Worked Examples: Chapter 8

Chapter 8 Worked Example 1
The decavanadate ion is a complex species with chemical formula V
10
O
28
6. It reacts with an excess of acid to form the dioxovanadium ion VO
2
+ , and water. Write a balanced equation and state the oxidation state of vanadium before and after reaction. Which vanadium oxide has the same oxidation state of V?
Solution
V
10
O
28
6+ 16H + 10 VO
2
+ + 8H
2
O
Oxidation state of V in both species is +5 {V(V)}
V
2
O
5 is the V(V) oxide
Chapter 8 Worked Example 2
Determine the oxidation state of the metal in each of the
Following coordination complexes:
[V(NH
3
)
4
Cl
2
], [Mo
2
Cl
8
] 4, [Co(H
2
O)
2
NH
3
Cl
3
] ,
[Ni(CO)
4
]
Solutions
[V(NH
3
)
4
Cl
2
] [Mo
2
Cl
8
] 4[Co(H
2
O)
2
NH
3
Cl
3
] [Ni(CO)
4
]
V = +2 Mo = +2 Co = +2 Ni = 0
Chapter 8 Worked Example 3
Give the chemical formula that corresponds to each of the following compounds.
(a) Sodium tetrahydroxozincate (II)
(b) Dichlorobis(ethylenediamine)cobalt(III) nitrate
(c) Triaquabromoplatinum (II) chloride
(d) Tetra-amminedinitroplatinum (IV) bromide
Solutions
(a) Na
2
[Zn(OH)
4
(b) [Coen
2
Cl
2
]NO
3
(c) [Pt(H
2
O)
3
Br]Cl
(d) [Pt(NH
3
)
4
(NO
2
)
2
]Br
2
Chapter 8 Worked Example 4
Determine the oxidation state of Mn in the following oxides and oxoanion.
MnO MnO
2
Mn
2
O
3
Mn
2
O
7
Mn
3
O
4
[MnO
4
] -
Solutions
+2 +4 +3 +7 +2, +3 +7
MnO MnO
2
Mn
2
O
3
Mn
2
O
7
Mn
3
O
4
[MnO
4
] -
Chapter 8 Worked Example 5
Draw the structures of all possible isomers for the following complexes. Indicate which isomers are enantiomeric pairs.
(a) Diamminebromochloroplatinum(II) (square planar)
(b) Diaquachlorotricyanocobaltate(III) ion (octahedral)
(c) Trioxalatovanadate(III) ion (octahedral)
Solutions
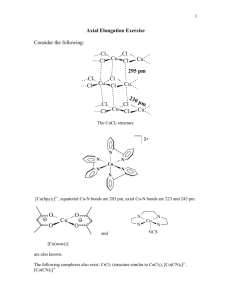
(a)
Br
Cl
Pt
NH
3
NH
3 ci s
NH
Br
3
Pt
NH
3
Cl t r ans
CN
_
OH
2
_
OH
2
(b)
H
2
O
H
2
O
Co
CN
CN
NC
NC
Co
(c)
O
C
O
C O
O
Cl f ac
Cl mer ( cis) mirror plane
O O
O
C
O
C
V
O
O
O
O C
O
O
C
C
O
O
O
C
Two enantiomers
C
O
C
O
V
O
CN
OH
2
O
O
C
O
C
O
NC
NC
Co
CN
Cl
OH
2 mer ( tr ans)
_
Chapter 8 Worked Example 6
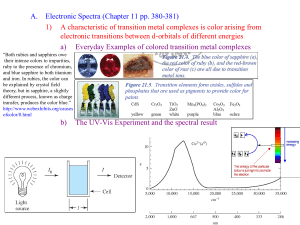
(a) Explain why octahedral complexes with three and eight d electrons on the central metal atom are particularly stable.
(b) Explain under what circumstances you would expect octahedral complexes with five or six d electrons on the central metal atom to be particularly stable.
Solutions
(a) According to the crystal field theory, the d-electron configurations of d
3 and d
8 octahedral complexes are e g
E d
3
(t
2g
)
3 half-filled t
2g subshell t
2g d
8
(t
2g
)
6
(e g
)
2 filled t
2g subshell and half-filled e g subshell
(b) According to the crystal field theory, the d-electron configurations of d
5
(high spin) and d
6
(low spin) octahedral complexes are e g
E d
5
(high spin)
(t
2g
)
3 and e
(e g g
)
2 half-filled t
2g subshells t
2g
(t
2g d
)
6
6
(low spin) filled t
2g subshell
Chapter 8 Worked Example 7
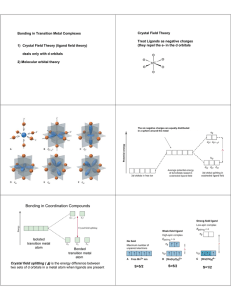
The three complex ions [Mn(CN)
6
] 5, [Mn(CN)
6
] 4, and [Mn(CN)
6
] 3have all been synthesized and are all low-spin octahedral complexes.
For each complex ion, determine the oxidation number of Mn, the configuration of the d-electrons (how many t
2g and the number of unpaired electrons.
Solution and how many e g
),
[Mn(CN)
6
]
5-
[Mn(CN)
6
]
4-
Mn(CN)
6
]
3e g
E t
2g
Mn(I) (+1)
2 upaired electrons
(full configuration is
[Ar]3d
5
4s
1
)
(t
2g
)
5
Mn(II) (+2)
1 upaired electron
(full configuration is
[Ar]3d
5
)
(t
2g
)
5
Mn(III) (+3)
2 upaired electrons
(full configuration is
[Ar]3d
4
)
(t
2g
)
4
Chapter 8 Worked Example 8
Three different compounds are known to have the empirical formula CrCl
3
.6H
2
O. When exposed to a dehydrating agent, compound 1 (dark green) loses 2 mol water per mol of compound, compound 2 (light green) loses 1 mol water and compound 3
(violet) loses no water.
Solution
Water of hydration is much easier to remove than water ligands:
Compound 1 is [Cr(H
2
O)
4
Cl
2
]Cl.2H
2
O
Compound 2 is [Cr(H
2
O)
5
Cl]Cl
2
.H
2
O
Compound 3 is [Cr(H
2
O)
6
]Cl
3