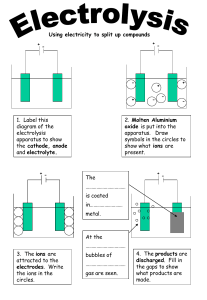
During the charging of a lithium-ion (Li-ion) cell, several electrochemical processes take place within the cell's components. A lithium-ion cell typically consists of three primary components: a positive electrode (cathode), a negative electrode (anode), and an electrolyte. Here's a simplified overview of the key processes that occur during the charging of a lithium-ion cell: 1. **Ion Movement:** During charging, an external voltage is applied to the cell, creating an electric field. Lithium ions in the positive electrode (cathode) move through the electrolyte towards the negative electrode (anode). 2. **Ion Insertion:** At the negative electrode (anode), lithium ions are inserted or intercalated into the host material of the anode, typically made of graphite. This process is known as lithiation. 3. **Electron Flow:** Simultaneously, electrons are released at the anode due to the chemical reactions, and they flow through the external circuit, providing electrical energy that can be used to power devices or stored for later use. 4. **Ion Flow Through Electrolyte:** Lithium ions move through the electrolyte from the anode to the cathode. 5. **Ion Insertion at Cathode:** At the positive electrode (cathode), typically composed of lithium metal oxide, lithium ions are deintercalated or extracted from the host material. This process is known as delithiation. 6. **Electron Reabsorption:** Electrons from the external circuit re-enter the cell at the cathode, completing the electrical circuit. 7. **Storage of Energy:** The energy required to separate lithium ions and electrons during charging is stored in the chemical bonds of the anode and cathode materials. It's important to note that these processes are reversible, allowing lithium-ion cells to undergo charging and discharging cycles. During discharging, the opposite reactions occur, with lithium ions moving from the anode to the cathode through the electrolyte, releasing stored energy. It's crucial to follow proper charging protocols, as lithium-ion batteries can be sensitive to overcharging or charging at too high a current, which can lead to safety issues, including overheating and potential battery failure. Advanced battery management systems are often incorporated into devices containing lithium-ion batteries to monitor and control the charging process for safety and optimal performance. Overcharging a lithium-ion (Li-ion) cell can have serious consequences and may lead to safety hazards. Here are some of the potential issues that can arise when a lithium-ion cell is overcharged: 1. **Lithium Plating:** Overcharging can cause the deposition of metallic lithium on the cell's anode instead of inserting lithium ions into the anode material. This process is known as lithium plating and can lead to the formation of dendrites, which are small, needle-like structures. These dendrites can pierce the separator between the anode and cathode, causing internal short circuits. 2. **Increased Heat Generation:** Overcharging increases the internal heat generated within the cell. Excessive heat can accelerate chemical reactions, degrade the electrolyte, and compromise the stability of the cell, leading to thermal runaway. 3. **Thermal Runaway:** Thermal runaway is a self-sustaining and uncontrolled increase in temperature within the cell. It can result from overcharging or other factors that cause excessive heat generation. During thermal runaway, the cell's temperature rises rapidly, potentially leading to the release of flammable gases, electrolyte leakage, and, in extreme cases, combustion or explosion. 4. **Loss of Capacity and Lifespan:** Prolonged overcharging can lead to a loss of the cell's capacity and overall lifespan. The excessive stress on the cell's components, especially the electrolyte, can cause chemical changes that degrade the performance of the battery over time. 5. **Voltage Stress:** Overcharging can subject the cell to higher voltage levels than it is designed to handle. This can result in voltage stress, leading to the breakdown of materials within the cell and compromising its structural integrity. To prevent overcharging and ensure the safe operation of lithium-ion cells, charging systems often incorporate protection mechanisms. These mechanisms may include voltage regulation, current limiting, and temperature monitoring. Battery management systems (BMS) are commonly used to monitor and control the charging process, protecting the cell from overcharging, over-discharging, and other potential issues. It's important to follow the manufacturer's guidelines and recommendations for charging lithiumion batteries to prevent overcharging and ensure the safe and efficient use of electronic devices. EV BIKE CHARGERS EV CAR CHARGERs