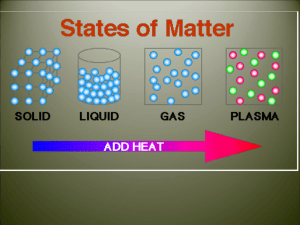
States of Matter
Phase Change
Science 6
Water
• Water exists on our
planet in three states.
• Ice, water, and water
vapor
• What causes water to
be in one phase or
another?
ENERGY
• When energy is
added to a substance
that energy causes
the particles in the
substance to move
faster and farther
apart.
• What happens to the
particles when energy
is taken away from a
substance?
Phase Change
• Energy content is responsible for the
different phases of matter.
• Matter can be made to change phase
when energy is added to or taken away
from a substance.
DO NOW
Page 2 in notebook
• Observe the ice cube.
• Describe everything you see happening.
• Why is this happening – be specific.
– Use knowledge you already have to explain
this process!
Melting- Solid to Liquid
• Melting is the
changing of a solid to
a liquid when the
substance absorbs
heat energy.
• Melting Point
– Water
0° C.
32° F
– Table salt 801° C.
1474 ° F
– Diamond 3550° C.
6422 ° F
click
Melting Point?
• What is a melting point?
Melting Point?
• What is a melting point?
• It is the precise temperature at which a
particular substance melts changing from
a solid to a liquid when the substance
absorbs heat energy.
• !
Phase Change Animation
Phase Change Diagram
If something melts, then it can freeze!
Freezing- Liquid to Solid
• Opposite of melting:
liquid changing to a
solid is freezing.
• Freezing occurs when
a substance loses
heat energy.
• The freezing point of
a substance is equal
to the melting point!
Water Molecules Freezing
• click
Questions
• What is a Freezing Point?
• What might someone find interesting
about the melting point and freezing point
of a particular substance?
Questions
• What is a Freezing Point?
• It is the precise temperature at which a
particular substance freezes!
• What might someone find interesting
about the melting point and freezing point
of a particular substance?
• They are the same temperature!
More Questions
• What is melting?
– Turning from a _______________ to a
______________ due to _________ of energy.
• What is freezing?
– Turning from a _______________ to a
______________ due to __________ of
energy.
More Questions
• What is melting?
– Turning from a ____solid_________ to a
______liquid____ due to _gain__ of energy.
• What is freezing?
– Turning from a ___liquid___ to a
__solid__ due to _loss_ of energy.
Vaporization- Liquid to Gas
• Vaporization is the
changing of a liquid to
a gas when the
substance absorbs
heat energy.
• Vaporization
occurring at the
surface of a liquid is
called evaporation.
The Water Cycle
Vaporization a.k.a. Boiling
• If enough heat energy
is applied to a
substance particles
inside the liquid can
change to gas.
• These particles travel
to the surface of the
liquid and then into
the air. This process
is called boiling.
Boiling Point
• Boiling Point –
temperature at which
a substance boils.
– Water
100° C.
212° F.
– Table salt 1413° C.
2575° F.
– Diamond
4827° C.
8720° F.
Water Phase Change Graph
What is the difference in the amount of energy need to melt and to boil?
MORE Questions
• What is the difference between
evaporation and boiling?
MORE Questions
• What is the difference between
evaporation and boiling?
– Evaporation happens only at the surface
– Examples – lake, ocean, perspiration
– Boiling, or vaporization, is when the particles
within a liquid take in enough heat (energy) to
become a gas. “Rolling boil”
Condensation- Gas to Liquid
• Gases can change
phase also - in a gas
to liquid phase
change.
• A substance in the
gas phase that loses
heat will change to a
liquid. This is called
condensation.
Condensation
• Water vapor in surrounding air loses heat energy when it
comes in contact with the cold glass. Water vapor
condenses and becomes liquid drops of water.
Condensation in the Water Cycle
THINK!!!
If water vapor
in the air must
condense to
form a cloud,
then a cloud is
made of
____________?
Yes, More Questions!
• What is condensation?
– It is when a gas ______________ energy and
changes to a ____________________.
• Think of examples in your life other than
the cold drink in the summer that shows
condensation.
Yes, More Questions!
• What is condensation?
– It is when a gas ______ energy and
changes to a __________.
• Think of examples in your life other than
the cold drink in the summer that shows
condensation.
• Steamy bathroom after a hot shower – “fog”
on mirror!
Phase Change Graph
Sublimation – Solid to Gas
• Solid to gas phase
change occurs when
a substance gains so
much energy it is able
to go directly from a
solid to a gas,
omitting the liquid
phase!
Sublimation: Solid to Gas
• Ice will sublimate in your freezer! The
molecules on the surface of the ice will
gain enough energy to turn into a gas.
• You may notice this in the cold winter
with snow. The snow does not melt, but
slowly disappears.
Deposition: Gas to Solid
• When water vapor
in air loses so
much energy it
turns directly into a
solid.
• Frost!
Questions – Just a couple…
• Sublimation occurs when a substance
__________ energy and goes directly
from a _____________ to a __________.
• Deposition occurs when a substance
__________ energy and goes directly
from a _____________ to a __________.
Questions – Just a couple…
• Sublimation occurs when a substance
__gains energy and goes directly from a
_solid to a _gas.
• Deposition occurs when a substance
_loses energy and goes directly from a
gas_ to a solid.
Energy in Phase Change
DEPOSITION
Deposition: Gas to Solid
• When water vapor
in air loses so
much energy it
turns directly into a
solid.
• Frost!
Add the correct terms to the
diagram…
Temperature Comparisons
• Boiling Point of H2O
212 F
100 C
• Melting Point of H2O
32 F
0 C
• Freezing Point of H2O
32 F
0 C
Your Assignment…
• Grass Garden Teams - Pick a number
• In numerical order choose from the following:
melting
freezing
vaporization
condensation
sublimation
deposition
Your TASK
Communicate to the rest of the class what your
phase change is. You may make a poster,
present a newscast, create a jingle, or produce
a skit.
See Assignment Sheet for details.
Phase Change Graph
Phase Change Graph
 0
0