b-Lactamase Inhibitors
advertisement

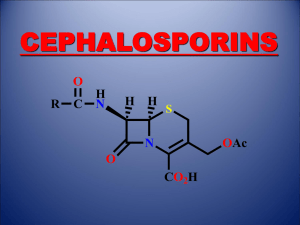
Patrick An Introduction to Medicinal Chemistry 3/e Chapter 16 ANTIBACTERIAL AGENTS Part 3: Other lactams ©1 Newer b-Lactam Antibiotics Thienamycin (Merck 1976)(from Streptomyces cattleya) Acylamino side chain absent OH Plays a role in ß-lactamase resistance Opposite stereochemistry to penicillins Carbon H H H3C NH3 S N O CO2 Double bond leading to high ring strain and an increase in b-lactam ring reactivity Carbapenam nucleus • • • • • Potent and wide range of activity vs Gram +ve and Gram -ve bacteria Active vs. Pseudomonas aeruginosa Low toxicity High resistance to b-lactamases Poor stability in solution (ten times less stable than Pen G) ©1 Newer b-Lactam Antibiotics Thienamycin analogues used in the clinic H OH Me O H OH Me NH HN H N CO2 H H O O C N Me Meropenem S N Me H N Me O H OH Imipenem S CO2 H H N Me O C N H Ertapenem(2002) S N CO2 CO2 ©1 Newer b-Lactam Antibiotics Nocardicins (Fujisawa 1975) HO2C D HC H2N O CH2 N OH C H N C CH2 H OH O Nocardicin A N C O H • • • • • • • CO2H Monocyclic b-lactam ring - monobactams Moderately active in vitro vs narrow group of Gram -ve bacteria Active vs. Pseusomonas aeruginosa Inactive vs. Gram +ve bacteria Different spectrum of activity from penicillins Thought to operate by a different mechanism from penicillins Low toxicity ©1 Newer b-Lactam Antibiotics Clinically useful monobactam Me N H2N Me O H N N S CO2H O O • • • • Me Aztreonam N SO3- Administered by intravenous injection Can be used for patients with allergies to penicillins and cephalosporins No activity vs. Gram +ve or anaerobic bacteria Active vs. Gram -ve aerobic bacteria ©1 b-Lactamase Inhibitors Clavulanic acid (Beechams 1976)(from Streptomyces clavuligerus) Sulphur replaced by O No acylamino side chain H 9 O 5 6 7 N 1 OH 4 3 2 H O H CO2H b-Lactam Oxazolidine ring • • • • • • Weak, unimportant antibacterial activity Powerful irreversible inhibitor of b-lactamases - suicide substrate Used as a sentry drug for ampicillin Augmentin = ampicillin + clavulanic acid Allows less ampicillin per dose and an increased activity spectrum Timentin = ticarcillin + clavulanic acid ©1 b-Lactamase Inhibitors Clavulanic acid - mechanism of action 1 2 NH 2 O NH 2 O CH2OH O OHH HN O N O CH2OH H Base CO2H CO2H 3 4 O 5 CH2OH H2N NH NH O HN O O H CH CH2OH HC O CO2H NH CO2H O O O H Irreversibly blocked ©1 b-Lactamase Inhibitors Penicillanic acid sulfone derivatives O S O 6 5 7 N O O • • • 6 2 3 Me CO2 Na Sulbactam • • S Me 1 O N Me N 3 N O N CO2 Tazobactam Suicide substrates for b-lactamase enzymes Sulbactam has a broader spectrum of activity vs b-lactamases than clavulanic acid, but is less potent Unasyn = ampicillin + sulbactam Tazobactam has a broader spectrum of activity vs b-lactamases than clavulanic acid, and has similar potency Tazocin or Zosyn = piperacillin + tazobactam ©1