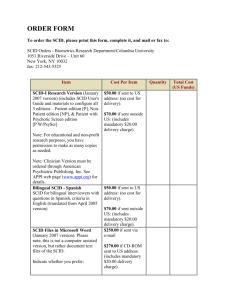
B - icord
advertisement

Research on rare diseases follows a long and complex pathway (1) Identification of a clinical phenotype Mendelian Genetics Gene identification Validation cohort clinical expertise databases technical platforms Genotyping Sequencing «Variants» (same phenotype without mutation) Genotype/phenotype correlation ? DIAGNOSIS Gene modifiers PROGNOSIS Pathophysiology Cell studies Gene expression analysis Animal models In vivo studies Bioinformatics, cell imaging… Animal transgenic facilities Phenotyping facilities (Imaging…) Research on rare diseases follows a long and complex pathway (2) Therapeutic research Compounds libraries Screening Industrial Collaborations Targets Pathophysiology Screening of molecules Class of molecules Biological molecules Cell and gene therapy in vitro Vectorology large animals facilities in vivo Animal models Clinical trials specific methodology THERAPEUTICS The "reference center" for primary immunodeficiencies (PID). Necker University Hospital Paris PID diagnosis laboratory Dpt of Biotherapy research Faculté deU550 Médecin e U429 UIH Pediatrics Infectiology, immunology adults Data base center SCID diseases Fatal conditions early in life X-linked or autosomal recessive inheritance T HSC Myeloid compartment THYMUS T B 1970s SCID diseases 1985 NK Abnormal thymus in SCID HSC CLP Myeloid compartment Control THYMUS SCID CD8 CD4 B X-linked SCID 1985-1995 3 22. 2 IL-15 1 p 21. 3 NK gc deficiency 2 1 11. 11. 12 4 3 23 22 21 1 1 2 TIMP DXS255 DXS14 DXZ1 DXS159 13 PGK1 DXS447 1 21. 2 q 23 XL-SCID Déficit immunitaire combiné sévère gc gene T CD4 IL-7 HSC T CD8 DXS3 3 22. CLP B DXS178 1 2 3 DXS94 24 DXS17 25 DXS42 IL-4 IL-2 IL-7 IL-9 IL-15 IL-21 DXS37 26 IL-2Rb IL-2Ra 27 28 X gc IL-4Ra gc IL-7Ra gc IL-9R gc IL-2Rb IL-15Ra gc IL-21R gc T-B-SCID p Ly NK T gd Pro T Chr. 10 T CD8 T CD4 Precursor Pre-B B Pro B "T-B-SCID" B IgG, A, E B IgM q 100 Genomic deletions / Splice site mutations 1-3 5-6 4 5 b-Lact N M1T S32C R81X H35D Artemis 5-8 G118V G135E 10-12 9 10 % Survival 3 11 b-CASP del. 1bp R191X del. 4bp Y199X SCIDA C-ter 10 1 C Control RS-SCID RS-SCID + Arte del. 7bp del. 17bp ins. 2bp 0 1 Gy 2 1990-2001 3 T-B-SCID Artemis KO Spleen Thymus 5.7 76.1 61.8 8.7 4.5 WT 0.2 0.3 TCR and BCR genes rearrangements during T and B cell differentiation v 0.1 Cleavage KO 1.8 CD8 13.0 RSS j Rag-1-Rag-2 v IgM CD4 Hairpin coding ends RSS B220 j Ku70-Ku80 DNA-PKcs Hairpin opening and processing Artemis j v End joining Excised DNA XRCC4, ligase 4 Generation of coding joints 2001-2004 SCID diseases 1.Prevention of cell apoptosis DNA replication (purin metabolism) ADA 2005 2.gc cytokine-dependent signals gc, JAK-3, IL7Ra NK 4.Pre TCR/TCR signalling) CD45, CD3d, HSC CD8 CLP CD4 Myeloid compartment THYMUS 3.V(D)J recombination Rag-1/-2, Artemis B More diseases to be described… Molecular analysis of SCID conditions, what for ? 1. Basic science: lymphocyte development 2. Molecular diagnosis/genetic counseling 3. Specific therapies: ADA enzyme substitution, gene therapy 4. Design of new drugs: . ADA deficiency lymphotoxic role of deoxyadenosine 2 chloro deoxyadenosine, a cytotoxic drug used in the treatment of lymphomas . gc/JAK-3 deficiencies role of JAK-3 in lymphocyte development/survival/function JAK-3 inhibitor as a new immunosuppressive agent used in transplantation… Schematic representation of a major signaling pathway activated by IL-2 JAK-3 inhibitor GAS Description of multiple SCID variants Three examples: Omenn's syndrome Hypomorphic mutations of Artemis Revertant in XL-SCID Omenn’s Syndrome: hypomorphic mutations in Rag-1/2 emergence of autoreactive T cell clones skin CD3 staining 1 1043 K86Fs. gu t G720S R1010H R511H L732F K1029E R404W R404Q D832N S875Fs. Hypomorphic mutations of Rag-1 Defective central T cell tolerance Patient Control T173Fs. Y333• Oligoclonal T cell expansion Hypomorphic mutations ofArtemis associated with B cell lymphomas b-Lact C-ter b-CASP “Catalytic domain” “Regulatory domain” T432 fs D451 fs Hypomorphic mutations Phenotype ? Occurrence of B cell lymphomas CD20 Ki67 Role of Artemis as a genome caretaker An atypical case of XL-SCID gc deficiency NK NK Reverse mutation T CD4 HSC T CD8 B Absence of T cells Absence of NK cells T CD4 gc gc gc T CD8 gc gc B T cell counts = 50 % of control memory phenotype Repertoire diversity generated from one precursor (revertant cell) ? Revertant analysis • 1 T cell precursor 1 x 103 TCRVB clones 10 to 11 cell division cycles ~ 1 % of T cell repertoire diversity A form of natural gene therapy ! Probability of survival in SCID patients after hematopoietic stem cell transplantation according to donor-recipient compatibility 1968: the 1st successful HSCT was achieved in a patient with SCID HLA id. sibling T cells/µl Survival % 3,000 HLA haplo id. parent Homeostatic expansion of donor mature T cells HLA id. Haplo id. 2,000 New T cell ontogeny 1,000 0 1 2 3 4 6 12 months Months European registry - 464 pts Persisting severe immunodeficiency Lymphopoiesis in SCID patients pre/post hematopoietic stem celltransplantation (HSCT) Pre HSCT Early post HSCT Donor Late post HSCT NK T HSC Exhaustion of precursors T Immunodeficiency ! Myeloid cells No donor cells in bone marrow Occurrence of severe chronic skin HPV disease in SCID patients after HSCT gc, JAK3 deficiencies Occurrence of severe chronic skin HPV disease in SCID patients after HSCT • A significant problem with no simple solution • An unexpected role for gc/JAK-3 cytokine receptor signaling pathways in defense of keratinocytes against papilloma virus New therapeutics ? Principle of ex vivo gene therapy of the SCID-X1 condition T Lymphocytes development after gene therapy of SCID-X1 9000 8000 P4 7000 * 6000 T Lymphocytes/µl P8 5000 * P9 4000 3000 P2 P7 2000 P5 P6 1000 * P1 P10 0 0 10 20 30 40 Months after Gene Therapy 50 60 70 High proliferation rate enables T cell diversification from a few precursors Transduction 1 clone Multiple T cell clones 1 integration site, diverse TCR CLP gc-dependent proliferation Hematopoietic stem cell Myeloid cells TCR rearrangements As in the reversion case, oligoclonal lymphopoiesis polyclonal repertoire Monoclonal gd T cell proliferation P4 T Lymphocytes/µl 1000000 100000 10000 1000 100 P4 10 1 0 5 10 15 20 25 30 35 40 46, XY, 6q21;13qter • Monoclonal g d T cell clone Serious adverse effect caused by retroviral insertional mutagenesis in a protooncogene 44,218 MFG gc P5 46,229 MFG gc 1 P4 54,614 66,467 66,867 71,646 76,883 2 3 4 5 6 Enhancer activity LMO-2 mRNA 2. 3 kb Aberrant expression ---> clonal proliferation 1 2 3 4 5 1077 6 1513 T cell clone Clonal lymphoproliferation Synergistic effect with the gc transgene Conclusion Clinical benefit with sustained efficacy • 6 additional successfully treated patients in UK (A.Thrasher) extension to other SCID ? ADA deficiency, Rag-1, Rag-2, Artemis deficiencies… • • But… a serious adverse effect vector modifications Vector modifications LTR gc Promoter LTR IRES suicide gene U3 Self inactivated LTR loss of the enhancer effect Principles for research on rare diseases • Clinical centers of expertise in limited numbers - well studied cohorts of patients • International collaborative effort • Multidisciplinary approach • Impact - basic science . lymphocyte development - knowledge and therapy of more common diseases . pathophysiology of papilloma virus disease, . hematopoietic stem cell transplantation, . gene therapy - new drugs . treatment of lymphomas, prevention of graft rejection An European institute supporting research on rare diseases • To organize disease-reference centers (national/regional), in an european network • To build a network of platforms available for research projects on rare diseases (genomics,animal house, post genomics, molecular screening… ) • To foster collaboration with pharmaceutical companies. - example: the ERDITI initiative ERDITI: European Rare Diseases Therapeutic Initiative A public/private partnership To provide academic teams with an access to a variety of compounds from pharmaceutical companies - based on pathophysiological hypotheses To provide a collaboration process between academic teams and pharma partners Under the European Science Foundation Coordinated by GIS – Institut des maladies rares Partners and supporting institutions Pharmaceutical partners Supporting institutions Aventis Austria, Medical University Vienna Glaxosmithkline Belgium, FNRS - Fonds national de la Roche Servier Recherche Scientifique Croatia, Academy of Science ans Arts Denmark, Medical research council France, CNRS and Inserm Germany, DLR Project managment Organisations – Health research Netherlands, ZonMw, Steering committee on Orphan Drugs Spain, Rare diseases Institute (IIER) Slovakia, Academy of sciences A charter of collaboration • Partnership based on a charter of collaboration • The charter has 3 sections: - working procedure - standard material transfer agreement (MTA) - standard intellectual property rights agreement • Assessment by a scientific advisory board - analysis of the accuracy of the proposal - advices www.erditi.org www.institutmaladiesrares.net