Chapter 2 Lecture 2 Alkanes
I.
II.
Functional Groups
A.
Hydrocarbons: organic molecules containing only C and H
1) Alkanes: saturated hydrocarbons
a) Saturated = all single bonds
b) Unsaturated = one or more double or triple bonds
2) Alkenes: hydrocarbons with one or more double bonds
3) Alkynes: hydrocarbons with one or more triple bonds
4) Aromatics: hydrocarbons containing
B.
Other functional groups
1) Table 2-1 p. 54/55
2) Become familiar with these
Alkanes
A.
Structure
1) Straight-chain (normal) Alkanes
n-hexane
2)
Branched Alkanes
a)
b)
3)
Cycloalkanes
a)
b)
4)
CH4
5)
Straight-chain and Branched alkanes are constitutional isomers
CnH2n+2 = general formula
General formula = CnH2n
Not constitutional isomers with straight or branched
Homologous Series: group of molecules differing only by a -CH2- group
CH3CH3
CH3CH2CH3
CH3CH2CH2CH3 CH3CH2CH2CH2CH3
Alkyl Group: fragment that is an alkane missing one H
1) R (radical or Residue) stands for any Alkyl Group
2) Methyl
Ethyl
CH3
CH2CH3
CH3(CH2)nCH3
B.
Nomenclature: systematic naming by IUPAC rules
1) Straight Chain Alkanes
a) C1 = Methane C2 = Ethane
C3 = Propane
b) C5-C10 = pent-, hex-, hept-, oct-, non-, decc) C11-C19 = un-, do-, tri-, tetra-, penta-….nona
d) C20 = Icosane
2)
C4 = Butane
-ane
-decane
Branched Alkanes
a) Primary(1o) Secondary(2o) Tertiary(3o) Quaternary(4o)
Tell how many other carbon atoms are connected to the subject carbon
3o
1o
4o
2o
b) Substituents = groups attached to the main chain of an alkane
c) Rules for naming Branched Alkanes (or drawing structure from name)
1. Find the longest chain and name it as a straight chain alkane
2. Name substituents as alkyl groups
3. Number the main chain starting from the end closest to a substituent
4. Write the name
i. Alphabetize by sub. (di-, tri-count only if part of sub. name)
ii. Order #’s from low to high; use smallest possible numbers
iii. Capitalize the first letter only
iv. Write as one word with commas and hyphens as needed
v. Complex substituents in parentheses
1
1
5-Ethyl-2,2-dimethyloctane
1’
4-ethyl-3,6-dimethyldecane
1
1
5-(1,1-Dimethylethyl)-3-ethyloctane
4-(1-Ethylpropyl)-2,3-dimethylnonane
C.
Properties of Alkanes
1) Structure
a) Very regular because bonds (C-C, C-H) are all sp3
H H
b) Each C is tetrahedral
H
H
H
c) n-pentane
H
2)
HH H H H
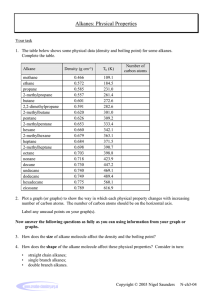
Physical Properties
a) Very predictable along homologous series
b)
Forces between Alkane Molecules
i. Ionic Solids: Ionic Forces (Coulombic) Na+Clii. Polar Solids: Dipole-Dipole Forces CH3Cl
iii. Nonpolar Solids: London Forces = weak attractive force due
to electron cloud correlation
c)
Melting Points of Alkanes
i. Very low, mostly liquids at room temperature
ii. Increases with size (more area for London Forces)
d)
Boiling Points of Alkanes: similar trends as m.p.
e)
Branched Alkanes have lower m.p., b.p than linear alkanes: less
surface area means less London Forces
D.
Conformations of Alkanes
1) C-C Bonds can freely rotate (~3 kcal/mol energy barrier due to e- repul.)
2)
Newman Projections
3)
Potential Energy Diagrams for Rotation of Alkane Conformations
a) Torsional Angle = degrees of rotation in a conformational change
b) Transition State = energy maxima; transition from one lower energy
state to another
c) Activation Energy = energy barrier to be overcome in changing
conformations
4)
Conformational Analysis of Substituted Ethanes
5)
Steric Hindrance = 2 fragments can’t occupy same space at same time
6) Butane: anti and gauche conformations
 0
0