Lecture 15: Review of Kraft Pulping
advertisement

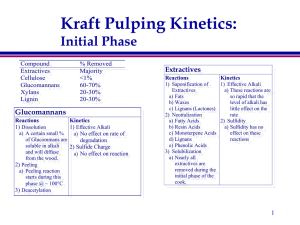
Pulping and Bleaching PSE 476/Chem E 471 Lecture #15 Kraft Pulping Review of Reactions/Kinetics Effective Alkali (g/l NaOH) Kraft Pulping 60 70°C 70°C 137°C 50 40 30 Impregnation zone 170° C 20 Initial Phase Bulk Phase Residual Phase 10 0 0 5 10 15 20 Yield of Lignin (%) 25 30 Kraft Pulping Kinetics: Initial Phase Compound Extractives Cellulose Glucomannans Xylans Lignin % Removed Majority <1% 60-70% 20-30% 20-30% Glucomannans Reactions 1) Dissolution Kinetics 1) Effective Alkali a) A certain small % a) No effect on rate of of Glucommans are degradation soluble in alkali 2) Sulfide Charge and will diffuse a) No effect on reaction from the wood. 2) Peeling a) Peeling reaction starts during this phase @ ~ 100C 3) Deacetylation Extractives Reactions Kinetics 1) Saponification of 1) Effective Alkali Extractives a) These reactions are a) Fats so rapid that the b) Waxes level of alkali has c) Lignans (Lactones) little effect on the 2) Neutralization rate a) Fatty Acids 2) Sulfidity b) Resin Acids a) Sulfidity has no c) Monoterpene Acids effect on these d) Lignans reactions e) Phenolic Acids 3) Solubilization a) Nearly all extractives are removed during the initial phase of the cook. Initial Reactions: Low Temperature • Carbohydrates » Alkaline hydrolysis of acetyl groups on xylan (see next slide). » Removal of certain soluble carbohydrates. - Certain galactoglucomannans. - Arabinogalactans. • Extractives » Alkaline hydrolysis of fats (saponification), waxes, and other esters. » Neutralization of extractives. - There are a number of acidic extractives which consume NaOH. Alkaline Hydrolysis: Example Using Acetyl Groups CH2OH OH HO OH CH2OH O OH - HO OH CH2OH OH HO O C CH3 O OH O OH O C CH3 O O HO + OH HO C CH3 O • Esters are cleaved in alkaline solutions through hydrolysis reactions forming carboxylic acids and alcohols. • Hydrolysis of acetyl groups occurs readily in alkaline solutions. » Reaction occurs rapidly even at room temperature. • Reaction consumes alkali. Saponification of Fats (Review slide from PSE 406) • Treatment of fats with alkali converts them to fatty acids and glycerol through saponification. OH O H2C O C HOCH2CHCH2OH R - OH Glycerol (glycerine) - O H2C O C OH R H2O O H2C OH R - O C Once again this reaction consumes part of the alkali charge. Removal of Glucomannans: 120 200 100 150 80 25% EA 60 100 15.8% EA 40 50 20 0 0 0 50 100 150 200 Time (minutes) Why doesn’t EA affect glucomannan removal? •Peeling reaction not sensitive to OH concentration •Rapid removal at the beginning of the cook (dissolving) •Over 100C greater rate of peeling •70% removed rate drops again 250 300 Temperature (C) Glucomannan Yield (%) Effect of Effective Alkali Kraft Pulping Kinetics: Initial Phase Xylans Lignin Reactions Kinetics 1) Cleavage of -O-4 1) Effective Alkali a) No affect on linkages rate during this a) Units with free phase phenolic hydroxyl b) Rate diffusion b) Very fast limited in this 2) Cleavage of -O-4 phase linkages 2) Sulfidity a) Units with free a) Sulfidity has no phenolic hydroxyl effect during or carbonyl. this phase b) Rapid reaction 3) Solubilization a) Small amount of lignin alkali soluble 4) Condensation a) Limited Reactions Kinetics 1) Dissolution 1) Effective Alkali a) Major reaction of a) Higher EA increases the Xylans during kraft rate of dissolution pulping 2) Sulfide Charge 2) Peeling a) No effect on reaction a) Very minor reaction starts @ ~ 100C 3) Stopping a) See 2a 4) Deacetylation Cellulose Reactions Kinetics 1) Peeling 1) Effective Alkali a) Loss of cellulose begins a) ? 2) Sulfide Charge @ ~ 120C - 130 C 2) Minor reaction as only 10% a) No effect on reaction of all cellulose lost during cook. Lignin Removal: Effect of Sulfidity • Sulfidity effect zero order in initial delignification (this means that changing sulfidity has no effect on initial rate) » Sulfidity is important during bulk delignification » Effects final lignin/carbohydrate ratio - HS- does not react with carbohydrates 40 KAPPA 30 It depends on: •Wood species •EA •Cooking temp. •Hardwoods 20% •Softwoods 25% 20 10 0 0 10 20 30 Sulfidity 40 50 60 Removal of Xylans: Effect of Effective Alkali 200 100 150 80 60 100 40 25% EA 15.8% EA 20 50 0 0 50 100 150 200 Time (minutes) Why does EA affect xylan removal? •Stable at the beginning •Hydrolysis and 2nd peeling 250 0 300 Temperature (C) Xylan Yield (%) 120 Kraft Pulping Kinetics: Bulk Phase Compound Extractives Cellulose Glucomannans Xylans Lignin % Removed (total) Majority <10% 60-70% 30-50% 80-85% Cellulose Reactions Kinetics 1) Peeling 1) Effective Alkali a) Loss of cellulose a) ? accelerates at temperature 2) Sulfide Charge due to glycosidic cleavage a) No effect on reaction 2) Stopping a) Stopping reactions slow loss of cellulose 3) Glycosidic Cleavage a) See 1a b) Starts to reduce cellulose viscosity Lignin Reactions Kinetics 1) Cleavage of non 1) Effective Alkali a) Higher EA = phenolic -O-4 faster rate of followed by: degradation a) Cleavage of -O-4 2) Sulfide Charge linkages in units a) Higher sulfide with free phenolic charge increases hydroxyl the rate of lignin b) Cleavage of -O-4 removal. linkages in units b) Higher lignin with free phenolic removal rate hydroxyl Rapid results in a reaction lower lig/carb at c) Condensation end of cook d) Cleavage of C-C bonds (double) Kraft Pulping Kinetics: Bulk Phase Xylans Reactions Glucomannans Kinetics 1) Dissolution 1) Effective Alkali a) Major reaction of a) Higher EA increases the Xylans during this rate of dissolution phase 2) Sulfide Charge 2) Peeling a) No effect on reaction a) Minor reaction 3) Glycosidic Cleavage a) Will increase rate of peeling reaction 4) Precipitation on fibers a) Starts as alkali consummed Reactions 1) Peeling Kinetics 1) Effective Alkali a) Peeling is b) No effect on rate of extensive until degradation about 70% of xylan 2) Sulfide Charge is lost and then a) No effect on reaction almost stops 2) Gylcosidic cleavage a) Increases the rate of peeling 3) Stopping a) Stops losses due to peeling Kraft Pulping Kinetics: Residual Phase Compound Extractives Cellulose Glucomannans Xylans Lignin % Removed (total) Majority ~10% 70-80% 40-50%% 85-95% Cellulose Reactions 1) Glycosidic Cleavage Kinetics 1) Effective Alkali a) Major reaction resulting in a) ? 2) Sulfide Charge lower molecular weight a) No effect on reaction and strength loss 2) Peeling/stopping a) Minor reactions Lignin Reactions Kinetics 1) Majority of ether 1) Effective Alkali linkages that will a) The rate slows cleave are gone: rapidly: 2) Cleavage of C-C b) amount of lignin bonds (double) remaining a 3) Condensation function of the a) Increase the Mw EA and sulfide and lower reactivity charge during of remaining lignin the cook Kraft Pulping Kinetics: Residual Phase Xylans Reactions 1) Peeling/stopping/ a) negligible 2) Precipitation on fibers a) Significant amount deposited as alkali continues to be consummed Kinetics 1) Effective Alkali a) Lower levels of alkali at end of the cook casuse deposition of xylans 2) Sulfide Charge a) No effect on reaction Glucomannans Reactions 1) Peeling Kinetics 1) Effective Alkali a) Limited b) No effect on rate of 2) Gylcosidic cleavage degradation a) Limited 2) Sulfide Charge 3) Stopping a) No effect on reaction a) Limited