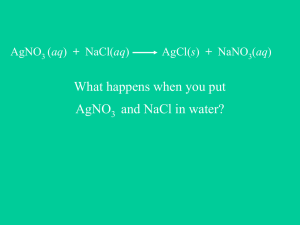8.3 ppt
advertisement

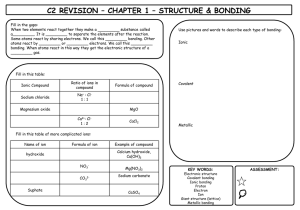
Warm Up Review Classify the type of reaction 1. AgNO3 + NaCl AgCl + NaNO3 2. 2 PbO2 2 PbO + O2 3. 2 Al + 3 CuSO4 Al2(SO4)3 + 3 Cu Predict the products of the following reactions: 4. Zn + 2 AgNO3 5. MgCl2 + Ca(OH)2 6. 2Mg + O2 Warm Up Review Classify the type of reaction 1. AgNO3 + NaCl AgCl + NaNO3 double displacement 2. 2 PbO2 2 PbO + O2 decomposition 3. 2 Al + 3 CuSO4 Al2(SO4)3 + 3 Cu single displacement Predict the products of the following reactions: 4. Zn + 2 AgNO3 Zn(NO3)2 + 2Ag 5. MgCl2 + Ca(OH)2 Mg(OH)2 + CaCl2 6. 2Mg + O2 2MgO Quiz 8.2 on Wed 12/17… Study 8.3 Reactions in Aqueous Solution H2O • Water is a combination of two ions H+ and OH• The reaction of ionic compounds in water (aqueous solution) allows the ionic compounds to disassociate in the solution. • (aq) indicates that in solution the ions are formed AgNO3 (aq) Ag+ (aq) and NO3- (aq) • Some ions combine in solutions to form insoluble solids. • These insoluble solids are called precipitates. • Precipitates are indicated by (s) – They do not dissolve in solution to form ions Ag+ + Cl- AgCl (s) Precipitates Writing Complete Ionic Equations • An equation that shows the dissolved ionic compounds (ie. The ions) Mg(NO3)2 (aq) + Na2CO3 (aq) MgCO3 (s) + 2 NaNO3 (aq) Writing Complete Ionic Equations • An equation that shows the dissolved ionic compounds (ie. The ions) Mg(NO3)2 (aq) + Na2CO3 (aq) MgCO3 (s) + 2 NaNO3 (aq) Mg2+ (aq) ** Your turn- try to complete the rest of ionic eqaution! Writing Complete Ionic Equations • An equation that shows the dissolved ionic compounds (ie. The ions) Mg(NO3)2 (aq) + Na2CO3 (aq) MgCO3 (s) + 2 NaNO3 (aq) Mg2+ (aq) + 2 NO3¯ (aq) + 2 Na+ (aq) + CO32- (aq) MgCO3 (s) + 2 Na+ (aq) + 2 NO3¯ (aq) • Ions that appear on both sides of the equation are not directly involved in the reaction. • They are called spectator ions Mg(NO3)2 (aq) + Na2CO3 (aq) MgCO3 (s) + 2 NaNO3 (aq) Mg2+ (aq) + 2 NO3¯ (aq) + 2 Na+ (aq) + CO32- (aq) MgCO3 (s) + 2 Na+ (aq) + 2 NO3¯ (aq) • The equation can be re-written with out the spectator ions. This is called a Net ionic equation. Mg(NO3)2 (aq) + Na2CO3 (aq) MgCO3 (s) + 2 NaNO3 (aq) Mg2+ (aq) + 2 NO3¯ (aq) + 2 Na+ (aq) + CO32- (aq) MgCO3 (s) + 2 Na+ (aq) + 2 NO3¯ (aq) Mg2+ (aq) + CO32- (aq) MgCO3 (s) Predicting the formation of a Precipitate 1. Write the ions formed by your reactants 2. Identify how the ions (cations and anions) will rearrange 3. Look at table 8.3 (pg. 227) to see if the newly formed products are soluble – If they are soluble, that means they will dissolve into ions – If they are insoluble, they will be solids (precipitates) Predicting the Formation of a Precipitate: Example SrBr2 (aq) + K2SO4 (aq) First: Identify the Ions formed Sr2+ (aq) + 2 Br¯ (aq) + 2 K+ (aq) + SO42- (aq) Predicting the Formation of a Precipitate: Example SrBr2 (aq) + K2SO4 (aq) First: Identify the Ions formed Sr2+ (aq) + 2 Br¯ (aq) + 2 K+ (aq) + SO42- (aq) Second: Identify how the ions will rearrange SrSO4 and KBr Predicting the Formation of a Precipitate: Example SrBr2 (aq) + K2SO4 (aq) First: Identify the Ions formed Sr2+ (aq) + 2 Br¯ (aq) + 2 K+ (aq) + SO42- (aq) Second: Identify how the ions will rearrange SrSO4 and KBr Third: Look at Table 8.3 to identify which compounds are soluble. – Notice the rule for sulfates (SO4); soluble except when in compounds of Pb, Ag, Hg, Ba, Sr, and Ca – Notice the rule for salts of alkali metals and ammonia (they are soluble) Predicting the Formation of a Precipitate: Example SrBr2 (aq) + K2SO4 (aq) First: Identify the Ions formed Sr2+ (aq) + 2 Br¯ (aq) + 2 K+ (aq) + SO42- (aq) Second: Identify how the ions will rearrange SrSO4 (s) and KBr (aq) Third: Look at Table 8.3 to identify which compounds are soluble. – Notice the rule for sulfates (SO4); soluble except when in compounds of Pb, Ag, Hg, Ba, Sr, and Ca – Notice the rule for salts of alkali metals and ammonia (they are soluble) Lets re-write the net ionic equation using this information! Sr2+ (aq) + 2 Br¯ (aq) + 2 K+ (aq) + SO4 (aq) SrSO4 (s) + 2 K+ (aq) + 2 Br¯ (aq) Sr2+ (aq) + SO42- (aq) SrSO4 (s) The End! Begin HW so that I am able to help you during class.