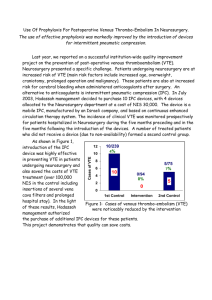
Venous thromboembolism (VTE) risk assessment tool
advertisement

NICS Venous Thromboembolism Risk Assessment Form Version 2 2008 1. Treating doctor or nurse to determine and document highest VTE risk category 2. Check for contraindications to VTE prophylaxis 3. Record drugs and orders for GCS/IPC as per hospital policy 4. Print name, sign and date on completion Surgical VTE Risk▲ Tick AFFIX PATIENT LABEL Recommended VTE prophylaxis▲ LMWH or Fondaparinux# AND IPC (with or without GCS) Hip arthroplasty Knee arthroplasty Major trauma Hip fracture surgery HIGH LMWH or LDUH or Fondaparinux# AND GCS (with or without IPC) Other surgery with prior VTE &/or active cancer LMWH or LDUH AND GCS (with or without IPC) Major surgery and age >40 yrs (Major surgery refers to intra-abdominal surgery and all other operations >45 mins) Other risk (please state): All other surgery LOWER Consider GCS All other surgery with additional VTE risk factors☼ (see over) Medical VTE Risk▲ (see over) Consider LMWH or LDUH & GCS Tick Ischaemic stroke History of VTE LMWH or LDUH Active cancer OR Decompensated heart failure HIGH Acute on chronic lung disease GCS &/or IPC If heparin contraindicated Acute inflammatory disease Age > 60 years Other risk (please state): LOW None of the above risk factors No prophylaxis recommended Are there any contraindications to chemical or mechanical prophylaxis? (indicate below) Chemical Tick Mechanical Tick Active bleeding Severe peripheral arterial disease High risk of bleeding e.g. Haemophilia, thrombocytopenia (platelet count <50 x 109/L), history of GI bleeding Severe peripheral neuropathy Severe hepatic disease (INR >1.3) Severe leg deformity Adverse reaction to heparin Recent skin graft On current anticoagulation Other (please state): Other (please state): No contraindications to mechanical prophylaxis No contraindications to chemical prophylaxis Patient risk status Prophylaxis required? → Document risk status as per hospital policy High Lower/ Low → Document risk status as per hospital policy Yes → All drugs and orders for GCS/IPC must be documented as per hospital policy No → Reason: Patient assessed by Name (PRINT): Position: Signature: Date: Key: # Fondaparinux for orthopaedic surgery only ▲ Based on Prevention of Venous Thromboembolism: Best Practice Guidelines for Australia and New Zealand 4th Edition, December 2007. Abbreviations: LMWH LDUH GCS IPC Low molecular weight heparin Low dose unfractionated heparin Graduated compression stockings Intermittent pneumatic compression Details of risk stratification and recommended prophylaxis Surgical Risk Stratification Recommended VTE Prophylaxis Duration HIGH LMWH(Enoxaparin 40mg/day OR Dalteparin 5000 units/day) OR Fondaparinux# 2.5mg/day AND IPC (with or without GCS) LMWH(Enoxaparin 40mg/day or Dalteparin 5000 units/day) OR Fondaparinux# 2.5mg/day OR LDUH 5000 units/TDS AND GCS (with or without IPC) LMWH (Enoxaparin 20mg/day OR Dalteparin 2500 units/day) OR LDUH 5000 units BD or TDS AND GCS (with or without IPC) Consider GCS If additional VTE risk factors☼ consider LMWH(Enoxaparin 20mg/day or Dalteparin 2500 units/day) or LDUH 5000 units BD or TDS 5-10 days 28-35 days for hip arthroplasty Hip arthroplasty Knee arthroplasty Major trauma Hip fracture surgery Other surgery with prior VTE &/or active cancer LOWER Major surgery & age >40 years (Major surgery refers to intraabdominal surgery and all other operations >45 mins) All other surgery Medical Risk Stratification HIGH Ischaemic stroke History of VTE Active cancer Decompensated heart failure Acute on chronic lung disease Acute inflammatory disease Age >60 years Other risk LOW None of the above 5-10 days 28-35 days for hip fracture surgery 5-10 days Until hospital discharge LMWH(Enoxaparin 40mg/day OR Dalteparin 5000 units/day) OR LDUH 5000 units BD or TDS OR GCS (with or without IPC) if heparin contraindicated Until resolution of acute medical illness or hospital discharge No prophylaxis recommended Notes for Medical Risk Stratification Please note: This is a guide only. If unsure please consult the treating doctor. Ischaemic stroke: acute, confirmed by CT or MRI and unable to walk unassisted because of motor impairment with a score of 2 or more (NIHSS) for motor function of the leg. History of VTE: Previous PE or DVT. Active cancer: prophylaxis is recommended for hospitalised or bed-ridden patients and ambulatory patients receiving thalidomide or lenalidomide with chemotherapy or dexamethasone. Decompensated heart failure: symptoms of heart failure that occur with minimal activity or at rest (NYHA Class III or IV). Acute on chronic lung disease: respiratory failure or exacerbation of respiratory disease with or without ventilation. Acute inflammatory disease: eg rheumatoid arthritis, systemic lupus erythematosus. ☼ Additional Risk Factors Immobility: patients with permanent immobility or significant period of past immobilisation Thrombophilia: evidence of a disorder of the haemostatic system which increases risk of thrombosis eg: antithrombin 3, protein C, or protein S deficiencies. Oestrogen therapy Pregnancy: current or puerperium. Active inflammation Strong family history of VTE and/or obesity References 1. Nicolaides AN, et al. Prevention and treatment of venous thromboembolism. International Consensus Statement (Guidelines according to scientific evidence).International Angiology. 2006; 25(2):101-61. 2. Sherman DG, et al. PREVAIL Study, Lancet. 2007; 369:1347-55. 3. Lyman G, et al. American Society of Clinical Oncology guideline: Recommendations for venous thromboembolism prophylaxis and treatment in patients with cancer. Journal of Clinical Oncology. 2007; 25(34):5490-505. NOTE: All hospitals should have a venous thromboembolism (VTE) prophylaxis policy for assessment of VTE risk in admitted patients. The policy should include information about appropriate prophylactic measures to minimise the risk of deep vein thrombosis and pulmonary embolism in every admitted patient based on best practice recommendations. This sample risk assessment form was developed by the National Institute of Clinical Studies in collaboration with the Private Hospital VTE Prevention Program Advisory Committee. The recommendations used in this form are based on Prevention of Venous Thromboembolism: Best Practice Guidelines for Australia and New Zealand, 4th Edition, December 2007, by the ANZ Working Party on the Management and Prevention of Venous Thromboembolism. This work was developed by NICS/NHMRC as part of the Private Hospital VTE Prevention Program. It may be reproduced in whole or part subject to inclusion of an acknowledgement of the source. It may not be reproduced for commercial use or sale.