Combined Gas Law

Combined Gas Law
The combined gas law combines
Boyle’s Law and Charles’ Law
Combined Gas Law
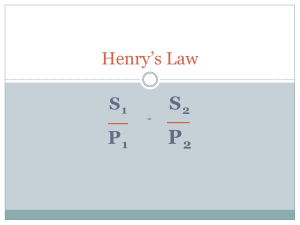
Boyle’s Law P
1
V
1
Charles’ Law
= P
V
1
T
1
=
V
T
2
2
Combined Gas Law
P
1
V
1
T
1
=
P
2
V
2
T
2
2
V
2
Combined Gas Law
For example: If I initially have a gas at a pressure of 12 atm, a volume of
23 L and a temperature of 200 K, then I raise the pressure to 14 atm and increase the temperature to 300
K, what is the new volume of the gas?
Combined Gas Law
Begin by converting to Kelvin. It is not necessary in this problem.
Combined Gas Law
Next, write down the information you know and want to know.
P
2
V
2
T
2
P
V
1
T
1
1
=
=
=
=
=
=
Combined Gas Laws
Next, write down the information you know and want to know.
P
2
V
2
T
2
P
V
1
T
1
1
= 12 atm
=
=
=
=
=
Combined Gas Law
Next, write down the information you know and want to know.
P
2
V
2
T
2
P
V
1
T
1
1
= 12 atm
= 23 L
=
=
=
=
Combined Gas Law
Next, write down the information you know and want to know.
P
2
V
2
T
2
P
V
1
T
1
1
= 12 atm
= 23 L
= 200K
=
=
=
Combined Gas Law
Next, write down the information you know and want to know.
P
2
V
2
T
2
P
V
1
T
1
1
= 12 atm
= 23 L
= 200K
= 14 atm
=
=
Combined Gas Law
Next, write down the information you know and want to know.
P
2
V
2
T
2
P
V
1
T
1
1
= 12 atm
= 23 L
= 200K
= 14 atm
= x
=
Combined Gas Law
Next, write down the information you know and want to know.
P
2
V
2
T
2
P
V
1
T
1
1
= 12 atm
= 23 L
= 200 K
= 14 atm
= x
= 300 K
Combined Gas Law
Now plug in the information you have . . .
P
1
V
1
T
1
=
P
2
V
2
T
2
12atm(23L)
200K
=
14 atm (x)
300K
Combined Gas Law
Cross multiply to solve for x
12atm(23 L)(300K) = 200 K (14L)(x)
82800 = 2800x x = 29.57 Liters