Chem 121 Worksheet – Ch. 4.1 – 4.5
advertisement

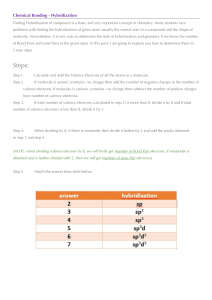
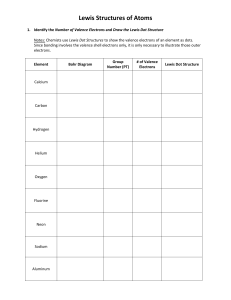
Chem 121 Worksheet – Ch. 4.1 – 4.5 You may work with a partner and use your textbook. This worksheet will not be graded, but you may want to write out all your work for practice and future reference. 1. Fill out the tables below. For each element, fill in the number of valence electrons each has, the number of bonds it will tend to form (if obeying the octet rule), and the number of lone pair electrons it will have. Element # of valence electrons number of bonds oxygen phosphorus carbon iodine 2. Draw Lewis structures for the following molecules. a. CHCl3 b. CO2 c. N2 d. C3H8 e. CH4O f. CH2O number of lone pairs 3. Each molecule below has more than one valid Lewis structure. Draw them. a. N2O b. CO3- c. SCN- 4. Give the name for each of the formulas below. a. H2O b. CCl4 c. P2S5 d. SF6