Properties of Aqueous Solutions
advertisement

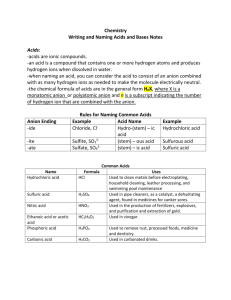
AP CHEMISTRY STUDENT NOTES – CH 3 & 4 REACTIONS AND STOICHIOMETRY 3.1 Chemical Equations the reactants are written to the left and the products are written to the right of the arrow s, g, l coefficients are used to show the relative amounts the law of conservation of matter balanced equations vs. skeleton equations relationship between amounts is stoichiometry 3.2 Balancing Formulas for reactants and products have to be written correctly Balancing Combustion Reactions Step 1: write correct formulas for reactants and products Step 2: Step 3: Step 4: Step 5: Combustion Complete oxidation of an organic compound to yield CO2 and H2O C2H4(g) + 3O2(g) 2CO2(g) + 2H2O(l) Nonmetallic hydrides combine with oxygen to form oxides and water SiH4(g) + 2O2(g) SiO2(s) + 2H2O(l) Nonmetallic sulfides combine with oxygen to form sulfur dioxide and oxides CS2(s) + 3O2(g) CO2(g) + 2SO2(g) 3.3 Chemical Equilibria Reactions are reversible, and many reactions lead to incomplete conversion of reactants to products. Chemical equilibrium Dynamic equilibrium Depending upon different factors, reactions can be product-favored or reactant-favored. 3.4 Properties of Aqueous Solutions Another name for a solution is a homogeneous mixture. Substance dissolved in water are called aqueous solutions. Polarity of water creates an attraction for ions. Electrolytes Compounds that conduct electricity in aqueous solution are called electrolytes. Strong electrolytes completely ionize in solution. Weak electrolytes only partially dissociate. Nonelectrolytes dissolve in water but don’t ionize. 3.6 Solubility Solubility rules on page 126 precipitation reactions form insoluble products lead nitrate and potassium iodide net ionic equations leave out the spectators Net Ionic Equations All AP equations “work”. The equations need to be written in net ionic form. All spectators must be left out and all ions written with charges. All molecular substances and insoluble compounds must be written together (not ionized). Weak electrolytes are not ionized, solids and pure liquids are written together. First classify the reaction as to type. If there is an acidic or basic solution, then it is redox. All solutions do not fit neatly into the five types you’ve already learned. Save all the reactions that you do and practice them before the AP test in May. Practice Problems Write a balanced net ionic equation for each of the following reactions: AlCl3 + Na3PO4 AlPO4 + NaCl Solutions of iron (III) chloride and potassium hydroxide give iron (III) hydroxide and potassium chloride when combined. Solutions of lead (II) nitrate and potassium chloride give lead (II) chloride and potassium nitrate when combined. 3.7 Acid and Base Reactions An acid is a substance that, when dissolved in water, increases the concentration of hydrogen ions, H+ (aq), in the water. A base is a substance that, when dissolved in water increases the concentration of hydroxide ion, OH-, in the water. ammonia produces hydroxide in water, forming ammonium and hydroxide ion common acids and bases on page 132 Strong Acids Strong Bases Group 1 Hydroxides Group 2 Hydroxides acids react with strong bases to produce a salt and water (ex. hydrochloric acid and sodium hydroxide) net ionic for strong base and strong acid: neutralization reaction Acids and Bases: The Bronsted-Lowry Definition an acid is a proton donor a base is a proton acceptor Acid-base reactions involve the transfer of a proton from an acid to a base to form a new acid and a new base. It is written as an equilibrium reaction and the equilibrium favors the weaker acid and base. Acids capable of transferring two or more hydrogen ions react with water in multiple steps. Different steps may favor different sides of the equilibrium. Some substances are amphiprotic and can function as either an acid or a base. Practice Problem Write the balanced, overall equation and the net ionic equation for the reaction of magnesium hydroxide with hydrochloric acid. Oxides of Nonmetals and Metals Nonmetal oxide + water acid o Carbon dioxide reacts with water to form carbonic acid, which then ionizes slightly to form hydrogen ion and bicarbonate ion. o Bicarbonate ion can further ionize to form carbonate. CO2 + H2O H2CO3 H2CO3 H+ + HCO3HCO3- H+ + CO32 Metal oxide + water base CaO(s) + H2O(l) Ca(OH)2(s) metal oxide + nonmetal oxide salt 6CaO(s) + P4O10(s) 2Ca3(PO4)2(s) 3.8 Gas-Forming Reactions Formation of a gas: gases may form directly in a double replacement reaction or can form from the decomposition of a product such as carbonic acid or sulfurous acid (table on page 165) Ex. Excess hydrochloric acid solution is added to a solution of potassium sulfite. H+ + SO3-2 H2O(l) + SO2(g) CuCO3(s) + H2SO4(aq) Ba(OH)2(s) + HNO3(aq) CuCl2(aq) + (NH4)2S(aq) 3.9 Oxidation-Reduction Reactions These reactions involve the transfer of electrons. The oxidation numbers of at least two elements must change. These may be single replacement, combination, and decomposition reactions. When a problem mentions an acidic or basic solution, it is probably redox. A substance that loses electrons is oxidized and is the reducing agent. A substance that gains electrons is reduced and is the oxidizing agent. LEO goes GER If one substance is oxidized, another substance must be reduced. Hence, redox reactions! 2Ag+ + Cu 2Ag + Cu+2 Oxygen may not be present; but a transfer of electrons results in a charge reduction. Silver ion accepts electrons from copper and is reduced to silver (silver ion is the oxidizing agent). Copper donates electrons to silver ion and is oxidized to copper (II) ion and is the reducing agent. Assigning Oxidation States The oxidation state of… an atom in an element is zero monatomic ion is same as charge oxygen is usually -2 except for peroxides hydrogen is +1 in covalent compounds, -1 in binary hydrides In a compound, the sum must be zero For ions, sum must equal overall charge Fluorine always has an oxidation number of -1 in compounds. Cl, Br, and I always have oxidation numbers of -1 in compounds, except when combined with oxygen and fluorine. Assign an oxidation number to the underlined atom in each ion or molecule. Fe2O3 H2SO4 CO32 NO2- 3.10 Classifying Reactions in Aqueous Solution Precipitation Reactions Acid-Base Reactions Gas-Forming Reactions Oxidation-Reduction 4.1 Mass Relationships stoichiometric factor: a mole ratio based on the coefficients of reactants or products in the balanced equation What mass of oxygen, O2, is required to completely combust 454 g of propane, C3H8? What masses of CO2 and H2O are produced? C3H8 + 5O2 3CO2 + 4H2O 4.2 Limited Reactants Pure silicon, required for computer chips and solar cells, is made by the reaction SiCl4 + 2Mg Si + 2MgCl2 If you begin with 225 g each of SiCl4 and Mg, which is the limiting reactant in this reaction? What quantity of Si, in grams, can be produced? 4.3 Percent Yield Methanol, CH3OH, can be burned in oxygen to provide energy, or it can be decomposed to form hydrogen gas, which can then be used as a fuel. CH3OH 2H2 + CO If 125 g of methanol is decomposed, what is the theoretical yield of hydrogen? If only 13.6 g of hydrogen is obtained, what is the percent yield of the gas? 4.4 Quantitative Analysis A substance of unknown concentration may be allowed to react with a known quantity of something else OR An unknown substance can be converted to one or more substances of known composition (back track) Chemical Analysis One method for determining the purity of a sample of titanium (IV) oxide, TiO2, an important industrial chemical, is to combine the sample with bromine trifluoride. 3TiO2 + 4BrF3 3TiF4 + 2Br2 + 3O2 Oxygen gas is evolved quantitatively. The gas can be captured readily, and its mass can be determined. Suppose 2.367 g of a TiO2-containing sample evolves 0.143 g of O2. What is the mass percent of TiO2 in the sample? Determining an Unknown Formula The unknown pure compound is decomposed into known products. The reaction products are isolated in pure form and the amount of each is determined. The amount of each product is related to the amount of each element in the original compound to give the empirical formula. Determining an Empirical Formula A 0.523 g sample of the unknown compound CxHy is burned in air to give 1.612 g of CO2 and 0.7425 g of H2O. A separate experiment gave a molar mass for CxHy of 114 g/mol. Determine the empirical and molecular formulas for the hydrocarbon. 4.5 Concentrations in Solutions 1 mole element = 6.02 x 1023 atoms 1 mole molecular compound = 6.02 x 1023 molecules 1 mole ionic compound = 6.02 x 1023 formula units Molarity Molarity = moles of solute per liter of solution Example: Prepare 2.00L of 0.250M NaOH from solid NaOH. Sodium bicarbonate, NaHCO3, is used in baking powder formulations and in the manufacture of plastics and ceramics, among other things. If 26.3 g of the compound is dissolved in enough water to make exactly 200mL of solution, what is the molar concentration of NaHCO3? What are the concentrations of the ions? An experiment in your laboratory requires 250.mL of a 0.0200M solution of AgNO3. You are given solid AgNO3, distilled water, and a 250.mL volumetric flask. Describe how to make up the required solution. Dilutions Prepare 2.00L of 0.250M NaOH from 1.00M NaOH. In one of your laboratory experiments, you are given a solution of CuSO4 that has a concentration of 0.15M. If you mix 6.0mL of this solution with enough water to have a total volume of 10.0mL, what is the concentration of CuSO4 in this new solution? An experiment calls for you to use 250. mL of 1.00M NaOH, but you are given a large bottle of 2.00M NaOH. Describe how to make the 1.00M NaOH in the desired volume. 4.6 pH pH can be calculated directly from hydrogen ion concentration Can use the antilog of pH to figure out hydrogen ion concentration What is the pH of a solution of HCl, where [HCl] = 2.6 x 10-2 M? What is the hydrogen ion concentration in saturated calcium hydroxide with a pH of 12.45? 4.7 Solution Stoichiometry If you combine 75.0 mL of 0.350M HCl and an excess of Na2CO3, what mass of CO2 (in grams) should be produced? Titrations A titration can be used to determine the concentration of an unknown solution. A substance, present in unknown quantity, can be allowed to react with a known quantity of another substance. If the stoichiometric ratio for their reaction is known, the unknown quantity can be determined. Acid-base indicators are used to tell when the reaction is complete. When the solution changes color, the equivalence point (also called end point) has been reached. The amount of OH- equals the amount of H+. Amount of base (mol) = conc. of base (M) x volume of base (L) A 25.0 mL sample of vinegar requires 28.33 mL of a 0.953 M solution of NaOH for titration to the equivalence point. What mass (in grams) of acetic acid is in the vinegar sample, and what is the concentration of acetic acid in the vinegar? CH3CO2H + NaOH NaCH3CO2 + H2O Often the concentration of the base is unknown and must be determined through standardization. primary standard: a sample of solid acid or base is titrated with a solution of the base or acid to be standardized titrate with another solution that is already standardized Standardization Hydrochloric acid can be purchased from chemical supply houses with a concentration of 0.100M, and such a solution can be used to standardize the solution of a base. If titrating 25.00 mL of a sodium hydroxide solution to the equivalence point requires 29.67mL of 0.100M mL, what is the concentration of the base? Redox Titration A sample containing vitamin C is titrated with Br2, an oxidizing agent. C6H8O6 + Br2 2HBr + C6H6O6 A 1.00g “chewable” vitamin C tablet requires 27.85mL of 0.102M Br2 for titration to the equivalence point. What is the mass of vitamin C in the tablet? 4.8 Spectrophotometry It is possible to measure the extent of light absorption through a solution that is colored, and then you can relate this to the concentration of the dissolved solute. This method of quantitatively measuring light absorption is called spectrophotometry. Transmittance Absorbance The Beer-Lambert Law Absorbance also increases as path length increases. There is a linear relationship between a sample’s absorbance and its concentration for a given path length. Spectrophotometic Analysis Record the absorption spectrum of the substance to be analyzed. Choose the wavelength for the measurement. Prepare a calibration plot/curve.