Chapter 2
advertisement

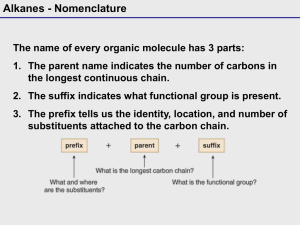
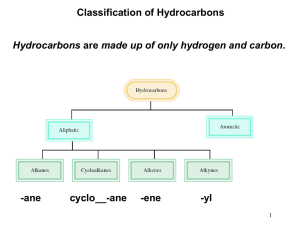
Chapter 2 ALKANES Classification of Hydrocarbons • Hydrocarbons contain only carbon and hydrogen. • A hydrocarbon that contains only single bonds is a saturated hydrocarbon or alkane (Chapter 2). • Representing Organic Compounds Molecular formula shows kind and number of atoms, not how they are arranged C4H10 Structural formulas show all atoms with bonds. Condensed formulas list all the atoms in order implying how they are bound together: CH3CH2CH2CH3 or CH3 (CH2) 2CH3 Ball and stick model Line –angle formula Each line represents a bond and each vertex and terminus represents a carbon atom Root Name Methethpropbutpenthexheptoctnondec- # of Carbons 1 2 3 4 5 6 7 8 9 10 CH3CH2CH2CH3 = BUTANE • Alkanes with 4 or more carbons can be straight chained (normal) or branched. C—C—C—C—C normal alkane C | C—C—C | C branched alkane •Constitutional Isomers: Compounds that have identical molecular formulas, but different arrangement of atoms (structural formulas). Molecular Formula C4H10 Constitutional Isomerism • Problem: draw structural formulas for the five constitutional isomers of molecular formula C6H14 2 1 6 4 3 5 S i x c arb on s i n an u n b ran ch e d ch ai n 4 1 2 3 2 5 1 4 3 4 2 5 Fi ve c arb on s i n a ch ai n ; o n e carb o n as a b ran ch 1 3 4 2 1 3 Fo u r carb o n s i n a ch ai n ; tw o carb o n s as b ran ch e s • Naming Alkanes – The IUPAC method consists of: • Alkyl group: a substituent group derived from an alkane by removal of a hydrogen atom. • commonly represented by the symbol R-. • named by dropping the -ane from the name of the parent alkane and adding the suffix -yl. N am e m e th y l C on d e n s e d S tru c tu ra l F o rm u l a - CH 3 e th y l - CH 2 C H 3 p ro p y l - CH 2 C H 2 C H 3 i s o p ro p y l N am e i s o b u ty l - CH s e c- b u ty l - CH CH - CH CH 3 CH CH 3 t er t - b u ty l b u ty l - CH 2 C H 2 C H 2 C H 3 C on S tru - CC CH Naming Alkanes • The ending –ane signifies the alkane family. Step 1: Identify and name the longest carbon chain. This gives the root and ending. Step 2: Number the longest carbon chain to give the lowest number to any carbon to which a group is attached(substituent). CH3 CH3 | | Example: CH2 — CH2 — CH — CH3 5 CH CH3 | | Example: CH2 — CH2 — CH — CH3 (pentane) 3 4 3 2 1 Step 3: Locate and name the attached alkyl group. Naming Alkanes, cont. Step 4: Combine the longest chain and the branches into the name. Use a hyphen to connect the number to the name. 5 CH CH3 | | CH2 — CH2 — CH — CH3 3 Example: 4 3 1 2-methylpentane CH 3 CHCH 3 2 4 5 3 2 1 • Draw structural, condensed and line-angle formula of 3-ethylhexane IUPAC Names 5. If the same substituent occurs more than once: • Number the parent chain from the end that gives the lower number to the substituent encountered first. • Indicate the number of times the substituent occurs by a prefix di-, tri-, tetra-. • Use a comma to separate position numbers. CH 3 CH 3 CH 3 CH 2 CHCH 2 CH CH 3 6 4 5 2 3 2,4-D i m e th y l h e xan e (n ot 3,5-d im e th y l h e xan e ) 1 IUPAC Names 6. If there are two or more different substituents: • list them in alphabetical order. • number the chain from the end that gives the lower number to the substituent encountered first. • If there are different substituents in equivalent positions on opposite ends of the parent chain, give the substituent of lower alphabetical order the lower number. CH 3 CH 3 CH 2 CHCH 2 CHCH 2 CH 3 2 1 4 3 CH 2 CH 3 3-E th y l -5-m e th y l h e p tan e (n o t 3-m e th y l -5-e th y l h e p tan e ) 6 5 7 IUPAC Names 7. Do not include the prefixes di-, tri-, tetra- in alphabetizing; • alphabetize the names of substituents first, and then insert these prefixes CH 3 CH 2 CH 3 CH 3 CCH 2 CHCH 2 CH 3 1 6 4 2 3 5 CH 3 4-Eth yl -2,2-d im e th y l h e xan e (n ot 2,2-d im e th y l -4-e th y l h e xan e ) Cycloalkanes • Cyclic hydrocarbon: a hydrocarbon that contains carbon atoms joined to form a ring. • Cycloalkane: a cyclic hydrocarbon in which all carbons of the ring are saturated. • Cycloalkanes of ring sizes ranging from 3 to over 30 carbon atoms are found in nature. • Five-membered (cyclopentane) and six-membered (cyclohexane) rings are especially abundant in nature. Cy cl o p e n tan e C yc l oh e xan e Cycloalkanes • Nomenclature • To name a cycloalkane, prefix the name of the corresponding open-chain alkane with cyclo-, and name each substituent on the ring. • If there is only one substituent on the ring, there is no need to give it a location number. • If there are two or more substituents, number the ring beginning with the substituent of lower alphabetical order. Example: CH2CH3 1 CH3 3 2 1-ethyl-3-methylcyclopentane Physical Properties • The most important physical property of alkanes and cycloalkanes is their almost complete lack of polarity. – The electronegativity difference between carbon and hydrogen is 2.5 - 2.1 = 0.4 on the Pauling scale. – Given this small difference, we classify a C-H bond as nonpolar covalent. – Alkanes are nonpolar compounds and have only very weak interaction between molecules. Methane molecules Oil molecules Physical Properties • Melting and boiling points – Boiling points of alkanes are lower than those of almost any other type of compound of the same molecular weight. – In general, both boiling and melting points of alkanes increase with increasing molecular weight. Physical Properties • Solubility: a case of “like dissolves like”. – Alkanes are not soluble in water; they are unable to form hydrogen bonds with water. – Alkanes are soluble in each other. – Alkanes are also soluble in other nonpolar organic compounds. Alkane Reactions • Alkanes are the least reactive of all organic compounds. • The most significant reaction of alkanes is combustion. • Combustion- A chemical reaction that produces CO2, H2O and a flame due to burning with oxygen. • Many alkanes are used this way – as fuels. Methane – natural gas Propane – used in gas grills Butane – lighters Gasoline – a mixture of hydrocarbons Complete Combustion (in the presence of adequate Oxygen) CH 4 + 2 O2 CO 2 + 2 H 2 O + 212 k c al /m ol M e th an e CH 3 CH 2 CH 3 + 5 O 2 Prop an e 3 CO 2 + 4 H 2 O + 530 k cal / m o l Incomplete Combustion (where there is not enough Oxygen available) Reactions • Reaction with halogens (halogenation) • Halogenation of an alkane is a substitution reaction. heat or light C H 4 + C l2 Methane CH 3 Cl + Cl2 CH3 Cl + HCl Chloromethane (Methyl chloride) h e at CH 2 Cl2 + HCl D i c h l o ro m e th an e (M e th y l e n e ch l o ri d e ) Cl2 CH 2 Cl2 Cl2 CHCl3 CCl4 h e at h e at T ri ch l o rom e th an e T e trach l o rom e th an e (C h l o ro f orm ) (Carb o n te trach l o ri d e ) The Chlorofluorocarbons • Chlorofluorocarbons (CFCs) • manufactured under the trade name Freon • CFCs are nontoxic, nonflammable, odorless, and noncorrosive. • Among the CFCs most widely used were CCl3F (Freon11) and CCl2F2 (Freon-12). • CFCs were used as; • heat-transfer agents in refrigeration systems. • industrial cleaning solvents to prepare surfaces for coatings and to remove cutting oils from millings. • propellants for aerosol sprays.