Ch04-alkanes-ques-12
advertisement

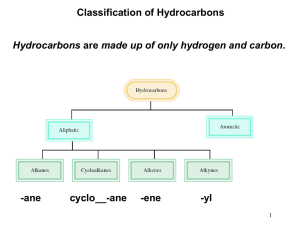
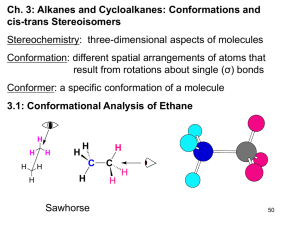
Chapter 4 Introduction to Hydrocabons Carbon Backbone, Nomenclature, Physical & Chemical Properties HYDROCARBONS • Compounds composed of only carbon and hydrogen atoms (C, H). Each carbon has 4 bonds. • They represent a “backbone” when other “heteroatoms” (O, N, S, .....) are substituted for H. (The heteroatoms give function to the molecule.) • Acyclic (without rings); Cyclic (with rings); Saturated: only carbon-carbon single bonds; Unsaturated: contains one or more carbon-carbon double and/or triple bond HYDROCARBONS • Alkanes contain only single ( ) bonds and have the generic molecular formula: [CnH2n+2] • Alkenes also contain double ( + ) bonds and have the generic molecular formula: [CnH2n] • Alkynes contain triple ( + 2) bonds and have the generic molecular formula: [CnH2n-2] • Aromatics are planar, ring structures with alternating single and double bonds: eg. C6H6 Types of Hydrocarbons Each C atom is tetrahedral with sp3 hybridized orbitals. They only have single bonds. Each C atom is trigonal planar with sp2 hybridized orbitals. There is no rotation about the C=C bond in alkenes. Question 4.1 • What is the hybridization of the starred carbon in humulene (shown)? • A) sp • B) sp2 • C) sp3 • D) 1s2 2s2 2p2 Question 4.2 • What is the hybridization of the starred carbon of geraniol? • A) sp • B) sp2 • C) sp3 • D) 1s2 2s2 2p2 Types of Hydrocarbons Each C atom is linear with sp hybridized orbitals. Each C--C bond is the same length; shorter than a C-C bond: longer than a C=C bond. The concept of resonance is used to explain this phenomena. Propane It is easy to rotate about the C-C bond in alkanes. Naming Alkanes C1 - C10 : the number of C atoms present in the chain. Each member C3 - C10 differs by one CH2 unit. This is called a homologous series. Methane to butane are gases at normal pressures. Pentane to decane are liquids at normal pressures. Nomenclature of Alkyl Substituents Examples of Alkyl Substituents Constitutional or structural isomers have the same molecular formula, but their atoms are linked differently. Naming has to account for them. Question 4.3 • How many hydrogens are in a molecule of isobutane? • A) 6 • B) 8 • C) 10 • D) 12 A compound can have more than one name, but a name must unambiguously specify only one compound C7H16 can be any one of the following: Question 4.4 • How many isomeric hexanes exist? • A) 2 • B) 3 • C) 5 • D) 6 Question 4.5 • The carbon skeleton shown at the bottom right accounts for 9 carbon atoms. How many other isomers of C10H22 that have 7 carbons in their longest continuous chain can be generated by adding a single carbon to various positions on this skeleton? • A) 2 • B) 3 • C) 4 • D) 5 Alkanes (Different types of sp3 carbon atoms) • Primary, 1o, a carbon atom with 3 hydrogen atoms: [R-CH3] • Secondary, 2o, a carbon atom with 2 hydrogen atoms: [R-CH2-R] • Tertiary, 3o, a carbon atom with 1 hydrogen atom: • [R-CH-R] R • Quaternary, 4o, a carbon atom with 0 hydrogen atoms: CR4 Different Kinds of sp3 Carbons and Hydrogens Question 4.6 • In 3-ethyl-2-methylpentane, carbon #3 (marked by a star) is classified as: • • • • A) B) C) D) primary (1°) secondary (2°) tertiary (3°) quaternary (4°) Question 4.7 • How many primary carbons are in the molecule shown at the bottom right? • A) 2 • B) 3 • C) 4 • D) 5 Nomenclature of Alkanes 1. Determine the number of carbons in the parent hydrocarbon 8 7 6 5 4 3 2 8 1 CH3CH2CH2CH2CHCH2CH2CH3 7 6 5 4 4 CH3CH2CH2CH2CHCH2CH3 CH3 2 2 1 CH3CH2CH2CHCH2CH2CH3 CH2CH2CH3 3 3 CH2CH2CH2CH3 1 5 6 7 8 2. Number the chain so that the substituent gets the lowest possible number 1 2 3 4 5 CH3CHCH2CH2CH3 CH3 2-methylpentane 1 2 3 4 5 6 7 8 CH3CH2CH2CHCH2CH2CH2CH3 CHCH3 CH3 4-isopropyloctane CH3 CH3CHCH2CH2CH3 common name: isohexane systematic name: 2-methylpentane 3. Number the substituents to yield the lowest possible number in the number of the compound CH3CH2CHCH2CHCH2CH2CH3 CH3 CH2CH3 5-ehtyl-3-methyloctane not 4-ethyl-6-methyloctane because 3<4 (substituents are listed in alphabetical order) 4. Assign the lowest possible numbers to all of the substituents CH3 CH3 CH3CH2CHCH2CHCH3 CH3 CH3 2,4-dimethylhexane CH3CH2CH2C CC H CH2CH3 H 3 2C CH3 CH3 3,3,4,4-tetramethylheptane CH3 CH3CH2CHCH2CH2CHCHCH2CH2CH3 CH2CH3 CH2CH3 3,3,6-triethyl-7-methyldecane 5. When both directions lead to the same lowest number for one of the substituents, the direction is chosen that gives the lowest possible number to one of the remaining substituents CH3 CH3 CH3CHCH2CHCH3 CH3 CH3 2,2,4-trimethylpentane not 2,4,4-trimethylpentane because 2<4 CH2CH3 CH3CH2CHCHCH2CHCH2CH3 CH3 6-ethyl-3,4-dimethyloctane not 3-ethyl-5,6-dimethyloctane because 4<5 6. If the same number is obtained in both directions, the first group receives the lowest number Cl CH2CH3 CH3CH2CHCH3 r B 2-bromo-3-chlorobutane not 3-bromo-2-chlorobutane CH3CH2CHCH2CHCH2CH3 CH3 3-ethyl-5-methylheptane not 5-ethyl-3-methylheptane 7. In the case of two hydrocarbon chains with the same number of carbons, choose the one with the most substituents 3 4 5 6 CH3CH2CHCH2CH2CH3 2 CHCH3 1 CH3 3-ethyl-2-methylhexane (two substituents) 1 2 3 4 5 6 CH3CH2CHCH2CH2CH3 CHCH3 CH3 3-isopropylhexane (one substituent) 8. Certain common nomenclatures are used in the IUPAC system CH3CH2CH2CH2CHCH2CH2CH3 CHCH3 CH3 4-isopropyloctane or 4-(1-methylethyl)octane CH3CH2CH2CH2CHCH2CH2CH2CH2CH3 CH2CHCH3 CH3 5-isobutyldecane or 5-(2-methylpropyl)decane Question 4.7 • The correct structure of 3-ethyl-2methylpentane is: • A) B) • C) D) CnH2n Cycloalkane Nomenclature Cycloalkanes • Cycloalkanes are alkanes that contain a ring of three or more carbons. • Count the number of carbons in the ring, and add the prefix cyclo to the IUPAC name of the unbranched alkane that has that number of carbons. Cyclopentane Cyclohexane Cycloalkanes • Name any alkyl groups on the ring in the usual way. A number is not needed for a single substituent. CH2CH3 Ethylcyclopentane Cycloalkanes • Name any alkyl groups on the ring in the usual way. A number is not needed for a single substituent. • List substituents in alphabetical order and count in the direction that gives the lowest numerical locant at the first point of difference. H3C CH3 CH2CH3 3-Ethyl-1,1-dimethylcyclohexane For more than two substituents, CH3 CH3CH2CH2 CH3 H 3C CH2CH3 4-ethyl-2-methyl-1-propylcyclohexane not 1-ethyl-3-methyl-4-propylcyclohexane because2<3 not 5-ethyl-1-methyl-2-propylcyclohexane because 4<5 CH3 1,1,2-trimethylcyclopentane not 1,2,2-trimethylcyclopentane because1<2 not 1,1,5-trimethylcyclopentane because 2<5 Question 4.8 • Which one contains the greatest number of tertiary carbons? • A) 2,2-dimethylpropane • B) 3-ethylpentane • C) sec-butylcyclohexane • D) 2,2,5-trimethylhexane Physical Properties of Alkanes and Cycloalkanes Naphtha (bp 95-150 °C) Kerosene (bp: 150-230 °C) C5-C12 Light gasoline (bp: 25-95 °C) C12-C15 Crude oil Gas oil (bp: 230-340 °C) Refinery gas C1-C4 C15-C25 Residue Question 4.9 Arrange octane, 2,2,3,3-tetramethylbutane and 2-methylheptane in order of increasing boiling point. • A) 2,2,3,3-tetramethylbutane < octane < 2-methylheptane • B) octane < 2-methylheptane < 2,2,3,3tetramethylbutane • C) 2,2,3,3-tetramethylbutane < 2-methylheptane < octane • D) 2-methylheptane < 2,2,3,3- tetramethylbutane < octane Crude Oil and Uses of Alkanes • The gasoline fraction of crude oil only makes up about 19%, which is not enough to meet demand. van der Waals Forces Weak Intermolecular Attractive Forces The boiling point of a compound increases with the increase in van der Waals force…and a Gecko uses them to walk! Gecko: toe, setae, spatulae 6000x Magnification Full et. al., Nature (2000) 5,000 setae / mm2 600x frictional force; 10-7 Newtons per seta Geim, Nature Materials (2003) Glue-free Adhesive 100 x 10 6 hairs/cm2 http://micro.magnet.fsu.edu/primer/java/electronmicroscopy/magnify1/index.html Intermolecular Forces Ion-Dipole Forces (40-600 kJ/mol) • Interaction between an ion and a dipole (e.g. NaOH and water = 44 kJ/mol) • Strongest of all intermolecular forces. Ion-Dipole & Dipole-Dipole Interactions: like dissolves like • Polar compounds dissolve in polar solvents & non-polar in non-polar Intermolecular Forces Dipole-Dipole Forces (permanent dipoles) 5-25 kJ/mol Intermolecular Forces Dipole-Dipole Forces Boiling Points & Hydrogen Bonding Hydrogen Bonding • Hydrogen bonds, a unique dipole-dipole (1040 kJ/mol). Intermolecular Forces London or Dispersion Forces • An instantaneous dipole can induce another dipole in an adjacent molecule (or atom). • The forces between instantaneous dipoles are called London or Dispersion forces ( 0.05-40 kJ/mol). Boiling Points of Alkanes • governed by strength of intermolecular attractive forces • alkanes are nonpolar, so dipole-dipole and dipole-induced dipole forces are absent • only forces of intermolecular attraction are induced dipole-induced dipole forces Boiling Points •Increase with increasing number of carbons • more atoms, more electrons, more opportunities for induced dipole-induced dipole forces •Decrease with chain branching • branched molecules are more compact with smaller surface area—fewer points of contact with other molecules Intermolecular Forces London Dispersion Forces Which has the higher attractive force? Question 4.10 • Which alkane has the highest boiling point? • A) hexane • B) 2,2-dimethylbutane • C) 2-methylpentane • D) 2,3-dimethylbutane Boiling Points •Increase with increasing number of carbons • more atoms, more electrons, more opportunities for induced dipole-induced dipole forces Heptane bp 98°C Octane bp 125°C Nonane bp 150°C Boiling Points •Decrease with chain branching • branched molecules are more compact with smaller surface area—fewer points of contact with other molecules Octane: bp 125°C 2-Methylheptane: bp 118°C 2,2,3,3-Tetramethylbutane: bp 107°C Sources and Uses of Alkanes • Gasoline is a mixture of straight, branched, and aromatic hydrocarbons (5–12 carbons in size). – Large alkanes can be broken down into smaller molecules by CRACKING. – Straight chain alkanes can be converted into branched alkanes and aromatic compounds through REFORMING. After using these processes, the yield of gasoline is about 47% rather than 19%. – Chemical Properties: Combustion of Alkanes •All alkanes burn in air to give carbon dioxide and water. Heats of Combustion Heptane 4817 kJ/mol 654 kJ/mol Octane 5471 kJ/mol 654 kJ/mol Nonane 6125 kJ/mol What pattern is noticed in this case? Heats of Combustion •Increase with increasing number of carbons • more moles of O2 consumed, more moles of CO2 and H2O formed Heats of Combustion 5471 kJ/mol 5 kJ/mol 5466 kJ/mol 8 kJ/mol 5458 kJ/mol 6 kJ/mol 5452 kJ/mol What pattern is noticed in this case? ENERGY Diagrams / Reaction Coordinate Diagrams 5471 kJ/mol 25 + O2 2 5466 kJ/mol 5458 kJ/mol + 25 O2 2 5452 kJ/mol + 8CO2 + 9H2O 25 O2 2 + 25 O2 2 Heat of Combustion Patterns •Increase with increasing number of carbons • more moles of O2 consumed, more moles of CO2 and H2O formed •Decrease with chain branching • branched molecules are more stable (have less potential energy) than their unbranched isomers Important Point •Isomers can differ in respect to their stability. •Equivalent statement: –Isomers differ in respect to their potential energy. Differences in potential energy can be measured by comparing heats of combustion. (Worksheet problems)