Real World Gasoline Analysis
advertisement

Gasoline Analysis
{
By: Travis Slaysman and Drew Landerman
Brainstorming
Urushiol extraction
Gasoline Comparison
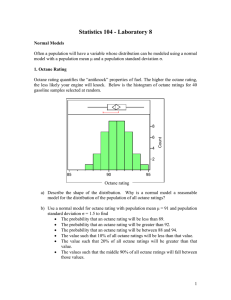
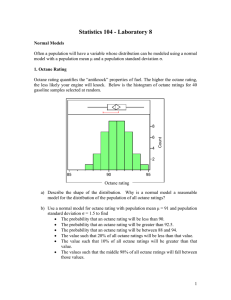
Octane Rating
Methodology
Sample Acquisition
Standard Creation
Sample Dilution
Experimentation
Standard Running
Sample Running
Data
Spectrum Development
Peak Determination
Standard
Formula
Boiling Point
Cyclohexane
C6H12
80.7°C
Heptane
C7H16
98.4°C
Octane
C8H18
125°C
Nonane
C9H20
151°C
Decane
C10H22
174.1°C
Benzene
C6H6
80.1°C
Ethanol
C2H6O
78.4°C
Toluene
C7H8
110.6°C
Analysis
Peak Comparison
Mass Spec. Utilization
Sample Spectrum Comparison
Octane Rating is based on comparison with the
burning characteristics of a mixture of nheptane and 2,2,4-trimethylpentane (isooctane).
Gasoline that isn’t formulated properly can
lead to engine knocking and increase the
incidence of incomplete combustion, resulting
in increased carbon monoxide production.
Octane Rating Formulation
Decane in Cyclohexane
Cyclohexane
Octane in Cyclohexane
Toluene in Cyclohexane
Hydrocarbon
Retention Time (min)
Hexane
2.561
Heptane
1.916
Octane
2.35
Nonane
4.212
Decane
5.275
Benzene
4.113
Toluene
2.609
Cyclohexane
1.829
1,2,4-Trimethyl benzene
~4.82
1,2,3,5-tetramethyl benzene
~5.79
o-xylene & p-xylene
~5.2
Retention Times
Volatile organic sample evaporation
Purity of the reagents we used
Mass Spec Utilization
Sources of Error
Conclusions
As the octane rating went up, the alkane
concentrations became smaller, as we expected.
Toluene, Benzene and related compounds
became more concentrated as octane rating
went up.
Cyclohexane was also increasing in
concentration as the octane rating went up.
Royal Farms seemed to have cleaner
chromatograms than Wawa did, this could be
due to possible detergents and other additives
put in the gasoline.