Bohr vs Electron Cloud
advertisement

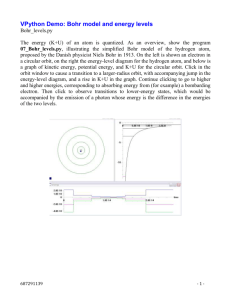
Bohr vs. Electron Cloud Where are we so far? • Students can: – Find the atomic number, the number of protons, neutrons, electrons, and atomic mass of an atom – Find the number of valance electrons – Draw a Bohr Model of the atom – Draw a Lewis Dot Diagram of an atom • Purpose – to find the number of bonding sites for atoms Date Bohr vs. Electron Cloud Models Objective: • Students will take notes and draw diagrams to explain the advantages and disadvantages of Bohr’s model and the Electron Cloud model of the atom. Page # Stable Atoms (Magic Number 8!) • Atoms with a full outer shell (8 e-) are stable – They don’t react or bond with any other element Ex: He = 2 valence electrons in the first shell Ne = 8 valence electrons in the second shell Ar = 8 valence electrons in the third shell These are the Noble Gasses! Niels Bohr’s Model (1913) • Electrons orbit the nucleus in circular paths of fixed energy (energy levels). The energy levels are like the rungs of a ladder but are not equally spaced. Energy levels. The energy levels in an atom are similar to the rungs of a ladder, but they get closer together as they get farther from the nucleus. Excited State and Ground State • Ground state: the lowest possible energy level an electron can be at. • Excited state: an energy level higher than the ground state. Niels Bohr’s Atom Cont’d • Electrons can jump from energy level to energy level. • Electrons ABSORB light energy when they jump from a lower energy level to a higher energy level. • Electrons EMIT light energy when they jump from a higher energy level to a lower energy level. (Draw this) E=hn Bohr Model for Hydrogen Hydrogen Emission Spectrum Violet Blue Red Balmer Series EVIDENCE: • The Bohr model explained the emission spectrum of the hydrogen atom ….. but was not able to explain the spectra of other elements. Successes and Problems of the Bohr Model • Introduced connection between spectra and electron structure • Concept of ‘allowed orbits’ or orbitals is developed further with new knowledge • Was an important contribution to the model of the atom - worthy of the Nobel prize • Couldn’t explain why orbits were allowed • Only successful agreement with experiment was with the H atom…not with any other elements The All New, Better Electron Cloud Model (Quantum Mechanical Model) A quantum of energy is the amount of energy required to move an electron from one energy level to another. Electron Cloud Model cont. • Electrons (e-) are located in specific energy levels. • There is no exact path for electrons around the nucleus… instead… • The model estimates the probability of finding an electron in a certain position… called an orbital or electron cloud Atomic Orbital / Electron Cloud A region in space in which there is high probability of finding an electron. Atomic Orbital / Electron Cloud (Draw this) Higher Probability for e- The higher the electron density, the higher the probability that an electron may be found in that region. http://www.chemeng.uiuc.edu/~alkgrp/mo/gk12/quantum/H_S_orbital.jpg Date Bohr vs. Electron Cloud Models Objective: • Students will take notes and draw diagrams to explain the advantages and disadvantages of Bohr’s model and the Electron Cloud model of the atom. Page #