). Recall, that all Balmer photon emissions involve the electron undergoing an energy level transition in which the final n = 2 and the initial energy level can be any n ≥ 3. ] Redding Homework 5 - Page 5 1 1 h•c/=∆Eelectron = Efinal - Einitial = Z2•Ry• 2 2 n n initial final (6.63x10-34J*s)(3.0x108m/s)=16 (2.18x10-18J) (1/9 – 1/4) (6.63x10-34J*s)(3.0x108m/s)=16 (2.18x10-18J) (-.139) (1.989x10-25J*m)=(-4.848x10-8J) (1.989x10-25J*m)=(-4.848x10-8J) (1.989x10-25J*m)= (-4.848x10-8J) =-4.103x10-8m =-41.02nm This does not seem correct… why is it negative? Why is it so small? f=c/ f=3.0x108m/s -4.102x10-8m f=-7.31x1015htz This does not seem correct… why is it negative? 4. Quantum Theory at work…telling the time…Cesium (Cs) clocks… “…The most accurate realization of a unit that mankind has achieved…” A bit of Déjà Vu… The excerpts below are from the following URL: http://tycho.usno.navy.mil/cesium.html “A “cesium(-beam) atomic clock” (or “cesium-beam frequency standard”) is a device that uses as a reference the exact frequency of the microwave spectral line emitted by atoms of the metallic element cesium, in particular its isotope of atomic weight 133 (“Cs-133”). The integral of frequency is time, so this frequency, 9,192,631,770 hertz (Hz = cycles/second), provides the fundamental unit of time, which may thus be measured by cesium clocks. Today, cesium clocks measure frequency with an accuracy of from 2 to 3 parts in 10 to the 14th, i.e. 0.00000000000002 Hz; this corresponds to a time measurement accuracy of 2 nanoseconds per day or one second in 1,400,000 years. It is the most accurate realization of a unit that mankind has yet achieved. A cesium clock operates by exposing cesium atoms to microwaves until they vibrate at one of their resonant frequencies and then counting the corresponding cycles as a measure of time. The frequency involved is that of the energy absorbed from the incident photons when they excite the outermost electron in a cesium Redding Homework 5 - Page 6 atom to jump (“transition”) from a lower to a higher orbit. According to quantum theory, atoms can only exist in certain discrete (“quantized”) energy states depending on what orbits about their nuclei are occupied by their electrons. … Cesium is the best choice of atom for such a measurement because all of its 55 electrons but the outermost are confined to orbits in stable shells of electromagnetic force. Thus, the outermost electron is not disturbed much by the others. The cesium atoms are kept in a very good vacuum of about 10 trillionths of an atmosphere so that the cesium atoms are little affected by other particles. All this means that they radiate in a narrow spectral line whose wavelength or frequency can be accurately determined.” Further down the same webpage: “In a cesium clock like these, liquid cesium is heated to a gaseous state in an oven. A hole in the oven allows the atoms to escape at high speed….” (a) Determine the wavelength (in cm) of the spectral line corresponding to this listed frequency (9,192,631,770 Hz). Also, determine the energy (in J) of the photon that has this frequency. f=c/ 9,192,631,770 Hz=3.0x108m/s =.0326m=3.263cm Energy in Joules: H*f=6.63x10-34J*s)( 9,192,631,770 Hz) Ephoton=6.095x10-24J (b) This outermost electron in Cs that is mentioned above, is in an energy level that the Bohr model would designate as n = 6. However, in this situation we are dealing with an electrically neutral Cs atom, there are as many protons as electrons. Thus, this outermost electron does not “feel” the full nuclear charge attraction of 55 protons (Z = 55) - because of the repulsion of the other 54 electrons. To a decent level of approximation, this outermost electron “feels” a nuclear charge of only one proton, i.e., “Z = 1” - just like hydrogen itself! Determine the energy (in J) and the orbital radius (in nm) of this outmost electron - assuming Z = 1 according to the Bohr model. Radius of electron orbit for H-like atom = n2•ao/Z (36)(.0529nm)=1.9044nm 1 Energy: Eelectron= -Z2*Ry/n2 Redding Homework 5 - Page 7 Eelectron= -12*2.18x10-18J/36 Eelectron= 6.056x10-20J (c) The webpage discussion (in blue) reveals that the gaseous Cs atoms are kept at a pressure of 10 trillionths of an atmosphere (i.e., 10-11 atmospheres) . Assuming that these Cs atoms are in a 2.00 L chamber maintained at 700ºC, determine the number of Cs atoms present if they exhibit ideal gas behavior. n=PV/RT n=(1x10-11)(2.0L)/(0.0821L*atm*mol-1*K)(973K) n=2.5x10-13 This seems like a really small amount!?!?! (d) The webpage discussion (in violet) reveals that the Cs atoms are heated in an oven to become a gas and then allowed to escape through a hole in the oven…sounds like effusion! :). [Recall Homework Set #3 - Problem #2 and the kinetic theory lecture slides (especially #15).] Assume a particular oven of fixed volume - maintained at constant temperature - contains some gaseous “Cs-133 atoms” (atomic weight = 133 g/mole) that are contaminated with some gaseous He atoms (atomic weight = 4.0 g/mole). The contaminated mixture contains 80 mole % of Cs-133 and 20 mole % He. • Determine the ratio of moles He : moles Cs-133 a few moments after the gaseous mixture begins to effuse through the hole in the oven into an evacuated chamber. • Now, determine the mole percent of each gas in this effused mixture. √My = √133g.mol = 5.77moles Cs: 1 He Mx 4g/mol Mole % Cs=5.77moles Cs = 85.23% 5.77 + 1.0 Mole % He=1.0moles He = 14.77% 5.77 + 1.0 I am not completely confident with this answer either…I feel as if there should be less Cs because it is a larger atom…