AP Chemistry Equation Writing on the AP Test
advertisement

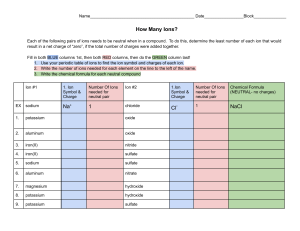
AP Chemistry Equation Writing on the AP Test Equation Writing is a very important part of chemistry. It is important to not only be able to predict the products and balance an equation, but to also know something qualitative about the reaction in question. This is the basis for equation writing on the AP Test, and will be the basis for many of the equation writing assignments in this class. It is our goal this year to not only be able to write and balance equations, but to be able to answer a question about the reaction. Some of the questions you will already know, some you will not. Some you might have to research. Either way, this is a required question on the AP Test and a task we will undertake this year. This is the format for the mandatory equation writing problem on the AP Test: For each of the following reactions, in part (i) write a BALANCED equation and in part (ii) answer the question about the reaction. In part (i), coefficients should be in terms of lowest whole numbers. (Assume that solutions are aqueous unless otherwise indicated. Represent substances in solutions as ions if the substances are extensively ionized. Omit formulas for any ions or molecules that are unchanged by the reaction.) Example: A strip of magnesium is added to a solution of silver nitrate. (i) Mg + 2 Ag+ → Mg2+ + 2 Ag (ii) Which substance is oxidized in the reaction? Answer: Magnesium (Mg) metal There are many possible questions that could have been asked about the above equation, such as… Which substance is being oxidized? Which substance is being reduced? What is the change in the oxidation number of the magnesium? What would you observe happening to the magnesium metal strip in this reaction? Any one of these questions -- and you may be able to come up with more -- is representative of what question 4 looks like. Below are more examples of question 4 on the AP Test. Look at the format. Examine the reactions. Get a feel on what is being asked. As the year progresses, you will gather more knowledge of reactions and the question will get easier. So remember… Part (i) asks for the balanced chemical equation, omitting any ions or molecules that are unchanged in the reaction; part (ii) asks one question that focuses on such things as the type of reaction (redox, proton-transfer, Lewis acid-base, and so forth), the stoichiometry of the reaction, and the physical observations that one might expect as the reaction occurs. Table 2: Question 4 Reactions - Examples (the answers are in bold) Reaction Question 1. Solid potassium chlorate is strongly heated and decomposes, resulting in a change in the oxidation numbers of both chlorine and oxygen. 1. What is the oxidation number of chlorine before and after the reaction? 2KClO3 → 2KCl + 3O2 2. Solid silver chloride is added to a solution of concentrated hydrochloric acid, forming a complex ion. AgCl + Cl− → [AgCl2]− 3. A solution of ethanoic (acetic) acid is added to a solution of barium hydroxide. HC2H3O2 + OH− → H2O + C2H3O2− 4. Ammonia gas is bubbled into a solution of hydrofluoric acid. NH3 + HF → NH4+ + F− 5. Zinc metal is placed in a solution of copper(II) sulfate. Zn + Cu2+ → Zn2+ + Cu Chlorine has an oxidation number of +5 in KClO3 and -1 in KCl. 2. Which species acts as a Lewis base in the reaction? Explain. The chloride ion acts as a Lewis base in the reaction because it donates an electron pair. 3. Explain why a mixture of equal volumes of equimolar solutions of ethanoic acid and barium hydroxide is basic. In the mixture there are initially twice as many moles of hydroxide ions as molecules of acid; since they react in a 1:1 ratio, there is an excess of hydroxide ions after the reaction is complete, leading to the basic solution. 4. Identify a conjugate acid-base pair in the reaction. NH3 (base) and NH4+ (acid) Or: HF (acid) and F− (base) 5. Describe the change in color of the solution that occurs as the reaction proceeds. The blue color of the solution due to the presence of the hydrated copper(II) ion fades as the copper(II) ion reacts and the colorless hydrated zinc(II) ion forms. 6. Hydrogen phosphide (phosphine) gas is added to boron trichloride gas. (i) PH3 + BCl3 → H3PBCl3 7. A solution of nickel(II) bromide is added to a solution of potassium hydroxide. (i) Ni2+ + 2 OH− → Ni(OH)2 6. Which species acts as a Lewis acid in the reaction? Explain. BCl3 acts as a Lewis acid in the reaction because it accepts the non-bonded pair of electrons of the phosphorus atom in PH3. 7. Identify the spectator ions in the reaction mixture. The spectator ions are the bromide ion (Br−) and the potassium ion (K+).