this lab is due october 12
advertisement

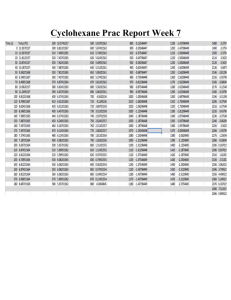
Lab. # 3: Cyclohexane Production Chemical Process Component Design 0906.401 THIS LAB IS DUE OCTOBER 12 SIMULATION OF A CYCLOHEXANE PLANT Part 1: Solve the heat and material balances of the cyclohexane plant shown in the Figure below: 500 psig C1 Recycle S13 S12 Eff. 80% S6 SP1 300 F Purge Gas S3 S11 E2 DP=15 psi S7 R1 S4 BENZENE 120 F S5 S1 HYDROGEN M1 F1 E1 S10 S9 E3 Product FEED STREAMS COMP.\STREAM BENZENE HYDROGEN Hydrogen (mole %) -- 97.5 Methane (mole %) -- 2.5 Benzene (mole %) 100 -- Cyclohexane (mole %) -- -- Temperature (F) 100 100 Pressure (psig) 0 500 Flowrate (bbl/hr) 100 Flowrate (std ft3/hr) 600,000 PROCESS CONDITIONS Use SRK for your initial run. Heat exchangers pressure drop = 5 psia. Reactor pressure drop = 15 psia. Specify heat exchanger E-1 and E-2 as having cold stream outlet temperature of 250 F and 300 F respectively. Set up splitter specification so that 20% of the splitter feed exits as the purge gas. For F-1 use an adiabatic flash (duty zero). 2 REACTION DATA Vapor Phase. The reactor is jacketed and controlled to 435 F. 95% conversion of benzene to cyclohexane Use a conversion reactor. Enter the reaction data through “Flowsheet / Reaction Package”. Part 2 Would Grayson-Streed. (GS) be better? If so, use and compare results. Part 3 Change the split ratio of the splitter. Explain how does that affect the levels of CH4 and H2 in the system. Keep in mind that you want to produce Cyclohexane at a minimum cost. a) Do we want a large split ratio or a small one? Is there any trade-off? b) If the feed of the reactor should have a relation of hydrogen to benzene of 15:1, how can you achieve it? 3