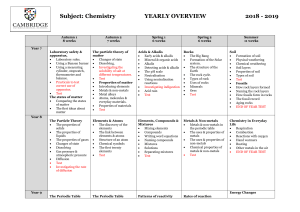
Metals & Non-Metals: Chemical Properties Lab Experiment
advertisement

Experiment: Investigating the Chemical Properties of Metals and Non-Metals Materials: Safety glasses, gloves and laboratory coat Samples of non-metals such as carbon and sulfur Samples of metals such as zinc, iron, aluminium, copper and tin Hammer and board Power pack Alligator clips Quartz stone Iron Nail Method: 1. Examine the metals and non-metals. Note any similarities and differences in their appearance. Record your observations. 2. Prior to performing each of the following tests predict what you think will happen. 3. Place a small piece of each substance you are testing on a board, one at a time, and hit with a hammer. Does it bend or shatter? 4. Try scratching each substance with the sharp point of an iron nail and then with a piece of quartz. Record which substances are scratched by each. 5. Set up the electric circuit as shown below. Check the circuit is working by starting with copper metal, the ammeter should produce a reading. 6. Replace the copper metal with the other substances and record the reading on the ammeter. Questions 1. What did you find out about the electrical conductivity of metals compared with non-metals? 2. Can you think of a property exhibited by some metals but not by others? Give examples to support your answer. 3. Which substances were hard? 4. State an everyday application of each of the following properties: a) Good electrical conductor b) Non-conductor of electricity c) Malleable d) Hard 5. How did your observations compare to your predictions? 6. Are there any generalisations that you can make about metals and non-metals based on this experiment?