1
Outline
•
•
•
•
Define terms and conventions
Introduce 1st law of thermodynamics
Contrast state and non-state properties
Describe the Carnot cycle
2
System and environment
• System = what we wish to study
– View as control mass or control volume
• Control mass (CM)
– Define some mass, hold fixed, follow it around
• Control volume (CV)
– Define and monitor a physical space
• Environment = everything else that may
interact with the system
3
System states
• Systems may be open or closed to mass
– Open systems permit mass exchange across system
boundaries
– Our CVs are usually open
– Strictly speaking, a CM is closed
• Closed systems may be isolated or nonisolated
– Isolated systems do not permit energy transfer with
environment
– Closed, isolated system = environment doesn’t matter
4
Lagrangian vs. Eulerian
• CM is the Lagrangian viewpoint
– Powerful, desirable but often impractical
– Total derivatives
– Freeway example
• CV is the Eulerian viewpoint
– Observe flow through volume
– Partial derivatives
5
Air parcel
• Our most frequently used system
• CM (usually!) – Lagrangian concept
• Monitor how T, p, and V change as we follow
it around
6
Conventions
• We often use CAPITAL letters for extensive
quantities, and lower case for specific quantities
– Specific = per unit mass
• Example:
– U is internal energy, in Joules
– u is specific internal energy, in J/kg
– Unfortunately, “u” is also zonal wind velocity
• Exceptions:
– Temperature T is essentially specific, but capitalized
(and isn’t per unit mass anyway)
– Pressure p is fundamentally extensive, but lower case
7
Energy and the 1st law
• Total energy = KE + PE + IE
– Conserved in absence of sources and sinks
• Our main use of 1st law: monitor changes in
internal energy (IE or u) owing to sources and
sinks
• How do we change system u? With energy
transfer via
– heat Q or q
– work W or w
• Caveat: w is also vertical velocity, and q will be
reused (briefly) for water vapor specific humidity
8
Work
• Work = force applied over a distance
– Force: N, distance: m
– Work: Nm = J = energy
• Our principal interest: CM volume
compression or expansion (dV) in presence of
external pressure (p)
• W > 0 if dV > 0
9
Work
W > 0 when system expands against
environment
10
Heat
• Diabatic heat
– Diabatic: Greek for “passable, to be passed
through”
– Internal energy exchanged between system and
environment
– q > 0 when energy flow is INTO system
• Adiabatic = system is isolated
– Adiabatic: impassable, not to be passed through
11
Caution on nomenclature
• We should use diabatic when the energy exchange is
between system and environment
• But, what if the heat source or sink is inside the system?
– That’s adiabatic, but q ≠ 0
– Our interior heat source will be water changing phase
• Dry adiabatic: q = 0
– No heat source, outside OR inside
– “dry” really means no water phase changes
• Moist adiabatic: q ≠ 0, but heat source/sink is inside system
– “moist” implies water phase change
– Synonyms include “saturated adiabatic” and “wet adiabatic”
– Can also be referred to as “diabatic”!
12
1st law
• In the absence of ∆KE and ∆PE
• Other ways of writing this
Most of my examples will be per unit mass.
13
State properties
• Internal energy u is a state property
• Changes in state properties are not pathdependent
• Other state properties include m, T, p, r, V,
etc.
14
State properties
15
Path-dependence
• Work and heat are path-dependent
16
Path-dependence
• A cyclic process starts
and ends with the same
state property values
• … but the cyclic process
can have net heat
exchange and do net
work
17
Path-dependence
18
Path-dependence
19
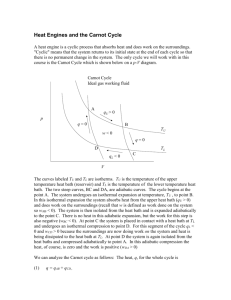
Carnot cycle
• 4-step piston cycle on a CM
• 2 steps of volume expansion, 2 of volume
compression
• 2 steps are isothermal, 2 are (dry) adiabatic
• Warm and cold thermal reservoirs external to
system
• Start and end with temperature T1 and
volume V1
20
Carnot – Step 1
Isothermal volume expansion
Add heat QA from warm
reservoir
T2 = T1
V2 > V1
21
Carnot – Step 2
Adiabatic volume expansion
No heat exchange
T3 < T2
V3 > V2
22
Carnot – Step 3
Isothermal volume compression
Lose heat QB to cold thermal
reservoir
T4 = T3
V4 < V3
23
Carnot – Step 4
Adiabatic volume compression
No heat exchange
T1 > T4
V1 < V4
Returned to original state T1, V1.
Cycle is complete.
24
25
Apply 1st law
26
Carnot on T-V diagram
27
Carnot on T-V diagram
28
Carnot on T-V diagram
29
Carnot on T-V diagram
30
Carnot on T-V diagram
31
Carnot on T-V diagram
32
Carnot on T-V diagram
No net ∆V
But did net W
33
Conceptual summary
Heat flow diverted
to do work
34
Question for thought #1
The isothermal expansion (QA) occurred at a
higher temperature than the
Isothermal compression (QB).
What does this imply for the work?
QB is waste heat.
What does this imply for the
efficiency of this heat engine?
Is there a limit to efficiency?
Is the limit found in the 1st law?
35
Question for thought #2
Can you design a cyclic process that does no net work?
What would it look like on a T-V diagram?
36
Summary
• 1st law says, in essence, if you can’t take the
heat, you can’t do the work
• Work and heat are path-dependent
• Carnot cycle illustrates isothermal and (dry)
adiabatic processes
– Heat diverted to do work, but some is wasted
W = Q A - QB
37
 0
0