the slides - ARV
advertisement

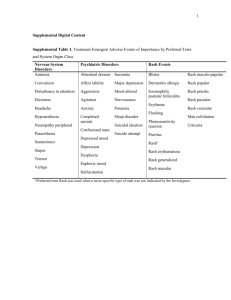
Switch to EVG/c/FTC/TDF STRATEGY-PI Study STRATEGY-NNRTI Study STRATEGY-PI Study: Switch PI/r to EVG/c Design HIV+ ≥ 18 years On FTC + TDF + PI/r HIV RNA < 50 c/mL > 6 months No virologic failure Genotype testing before ART with no resistance to study drugs Integrase inhibitor naïve eGFR > 70 mL/min Randomisation 2:1 Open-label N = 293 N = 145 W48 W96 Switch to EVG/c/FTC/TDF Continue PI/r + FTC + TDF Endpoints – Primary: proportion of patients maintaining HIV RNA < 50 c/mL at W48 (mITT, snapshot) ; non-inferiority if lower margin of a two-sided 95% CI for the difference = -12%, 85% power. If non-inferiority and lower margin > 0, assessment for superiority – Secondary: proportion of patients maintaining HIV RNA < 50 c/mL at W48 (TLOVR algorithm), CD4, safety, tolerability to W96 STRATEGY-PI Arribas J.R. Lancet Infect Dis 2014;14:581-9 STRATEGY-PI Study: Switch PI/r to EVG/c Baseline characteristics and patient disposition EVG/c/FTC/TDF N = 293 PI/r + FTC + TDF N = 140 41 40 15% 14% 4 4 77% 83% Atazanavir 42% 37% Darunavir 39% 43% Lopinavir 17% 16% Fosamprenavir 2% 4% Saquinavir 1% 0 564 585 Hepatitis B / hepatitis C coinfection 3% / 7% 2% / 7% Discontinuation by W48 25 (8.5%) 26 (18.6%) Median age, years Female Time since HIV diagnosis, median years On first ARV regimen PI at randomisation CD4 cell count (/mm3), median STRATEGY-PI Arribas J.R. Lancet Infect Dis 2014;14:581-9 STRATEGY-PI Study: Switch PI/r to EVG/c Virologic outcome at W48 (mITT, snapshot) EVG/c/FTC/TDF HIV RNA < 50 c/mL PI/r + FTC + TDF HIV RNA ≥ 50 c/mL No virologic data % 100 94 87 80 60 40 20 6 1 0 Difference (95% CI) = 6.7% (0.4 ; 13.7) STRATEGY-PI 12 1 N=2 N=2 Arribas J.R. Lancet Infect Dis 2014;14:581-9 STRATEGY-PI Study: Switch PI/r to EVG/c HIV RNA < 50 c/mL Sensitivity and secondary analysis Per-proctol EGV/c/FTC/TDF PI/r + FTC + TDF 99.5% 99.2% Difference: 0.1% (95% CI = - 2.1 ; 3.7) ITT-TLOVR 91.7% 84.2% Difference: 7.6% (95% CI = 0.9 ; 15.0) No participants met the criteria for resistance testing (HIV RNA > 400 c/mL at virologic failure or early discontinuation) STRATEGY-PI Arribas J.R. Lancet Infect Dis 2014;14:581-9 STRATEGY-PI Study: Switch PI/r to EVG/c Virologic sucess overall and by subgroup at W48 (mITT) n/N 272/290 121/139 122/130 61/68 150/160 60/71 231/247 103/120 41/43 18/19 217/231 98/113 53/57 22/24 114/121 41/51 107/113 55/60 45/49 20/23 213/225 104/115 53/59 16/23 Overall Age < 40 years Age > 40 years Male Female White Non-white Atazanavir Darunavir Lopinavir On first regimen at baseline On second regimen at baseline 0 STRATEGY-PI 10 20 30 40 50 60 70 80 90 100 Virological success (%) Switch group No-switch group -50 -40 -30 -20 -10 0 10 20 30 40 50 Difference (%) Favours not switching Favours switching Arribas J.R. Lancet Infect Dis 2014;14:581-9 STRATEGY-PI Study: Switch PI/r to EVG/c Adverse events and grade3-4 laboratory abnormalities EVG/c/FTC/TDF PI/r + FTC + TDF 79% 74% 4% 8% 6% 6% N = 6 (2%) N = 4 (3%) 0 N=1 14% 23% Gamma-GT > 5 x ULN 3% 1% CK ≥ 10 x ULN 2% 6% ALAT > 5 x ULN 2% 1% Haematuria 2% 1% 0 12% Any adverse event, Grade 3 or 4 AE Serious adverse event Discontinuation because of AE Death Any Grade 3 or 4 laboratory abnormality Bilirubin > 2.5 x ULN Improvement in lipids in the switch group HIV Symptom Index : rates of diarrhea and bloating decreased in the switch group Higher tretament satisfaction scores in the switch group STRATEGY-PI Arribas J.R. Lancet Infect Dis 2014;14:581-9 STRATEGY-PI Study: Switch PI/r to EVG/c Conclusion – Coformulated EVG/c/FTC/TDF is an effective, safe, and tolerable simplification from a PI/r plus FTC and TDF regimen in virologically suppressed, HIV-infected adults with no history of virological failure or resistance to FTC or TDF – Low frequency of virologic failure and absence of emergent resistance in the group switched to EVG/c/FTC/TDF – Rare discontinuations because of adverse events – Nausea more frequent in the switch group ; diarrhea and bloating improved – Small increase in creatinine, moderate improvement in lipids – EVG/c/FTC/TDF is a switch option in virologically suppressed patients with no history of virological failure who want to simplify their existing PI/r regimen, or who have concerns about the long-term safety and side-effects of their existing regimen STRATEGY-PI Arribas J.R. Lancet Infect Dis 2014;14:581-9