Chemistry: Matter and Change
advertisement

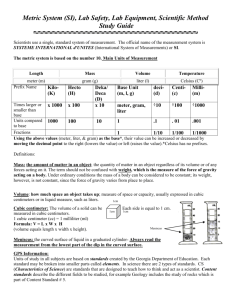
Analyzing Data Section 2.1 Units and Measurements Section 2.2 Scientific Notation and Dimensional Analysis Section 2.3 Uncertainty in Data Section 2.4 Representing Data Click a hyperlink or folder tab to view the corresponding slides. Exit Section 2.1 Units and Measurements • Define SI base units for time, length, mass, and temperature. • Explain how adding a prefix changes a unit. • Compare the derived units for volume and density. mass: a measurement that reflects the amount of matter an object contains Section 2.1 Units and Measurements base unit kelvin second derived unit meter liter kilogram density Chemists use an internationally recognized system of units to communicate their findings. (cont.) Standards of Measurement 2 Units and Standards • A standard is an exact quantity that people agree to use to compare measurements. Standards of Measurement 2 Measurement Systems • Suppose the label on a ball of string indicates that the length of the string is 150. • Is the length 150 feet, 150 m, or 150 cm? • For a measurement to make sense, it must include a number and a unit. Standards of Measurement 2 Measurement Systems • The English system of measurement is commonly used in the United States. • Most other nations use the metric system—a system of measurement based on multiples of ten. Standards of Measurement 2 International System of Units • All SI standards are universally accepted and understood by scientists throughout the world. Standards of Measurement 2 International System of Units • Each type of SI measurement has a base unit. • The meter is the base unit of length. Standards of Measurement 2 International System of Units • Every type of quantity measured in SI has a symbol for that unit. • All other SI units are obtained from these seven units. Standards of Measurement 2 SI Prefixes • The SI system is easy to use because it is based on multiples of ten. Standards of Measurement 2 SI Prefixes • Prefixes are used with the names of the units to indicate what multiple of ten should be used with the units. • The most frequently used prefixes are shown. Standards of Measurement 2 Converting Between SI Units • A conversion factor is a ratio that is equal to one and is used to change one unit to another. • For example, there are 1,000 mL in 1 L, so 1,000 mL = 1 L. Standards of Measurement 2 Converting Between SI Units • To convert units, you multiply by the appropriate conversion factor. • For example, to convert 1.255 L to mL, multiply 1.255 L by a conversion factor. Standards of Measurement 2 Converting Between SI Units • Use the conversion factor with new units (mL) in the numerator and the old units (L) in the denominator. Standards of Measurement 2 Measuring Distance • In scientific measurement length is the distance between two points. • The SI base unit of length is the meter, m. • Metric rulers and metersticks are used to measure length. Standards of Measurement 2 Choosing a Unit of Length • The size of the unit you measure with will depend on the size of the object being measured. • You probably would use the centimeter to measure the length of your pencil and the meter to measure the length of your classroom. Standards of Measurement 2 Choosing a Unit of Length • By choosing an appropriate unit, you avoid large-digit numbers and numbers with many decimal places. • Twenty-one kilometers is easier to deal with than 21,000 m. And 13 mm is easier to use than 0.013 m. Standards of Measurement 2 Measuring Volume • The amount of space occupied by an object is called its volume. • If you want to know the volume of a solid rectangle, such as a brick, you measure its length, width, and, height and multiply the three numbers and their units together (V = 1 x w x h). Standards of Measurement 2 Measuring Volume • For a brick, your measurements probably would be in centimeters. • The volume would then be expressed in cubic centimeters, cm3. Standards of Measurement 2 Measuring Liquid Volume • In measuring a liquid’s volume, you are indicating the capacity of the container that holds that amount of liquid. • The most common units for expressing liquid volumes are liters and milliliters. Standards of Measurement 2 Measuring Liquid Volume • A liter occupies the same volume as a cubic decimeter, dm3. • A cubic decimeter is a cube that is 1 dm, or 10cm, on each side. Standards of Measurement 2 Measuring Liquid Volume • Sometimes, liquid volumes such as doses of medicine are expressed in cubic centimeters. • Suppose you wanted to convert a measurement in liters to cubic centimeters. • You use conversion factors to convert L to mL and then mL to cm3. Units • Système Internationale d'Unités (SI) is an internationally agreed upon system of measurements. • A base unit is a defined unit in a system of measurement that is based on an object or event in the physical world, and is independent of other units. There are four main categories of measurement • Time • Length • Matter/mass • Temperature 25 Each category of measurement has a base unit • Time – second (s) • Length – meter (m) • Matter/mass – gram (g) • Temperature – Kelvin (K) 26 Units (cont.) • The SI base unit of temperature is the kelvin (K). • Zero kelvin is the point where there is no particle motion or kinetic energy, also known as absolute zero. • Two other temperature scales are Celsius and Fahrenheit. • Some measurements are considered to be derived units. For example, density is a mass to volume comparison and can be calculated using the following equation: • d = m/v 28 • Volume is considered to be a derived unit. Why? 29 Standards of Measurement 2 Measuring Volume • The amount of space occupied by an object is called its volume. • If you want to know the volume of a solid rectangle, such as a brick, you measure its length, width, and, height and multiply the three numbers and their units together (V = 1 x w x h). Section 2.1 Assessment Which of the following is a derived unit? A. yard B. second C. liter D C A 0% B D. kilogram A. A B. B C. C 0% 0% 0% D. D Section 2.1 Assessment What is the relationship between mass and volume called? A. density B. space D A 0% C D. weight A. A B. B C. C 0% 0% 0% D. D B C. matter Section 2.3 Assessment A substance has an accepted density of 2.00 g/L. You measured the density as 1.80 g/L. What is the percent error? A. 0.20 g/L A 0% D D. 0.90 g/L C C. 0.10 g/L A. A B. B C. C 0% 0% 0% D. D B B. –0.20 g/L