Disseminated intravascular coagulation
advertisement

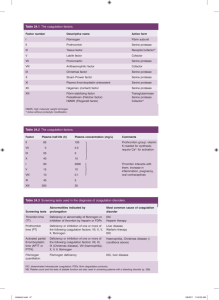
Disseminated intravascular coagulation Disseminated intravascular coagulation (DIC) is the term most commonly used for a clinicopathologic syndrome in which widespread intravascular coagulation is induced by procoagulants that are introduced or produced in the blood circulation and overcome the natural anticoagulant mechanisms. DIC may cause tissue ischemia from occlusive microthrombi and bleeding from both the consumption of platelets and coagulation factors and the anticoagulant effects of products of compensatory or secondary fibrinolysis. [Etiology] Numerous disorders can provoke DIC: ① Infections (Viral, bacterial, rickettsial, mycotic, protozoal); ② Malignant diseases; ③ Obstetric complications (Abruptio placentae, septic abortion and chorioamnionitis, amniotic fluid embolism, intrauterine fetal death, miscellaneous); ④ Surgery and trauma ; ⑤ Miscellaneous diseases. [Pathogenesis] The mechanisms that activate or “trigger” DIC act on processes that are involved in normal hemostasis; namely, the processes of platelet adhesion and aggregation and the contact-activated (intrinsic) and tissue-activated (extrinsic) pathways of coagulation. 1. Activation of the extrinsic coagulation system by tissue factor expressed on cell surfaces: trauma, cancer, obstetric events, ascites (shunt) etc; 2. Activation of intrinsic coagulation system by causing injury to endothelial cells: hypotension, endotoxin, heat stroke, vasculitis, aneurysm, hemangioma etc; 3. Activation of coagulation factors, e.g., factor Ⅹ by cancers, factor Ⅱ by snake venoms; All of these lead to thrombin generation that in the presence of failure of the control mechanisms results in intravascular coagulation. This in turn, can lead to thrombosis and consumption of platelets, fibrinogen, and other coagulation factors. Bleeding can be caused by depletion of these essential hemostatic components; by the anticoagulant effects of fibrinogen/fibrin degradation products; and by further depletion of fibrinogen, factor Ⅴ, and factor Ⅷ by plasmin if generated in excess by the secondary fibrinolysis or if uninhibited due to diminished antiplasmin levels. [Clinical features] The manifestations of DIC depend on the magnitude and rate of exposure to the DIC trigger. For example, the dramatic cases of “acute” DIC, characterized by severe bleeding due to excessive consumption of hemostatic components, may develop when blood is exposed to large amounts of tissue factor over a brief period of time. Alternatively, “chronic” DIC develops when blood is continuously or intermittently exposed to small amounts of tissue factor. Under these circumstances, clinical signs may be minimal or altogether absent, and most coagulation tests will be only slightly impaired. 1. Bleeding Acute DIC is frequently heralded by hemorrhage into the skin at multiple sites. Petechiae, ecchymosis, and oozing from veni-punctures, arterial lines, catheters, and injured tissues are common. Bleeding may also occur on mucosal surfaces. Hemorrhage may be life-threatening, with massive bleeding into the gastrointestinal, lungs, central nervous system, or orbit. Patients with chronic DIC usually exhibit minor skin and mucosal bleeding. 2. Thromboembolism Extensive organ dysfunction can result from microvascular thrombi or from venous and/or arterial thromboembolism. For example, involvement of the skin can cause hemorrhage bullae, acral necrosis, and gangrene; involvement of the lungs can cause acute respiratory distress syndrome, hypoxemia, edema, hemorrhage; involvement of liver can cause jaundice, parenchymal damage; involvement of gastrointestinal can cause bleeding, mucosal necrosis, and ulceration; involvement of central nervous system can cause stupor, coma, convulsions, focal lesions and intracranial bleeding. 3. Circulatory disturbance, shock Both the diseases underlying DIC and DIC itself can cause shock. For example, septicemia or excessive blood loss to trauma or to obstetric complications can by themselves cause shock. 4. Microangiopathic hemolytic anemia Erythrocytes are injured mechanically during passage through fibrin networks in the microcirculation. Such microangiopathic hemolysis leads to the production or schistocytes and microspherocytes. [Laboratory features] 1. Basic blood examinations (1) Platelet count: thrombocytopenia is an early and consistent sign of DIC. (2) Peripheral blood smear: Examination of the blood smear reveals schistocytes in approximately 50% of cases. 2. The coagulation defect (1) Partial thromboplastin time (PTT), Prothrombin time, and thrombin time: The PTT, PT, TT are prolonged in most patients with acute DIC. Early in the course of the disorder, and in chronic DIC, the PTT may be normal, or even shorter than normal, which may be the result of the procoagulant effects of activated coagulation factors. (2) Fibrinogen concentration: The plasma levels of fibrinogen usually are significantly depressed. 3. Tests for fibrinolysis (1) Fibrinogen degradation products (FDP): There is usually an increased level of FDP. (2) D-dimer: Assay of plasma D-dimer is very useful for evaluation of patients with DIC. Increased levels indicate that cross-linked fibrin generated by thrombin has been digested by plasmin. (3) Plasma protamine paracoagulation test (3P) (4) Euglobulin lysis time 4. Other laboratory findings (1) Fibrinopeptide A (FPA): FPA provide direct evidence of the action of thrombin on fibrinogen, it is exceptionally sensitive indicators of DIC and may be normal even in patients with normal levels of FDP. (2) Antithrombin Ⅲ: Levels of antithrombin Ⅲ may be diminished in some cases. (3) Factor Ⅷc levels (4) Proteins C and S levels. Laboratory data change with remarkable rapidity in DIC, and in doubtful cases, it is often important to repeat the tests at frequent intervals, even every 8 to 12 hours and observing the dynamics of the process. [Differential diagnosis] 1. DIC and primary fibrinogenolysis. 2. DIC and severe liver disease. [Treatment] 1. Management of underlying disorders The survival of patients with DIC depends on vigorous treatment of the underlying disorders and curtailing the triggers of blood coagulation. For example, intensive antibiotic treatment in patients with gram-negative bacteremia, hysterectomy in patients with abruptio placenta, resection of an aortic aneurysm, and debridement of crushed tissues are the most essential steps in the management of such patients. 2. Heparin administration Heparin is a specific activator of the physiologic antithrombin Ⅲ system and thereby inhibits a number of proteolytic enzymes, including factors Ⅸa,Ⅹa and thrombin. Indications: Administration of heparin is beneficial in some categories of acute and chronic DIC, including purpura fulminant, acute promyelocytic leukemia, dead fetus syndrome (at time of removal), and aortic aneurysm (prior to resection). Heparin is also indicated for treating thromboembolic complications in large vessels and before surgery in patients with metastatic carcinoma. Heparin administration may also be helpful when intensive blood component replacement fails to improve excessive bleeding and fails to increase the level of hemostatic factors. Dosage: The optimal dosage of heparin, however, is the source of some disagreement. Good therapeutic responses have been documented even with minimal dose regimens that do not significantly prolong the coagulation time, e.g., 5000 to 10000 U subcutaneously every 12 to 24 hours or 5 to 10 u/kg/hr by continuous drip. Laboratory control of heparin therapy for DIC is difficult. A rough estimate of the heparin level can be obtained from the coagulation time. 3. Antiplatelet drugs Dipyridamole 200-400mg/d intravenous drip; Aspirin 0.9-1.2g/d; Low molecular weight dextran 500-1500ml/d. Indications: (1) in hypercoagulability state; (2) the diagnosis of DIC is still not certain; (3) in mild cases. 4. Haemostatic support In DIC the concentration of both procoagulant factors and their control protein may be reduced. It is conventional offer haemostatic replacement therapy after massive blood transfusion with: (1) platelet concentrates, 1 donor unit/10kg body weight when the platelet count is below 50×109/L; (2) fresh frozen plasma (FFP) 10-15ml/kg body weight when the PTR is greater than 1.5; (3) additional cryoprecipitate when the fibrinogen concentration is 0.8g/l or less. 5. Fibrinolytic inhibitors Patients with early stage of DIC should not be treated by antifibrinolytic agents like ε-aminocaproic acid and tranecamic acid. These drugs block the secondary compensatory fibrinolysis that accompanies DIC and partially preserves tissue perfusion. In these conditions the use of fibrinolytic inhibitors can be considered, provided that: (1) the patient is bleeding profusely and has not responded to replacement therapy; (2)excessive fibrinolysis is observed, i.e., a very short euglobulin lysis time. In such circumstances the use of antifibrinolytic agents should be preceded by replacement of depleted blood components and continuous heparin infusion.