Confirmatory Testing Considerations – SCID
advertisement

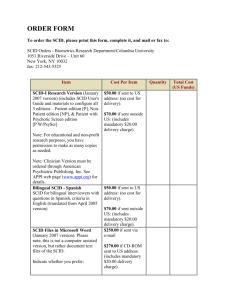
Confirmatory Testing Considerations BETH VOGEL, MS, CGC GENETIC COUNSELOR NEWBORN SCREENING PROGRAM WADSWORTH CENTER NEW YORK STATE DEPARTMENT OF HEALTH Overview Implementation Short-term follow-up Case review meetings Diagnostic testing Implementation: Guidance Partnership with clinicians New York State SCID Consortium 9 external group members National recommendations Newborn Screening Translational Research Network, CLSI Guidelines Implementation: Guidance Discussion points for NBS and clinician group Minimum standard confirmatory tests Short-term follow-up Diagnostic categories Transplants Algorithm changes Detection of non-SCID disorders Implementation: Education Target hospitals and primary care providers TREC assay is not well known! Developed educational materials Letter to hospitals Phone consultations NBS and Specialty Care Centers WHAT IS A TREC?! Short-term Follow-up Process Eight Specialty Care Centers Referral is called to primary care provider and specialty care center (SCC) Diagnostic form completed by clinician Case Review Meetings Identify topics for Consortium conference calls Unusual cases Identify need for new diagnostic categories Algorithm changes Correlation of laboratory findings and clinical outcomes Case Review Meetings Maintain consistency in case closure Incidence Algorithm changes Long-term follow-up Diagnostic Categories From NBSTRN website: 1. SCID 2. Leaky SCID/Omenn syndrome 3. Variant SCID 4. Syndromes with T cell impairment 5. Secondary T cell lymphopenia other than preterm alone 6. Preterm alone SCID Classic phenotype <300 autologous T cells/μL Emergent treatment required (transplant, gene therapy, enzyme replacement therapy) Leaky SCID/Omenn Syndrome Low T cell count 3oo to 1500 T cells/μL Requires treatment (transplant, gene therapy, enzyme replacement therapy) Omenn syndrome Erythrodermia, hepatosplenomegaly, eosinophilia, elevated IgE, restricted TCR diversity of T cells Variant SCID Moderately decreased T cell counts May not require transplant Syndromes with T Cell Impairment DiGeorge syndrome/22q11.2 deletion syndrome CHARGE syndrome Jacobsen syndrome RAC2 defect DOCK8 deficiency Ataxia telangiectasia VACTERL association Barth syndrome TAR syndrome Down syndrome Ectrodactyly ectodermal dysplasia syndrome Other Secondary T Cell Lymphopenia Other Than Preterm Alone Birth defects Gastroschisis, intestinal lymphangiectasia, congenital heart defects, cardiac surgery +/- thymectomy Metabolic disorder, degenerative neuromuscular disease Preterm Alone <37 weeks gestation TREC copy number typically improves over time If TRECs are undetectable, then full work-up warranted regardless of gestational age Idiopathic T-cell Lymphopenia Non-SCID No congenital anomalies Low T-cells that require clinical monitoring Diagnostic Testing: Complete Blood Count Lymphocytes T cells = 70% of circulating lymphocytes Proposed as a method to screen for SCID prior to current technologies “ALC (absolute lymphocyte count) <2500/uL in early infancy requires further evaluation” • R. Buckley, MD Diagnostic Testing: Flow Cytometry Evaluation of lymphocyte subpopulations Normal ranges vary by age Diagnostic Testing: Flow Cytometry CD = Cluster of differentiation Diagnostic Testing: Flow Cytometry CD Cell Type CD3 T cells CD4 Helper T cells CD8 Suppressor T cells CD19 B cells CD16CD56 NK cells Diagnostic Testing: Flow Cytometry Diagnostic Testing: Lymphocyte Proliferation to Mitogens Test of T-cell function Less sensitive, but more specific test of T-cell function Culture human peripheral blood mononuclear cells with plant lectin mitogens) phytohemagglutinin (PHA), pokeweed mitogen (PWM), concanavalin (conA) Response highest in newborns and decreases with age Diagnostic Testing: Mitogens Diagnostic Testing: Naïve and Memory T-cell Count Not typically included as part of standard flow cytometry CD45RA+ = Naive CD45RO+ = Memory Genetic Testing Multiple genes Mutation info often not needed before transplant May be important if diagnosis is unclear Important for genetic counseling 45% of SCID cases are X-linked (IL2RG-related) Remainder of the cases are recessive Diagnostic Form Diagnostic Form References Hicks MJ, Jones JK, Thies AC, et al: Age-related changes in mitogen-induced lymphocyte function from birth to old age. Am J Clin Pathol 1983;80:159-163 Baker M, Grossman W, Laessig R, et al. Development of a routine newborn screening protocol for severe combined immunodeficiency. J Allergy Clin Immunol. 2009; 124: 522-527 Puck M, et al. Population-based newborn screening for severe combined immunodeficiency: Steps toward implementation. J Allergy Clin Immunol. 2007; 120 (4): 760-768. Chan K, Puck J. Development of population-based newborn screening for severe combined immunodeficiency. J Allergy Clin Immunol. 2005; 115: 391-398. Comeau A, Hale J, Pai S, et al. Guidelines for implementation of population-based newborn screening for severe combined immunodeficiency. J Inherit Metab Dis. 2010 https://www.nbstrn.org/sites/default/files/SCID%20National%20Monthly%20March%20201 2.pdf Buckley RH. The long quest for neonatal screening for severe combined immunodeficiency. Clinical Reviews in Allergy and Immunology. 2012 Adeli MM and Buckley RH. Why newborn screening for severe combined immunodeficiency is essential: a case report. Pediatrics. 2010; 126(2):e465-e469. Chase NM, Verbsky JW, Routes JM. Newborn screening for SCID: three years of experience. Ann. N.Y. Acad. Sci. 2011; 1238: 99-105. Buckley RH, Transplantation of hematopoietic stem cells in human severe combined immunodeficiency: longterm outcomes. Immunol Res. 2011; 49: 25-43. Thank you QUESTIONS?