Chapter 20 Review
advertisement

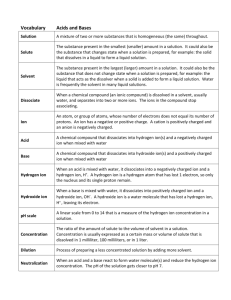
Chapter 20 – Review “Acids and Bases” Milbank High School 1 Chapter 20 - Review When an acid reacts with a base, what compound(s) are formed? What is the name of H2SO3? What is the formula for phosphoric acid? Give some properties of acids. Give some properties of bases. 2 Chapter 20 - Review If the hydrogen ion concentration is 10-10 M, is the solution acidic, alkaline, or neutral? If the [H1+] in a solution is 1 x 10-1 mol/L, what is the [OH1-]? 3 Chapter 20 - Review If the hydrogen ion concentration is 10-7 M, what is the pH of the solution? If the hydroxide ion concentration is 10-10 M, what is the pH of the solution? If the pH is 9, what is the concentration of the hydroxide ion? 4 Chapter 20 - Review For a solution to be classified as acidic, the hydrogen-ion concentration must be _____ than the hydroxide-ion concentration. If the pH is 6, what is the concentration of the hydrogen ion? 5 Chapter 20 - Review A solution in which the hydroxide ion concentration is 1 x 10-4 M is ______. A solution in which the hydroxide ion concentration is 1 x 10-8 M is ______. The formula of the hydrogen ion is often written as ______. 6 Chapter 20 - Review The products of self-ionization of water are _____. is most basic: a) [OH1-] = 1 x 10-4 M, or b) [H1+] = 1 x 10-11 M? In a neutral solution, the [H1+] is ______. What is pH? Which 7 Chapter 20 - Review In the reaction of water with hydrogen ions, which compound is the Lewis base? What is an acid according to Arrhenius? What type of acid (mono-, di-, or triprotic) is sulfuric acid? 8 Chapter 20 - Review Which is an Arrhenius base: a) LiOH, or b) CH3COOH? What is an acid, according to Bronsted? Which of the following is a Bronsted-Lowry base, but not an Arrhenius base: a) sodium hydroxide, or b) ammonia? 9 Chapter 20 - Review Which compound can act as both a Bronsted-Lowry acid and a Bronsted-Lowry base: a) water, or b) sodium chloride? Which type of solution is one with a pH of 8? 10 Chapter 20 - Review What is transferred between a conjugate acid-base pair? What is the term used to describe a substance that can be both an acid and a base? the reaction: NH41+ + H2O NH3 + H3O1+, water is acting as a(n) ______. In 11 Chapter 20 - Review A Lewis acid is a substance that can _____. What is the acid dissociation constant of a weak acid, if a concentration of 0.3 M gives a hydrogen ion concentration of 0.001 M? 12 Chapter 20 - Review What is the hydrogen ion concentration, if the acid dissociation constant is 1 x 10-6 M and the acid concentration is 0.01 M? What characterizes a “strong” acid or base? 13 Chapter 20 - Review A 0.12 M solution of an acid that ionizes only slightly in solution would be termed ___ and ___. A 12.0 M solution of an acid that ionizes completely in solution would be termed ___ and ____. 14 Chapter 21 – Review “Neutralization” Milbank High School 15 Chapter 21 - Review What products result from a neutralization reaction? What is obtained when phosphoric acid and magnesium hydroxide are mixed? What are the products of the reaction of one mole of Mg(OH)2 and one mole of H2SO4? 16 Chapter 21 - Review What is the gram-equivalent mass of magnesium hydroxide if magnesium hydroxide has a gram-molecular mass of 58.0 g? What is the purpose of a titration? 17 Chapter 21 - Review How many equivalents per mole are there in nitric acid? What is the gram-equivalent mass of chromic acid, H2CrO4? 18