NMR – Effect of Magnetic Field

Chapter 13 NMR Spectroscopy
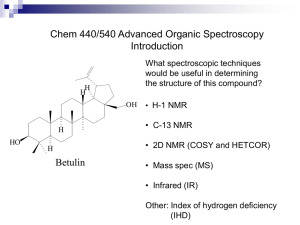
NMR - Nuclear Magnetic Resonance
NMR is a form of spectroscopy that uses an instrument with a powerful magnet to analyze organic compounds.
Invented by physicists (1950’s), then used by chemists (1960’s).
Why is it called NMR?
Nuclear Magnetic Resonance
Nuclear – because it looks at the nucleus of an atom, most commonly a hydrogen atom.
A hydrogen atom nucleus consists of one proton with a
+1 charge and “spin” of ½. It acts like a tiny bar magnet.
generates magnetic field
NMR – Effect of Magnetic Field
No external magnetic field applied to sample
Random orientation of nuclear spins
Sample placed in an external magnetic field
NMR: Absorption of Energy
Initial State – nucleus at low energy level
NMR: Information Obtained from a Spectrum
An NMR Spectrum will generally provide three types of information:
Chemical Shift
– indicates the electronic environment of the nucleus (shielded or deshielded)
Integration – gives the relative number of nuclei producing a given signal
Spin-Spin Coupling – describes the connectivity
1 H NMR Spectrum – H
2
O
A sample of water is placed in an NMR instrument, and a proton spectrum is recorded (scanned from left to right).
When does nucleus absorb energy?
2, External Field (H o
) from magnet
3.
NMR: Simple 1 H NMR Spectrum
Showing Chemical Shift
Two types of protons (a CH
2 and a CH
3
) give two separate signals at two different chemical shifts.
NMR: Chemical Shift Practice
Group
-O-CH
3
-Si-CH
3
-C-CH
3
Cl
3
C-H
EN
Assign the four groups shown to the four NMR singals, based on each element’s electronegativity.
NMR: Chemical Shift Regions
Chemical shift zero is set to
TMS (tetramethylsilane), a reference compound
Chemical shift measured in ppm.
NMR: Chemical Shift Regions
Alkane Region (high electron density):
Heteroatom Region:
Double Bond Region:
NMR: Chemical Equivalence and
Number of Signals
How many signals will the following compounds show in their
1 H NMR Spectrum? (Hint: check for symmetry)
O
OMe
Br
H
H
H
H
H
H
NMR: Chemical Equivalence and
Number of Signals
How many signals should appear in the proton NMR spectrum for these compounds?
O octane
In theory:
Signals actually resolved:
NMR: Overlapping Proton Signals
Protons b, c, and d are all nearly the same, and their signals are not resolved in this spectrum.
Review: How Many NMR Signals?
How many signals will the following compounds show in their
1 H NMR Spectrum? (Hint: check for symmetry)
CH
3
CH
2
Cl
H
H
H
H
H
C
Cl
C
H
H
H
H
H
H H
CH
3
No rotation about double bonds
NMR: Chloroethane
H
C
H H
H H
C
Cl
Fast rotation around single bonds gives an “averaged” spectrum for the three methyl hydrogens.
NMR: A Second Proton Spectrum
Note: the signal for the nine methyl H’s is larger than the CH
2 signal
NMR: Integration Indicates
Relative Number of Nuclei
The height of the integration line (“integral”) gives you the relative number of nuclei producing each signal.
NMR: Splitting into a Doublet doublet
Note that the signal at 1.6 ppm for the methyl group is split into two peaks.
Remember that this is one signal, composed of two separate peaks.
NMR: Signal Splitting, n+1 Rule
• A signal is often split into multiple peaks due to interactions with protons on carbons next door.
Called spin-spin splitting
• The splitting is into one more peak than the number of H’s on adjacent carbons (“n+1 rule”)
• Splitting of a signal can give doublets (two peaks), triplets (three peaks), quartets (4 peaks), ect.
• The relative intensities given by Pascal’s Triangle: doublet 1 : 1 triplet 1 : 2 : 1 quartet 1 : 3 : 3 : 1 pentet: 1 : 4 : 6 : 4 : 1
NMR: Signal Splitting, n+1 Rule n+1 Rule:
A signal in the proton NMR spectrum will be split into n+1 peaks, where n is the number of protons on adjacent carbons.
Example: CH
3
-CH
2
-Br
For the Methyl Group –
There are two protons ‘next door’ (n=2), so the methyl signal will be split into three peaks (2+1), which is called a triplet.
For the CH
2
Group:
Three protons next door means the CH
2
4 (3+1) peaks, called a quartet. signal will be split into
1 H NMR Spectrum for Bromoethane integration:
2 H 3 H
Note the expansions printed above
NMR: Signal Splitting, n+1 Rule
NMR: Signal Splitting, n+1 Rule
H
NMR: Origin of Spin-Spin Splitting
Net result:
NMR: Doublets and Triplets
Doublet: the one proton next door can be either up or down ( α or β)
Triplet: for the two protons next door, there are four combinations possible:
α α α β β β
β α
NMR: Signal Splitting, n+1 Rule
NMR: Using the n+1 Rule
Using the n+1 rule, predict the 1 H NMR spectrum of 2-iodopropane.
Give splitting pattern, integration, and approximate chemical shift.
I
H
3
C
I
C
H
CH
3
Note that the methyl groups are equivalent, so they will give one signal in the NMR spectrum.
NMR: Spectrum of 2-iodopropane doublet
Seven line pattern
NMR: Rules for Spin-Spin Splitting
• The signal of a proton with n equivalent neighboring H’s is split into n + 1 peaks
• Protons farther than two carbon atoms apart do not split each other
• Equivalent protons do not split each other
Common
1
H NMR Patterns
1. triplet (3H) + quartet (2H)
2. doublet (1H) + doublet (1H)
3. large singlet (9H)
4. singlet 3.5 ppm (3H)
5. large double (6H) + muliplet (1H)
6. singlet 2.1 ppm (3H)
Common
1
H NMR Patterns
7. multiplet ~7.2 ppm (5H)
8. multiplet ~7.2 ppm (4H)
9. broad singlet, variable chemical shift