PowerPoint 簡報
advertisement

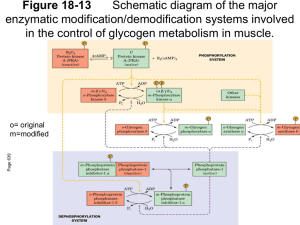
Coordinated regulation of glycolysis/gluconeogenesis Regulation of glycolysis Hexokinase Phosphofructokinase-1 Pyruvate kinase Hexokinase • There are four isozymes (I, II, III and IV) of hexokinase encoded by four different genes. • Hexokinase I and II are allosterically inhibited by their product, glucose 6phosphate. Hexokinase IV is not inhibited by G-6-P. Hexokinase • Hexokinase I and II are the predominant forms existing in muscle. Hexokinase IV is the predominant form in liver. • Hexokinase I and II will be half-saturated at about 0.1mM, but hexokinase IV will not be half-saturated until 10mM. Hexokinase • Hexokinase has different functions in liver and muscle. • Muscle consumes glucose, using it for energy production. • Liver maintains blood glucose homeostasis by removing or producing glucose. Muscle hexokinase • Because blood glucose concentration is about 4 to 5 mM, hexokinase in the muscle (which will be half saturated at 0.1mM) is always working at or near its maximal rate. Liver hexokinase • However, liver hexokinase (halfsaturated at 10mM) will not ever reach its maximal rate even after meal. Liver hexokinase • Liver hexokinase (glucokinase) is also regulated by a regulatory protein. When glucokinase is bound by it (which is enhanced by F-6-P), it become nuclear-localized and inactive. Liver hexokinase • After meal, glucose enter hepatocytes by GLUT2 transporter and is converted to G-6-P. G-6-P competes with F-6-P for glucokinase, which relieves its inhibition by regulatory protein. Phosphofructokinase-1 • PFK-1 catalyze the committing step of glycolysis. • This enzyme is regulated by ATP, AMP, ADP, citrate and fructose 2,6bisphosphate. ATP regulate the affinity of PFK-1 towards its substrate F-6-P • Not only as a substrate, ATP is also one of the end product of the glycolytic pathway. • ATP inhibit PFK-1 by binding to an allosteric site and lowering the affinity of the enzyme for F-6-P. Other molecules regulate PFK-1 • ADP and AMP relieve the inhibition by ATP. • Citrate increases the inhibitory effect of ATP. • F-2,6-BP is the strongest activator of PFK-1. Pyruvate kinase • Pyruvate kinase has at least three isozymes and one of them is liver-specific. • The liver pyruvate kinase is being regulated differently than other tissue type. Regulation of pyruvate kinase cAMP dependent Regulation of gluconeogenesis Pyruvate carboxylase FBPase-1 Pyruvate carboxylase • Pyruvate carboxylase is being positively regulated by acetylCoA. • The accumulation of acetylCoA signals that cell’s energy demands are met. • Acetyl-CoA also indirectly inhibit pyruvate dehydrogenase complex. How acetyl-CoA regulate PDC E2 • Acetyl-CoA indirectly inhibit PDC by stimulating a protein kinase that inactivates the dehydrogenase. FBPase-1 is inhibited by AMP F-2,6-BP: a potent regulator F-2,6-BP reciprocally regulate PFK-1 and FBPase-1 F-2,6-BP activates PFK-1 F-2,6-BP inhibit FBPase-1 The synthesis and breakdown of F-2,6-BP • F-2,6-BP is synthesized by PFK-2 and brokedown by FBPase-2, which is a single, bifunctional protein. When it is phosphorylated, it is FBPase-2. The bifunctional protein PFK-2/FBPase-2 Insulin and glucagon levels affect the balance between PFK-2/FBPase-2 A PP2A activated by xylulose 5phosphate also activate FBPase-2 PP2A Regulation of glycogen metabolism Glycogen phosphorylase Glycogen synthase Muscle glycogen phosphorylase • Muscle glycogen phosphorylase has two forms: the active a form and the less active b form. The active form is phosphorylated. Muscle glycogen phosphorylase • Glucagon and epinephrine stimulate the kinase that phosphorylate phosphorylase b, therefore active the whole glycogen breakdown process. How glucagon/epinephrine activate phosphorylase b kinase • When epinephrine/glucagon is secreted, it started the whole enzyme cascade by activate a GTP-binding protein. • Enzyme cascade allows for large amplification of the initial signal. Muscle glycogen phosphorylase • At resting stage, PP1 (phosphorylase a phosphatase) will dephosphorylate phosphorylase a, which will make it returning to the less active form (phosphorylase b). Liver glycogen phosphorylase • The dephosphorylated form (b) of liver glycogen phosphorylase is essentially inactive. Phosphorylation activates it, but when blood glucose is high, glucose will bind to the inhibitory allosteric site, induces a conformational change that will expose its phosphorylated Ser for PP1 to dephosphorylate (inactivate) this enzyme. Glycogen synthase • The activate form of glycogen synthase is not phosphorylated. • To inactivate glycogen synthase, it must be phosphorylated by casein kinase II (CKII) first, then glycogen synthase kinase 3 (GSK3) will add phosphoryl groups to three Ser residues near the carboxyl terminus of this protein. GSK3 inactivate glycogen synthase by phosphorylation Glycogen synthase • The activation of glycogen synthase requires PP1. • Glucose 6-phosphate will bind to the allosteric site of glycogen synthase b, making the enzyme a better substrate for PP1. GSK3 can be inactivated by phosphorylation • Insulin triggers activation of a protein kinase B to phosphorylate GSK3 at a Ser residue near the amino terminus, converting that region of the protein to a pseudosubstrate, preventing GSK3 from binding the real substrate (glycogen synthase). Insulin enhance glycogen synthesis by inhibiting the kinase that inactivate glycogen synthase Phosphoprotein phosphatase 1 (PP1) • PP1 can remove phosphoryl group from phosphorylase kinase, glycogen phosphorylase (inactivation), and glycogen synthase (activation) in response to glucagon/epinephrine. • By activating PP1 and inactivating GSK3, insulin stimulates glycogen synthesis. PP1 binds to glycogen-targeting protein (GM) and also other proteins Muscle has a different glucose transporter • GLUT2 is not present in myocyte. Instead, GLUT4 is present in myocyte and its expression is regulated by insulin. Insulin regulated the externalization/internalization of GLUT4