Unit 6 Study Ans
advertisement

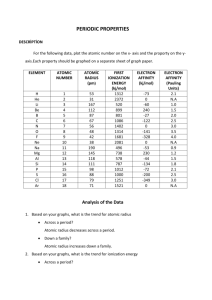
Study Guide: Periodic Table and Periodic Trends (Honors) 1. Differentiate between Mendeleev’s periodic table and Moseley’s periodic table. Mendeleev put the elements in order by atomic mass, but he noticed that in several places, the atomic masses did not always increase sequentially. Moseley discovered that ordering by atomic number was the correct way to describe the order. 2. State the periodic law. The properties of the elements are periodic functions of their atomic numbers. 3. Vertical columns on the periodic table are called groups or families and horizontal rows are called periods. 4. Elements with similar chemical properties are placed in the same group. 5. Complete the table below. Electron Config. [Xe] 6s24f145d106p6 [Xe] 6s1 [Ar] 4s13d5 [He] 2s2 [Kr] 5s1 4d10 [He] 2s2 2p5 Block Period p s d s d p 6 6 4 2 5 2 Group/Type Element 18 / Noble Gas 1 / Alkali Metal 6 / Trans. Metal 2 / Alk. Earth Metal 11 / Trans. Metal 17 / Halogen Radon Cesium Chromium Beryllium Silver Fluorine 6. Determine whether the following statements are describing alkali metals (A), alkaline earth metals (AE), transition metals (TM), halogens (H), noble gases (NG): a) most reactive non-metals H b) s2p6 valence configuration NG c) second most reactive metals AE d) s1 valence configuration A e) exceptions found to e- configurations TM f) soft metals A g) d orbitals being filled TM 7. Would you expect Li and S to have different chemical properties? Why? Yes; Li is an alkali metal with 1 valence e-, and S is a nonmetal with 6 valence e-. 8. Explain why a magnesium atom is smaller than: a sodium atom; a calcium atom. Mg is smaller than Na because the greater nuclear charge in Mg pulls the e- cloud in tighter (periodic trend). Mg is smaller than Ca because it has fewer energy levels (group trend). 9. Predict the size of the astatine atom compared to the sulfur atom. At is larger than S, because At is 3 periods down from S, but only 1 group over. The trends compete, but the the sizeable increase in energy levels overcomes the slight difference in nuclear charge. 10. Would you expect at Cl- ion to be larger or smaller than an Mg2+ ion. Explain. A Cl- ion is larger than a Mg2+ ion, because the chloride ion has 3 energy levels, matching the e- configuration of argon, while the magnesium ion has only 2 (matching the e- configuration of neon). 11. Explain why the sulfide ion (S2-) is larger than the chloride ion (Cl-). Both of these ions match the e- configuration of argon, but S2- is larger because the nuclear charge is less, resulting in less “pulling in” of the e- cloud. 12. Compare the ionization energy of sodium to that of potassium. Ionization energy decreases down a group, so the IE of sodium is greater than that of potassium. 13. Explain the difference in ionization energy between lithium and beryllium. Beryllium has a greater IE than lithium because Be’s greater nuclear charge holds the electrons tighter, resulting in increased IE required to remove an e-. 14. Will the electronegativity of barium be larger or smaller than that of strontium? smaller 15. Will a magnesium have a tendency to form a cation or an anion? cation (loses 2 e-) 16. Arrange oxygen, fluorine, and sulfur in order of increasing electronegativity. S, O, F 17. The most metallic element on the periodic table is francium; the least metallic element is fluorine. 18. As you go across a period, the electron affinity decreases (becomes a larger negative number) . Explain why. Nuclear charge increases across a period, leading to a stronger attraction for electrons. 19. When an electron is added to a neutral atom, energy is released (exo, negative). This is electron affinity. When an electron is removed from a neutral atom, energy is absorbed (endo, positive). This is ionization energy. A+ + e-, energy is a reactant and ionization 20. In an ionization energy equation, A energy is an endothermic process. A-, energy is a product and electron affinity 21. In an electron affinity equation, A + e- is an exothermic process. 22. Write an equation for the formation of F- from F. (energy = - 327.8 kJ/mol) Does this represent ionization energy or electron affinity? F (g) + e - F – (g) + 327.8 kJ electron affinity 23. For Group 3, list the element symbol for a) most metallic Ac b) least metallic Sc c) largest atomic radius Ac d) smallest atomic radius Sc e) largest ionic radius Ac f) largest ionization energy Sc g) largest electronegativity Sc h) smallest electron affinity Sc 24. For Period 3, list the element symbol for a) most metallic Na b) least metallic Cl c) largest atomic radius Na d) smallest atomic radius Ar e) largest ionic radius P f) largest IE Ar g) largest electronegativity Cl h) smallest electron affinity Cl