Chap 5B1.pptx
advertisement

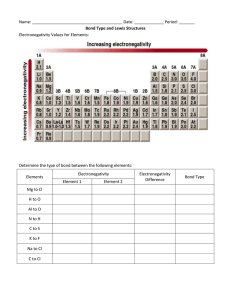
CHAPTER OUTLINE Electronegativity Polarity & Electronegativity Lewis Structures Molecular Shapes 1 ELECTRONEGATIVITY Electronegativity Linus Pauling derived (E.N.)a is relative the ability of an atom involved in aScale covalent bond to attract Electronegativity based on Bond Energies. the bonding electrons to itself. Cs F 0.7 4.0 Least electronegative Most electronegative 2 ELECTRONEGATIVITY 3 BOND POLARITY & ELECTRONEGATIVITY Polarity is a measure of the inequality in the sharing of bonding electrons The more different the electronegativity of the elements forming the bond The larger the electronegativity difference (EN) The more polar the bond formed 4 POLARITY & ELECTRONEGATIVITY As difference in electronegativity increases Least polar Most polar Bond polarity increases 5 POLARITY & ELECTRONEGATIVITY Electronegativity difference Bond Type EN = 0 Non-polar covalent 0 < EN <1.7 Polar covalent 1.7 < EN Ionic 6 POLARITY & ELECTRONEGATIVITY The molecule is nonpolar covalent Electronegativity 2.20 H H EN = 0 Electronegativity 2.20 Hydrogen Molecule 7 POLARITY & ELECTRONEGATIVITY The molecule is polar covalent + - H Electronegativity 2.20 Cl EN = 0.96 Electronegativity 3.16 Hydrogen Chloride Molecule 8 POLARITY & ELECTRONEGATIVITY No molecule exists The bond is ionic Na+ Electronegativity 0.93 ClEN = 2.23 Electronegativity 3.16 Sodium Chloride 9 SUMMARY OF BONDING EN > 1.7 Ionic Bond (large EN) Non-polar (similar electronegativities) EN = 0 Polar (moderate EN) Covalent Bond (small to moderate EN) 0 < EN < 1.7 10 COMPARING PROPERTIES OF IONIC & COVALENT COMPOUNDS Ionic Covalent Structural Unit Ions Molecules Melting Point High Low Boiling Point High Low Solubility in H2O High Low or None Electrical Cond. Examples High NaCl, AgBr None H2, H2O 11 LEWIS STRUCTURES Lewis In Lewis structures symbols symbols, foruse valence theLewis firstelectrons 3symbols periodsfor toofshow each valence electrons representative element are shown elements inas molecules a dot. are shown and ions below: of compounds. 12 LEWIS STRUCTURES In a Lewis structure, a shared electron pair is indicated by two dots between the atoms, or by a dash connecting them. Unshared pairs of valence electrons (called lone pairs) are shown as belonging to individual atoms or ions. 13 LEWIS STRUCTURES Writing correct Lewis structures for covalent compounds requires an understanding of the number of bonds normally formed by common nonmetals. 14 LEWIS STRUCTURES Structures Covalent molecules must satisfy are octet best represented rule (8 electrons with around electron-dot each or atom). Lewis structures. Hydrogen is one of the few exceptions and forms a doublet (2 electrons). 15 LEWIS STRUCTURES Bonding Non-bonding electrons electrons can be must displayed be displayed by a dashed as dots. line. 16 LEWIS STRUCTURES More complex Lewis structures can be drawn by following a stepwise method: 1. Count the number of electrons in the structure. 2. Draw a skeleton structure. 3. Connect atoms by bonds (dashes or dots). 4. Distribute electrons to achieve Octet rule. 5. Form multiple bonds if necessary. 17 Example 1: Write Lewis structure for H2O Step 1: H2O = 8 electrons Step 2: H Step 3: Step 4: 2 (1) + 6 = 8 O H Skeleton Hydrogen structure has doublet 4 electrons used Octet rule is satisfied should be 4 electrons remaining symmetrical 18 Example 2: Write Lewis structure for CO2 Step 1: CO2 = 16 electrons 4 + 2(6) = 16 Step 2: Step 3: Step 4: Step 5: O C O Skeleton structure Octet 10 4 electrons rule is NOT used Octet rule is satisfied should be 6 electrons 12satisfied electrons remaining symmetrical remaining 19 Example 3: Write Lewis structure for CO32Step 1: CO32- = 24 electrons 4+3(6)+2 = 24 Step 2: O O Step 5: C Step 4: O Step 3: 12 18 06 electrons electrons remaining Octet Octetrule ruleisissatisfied NOT satisfied 20 Example 4: Write Lewis structure for NH3 Step 1: NH3 = 8 electrons Step 2: Step 3: Step 4: 5 + 3(1) = 8 H N H H Octet rule is satisfied 21 Example 5: Write Lewis structure for ClO3 Step 1: ClO3 = 26 electrons 7+3(6)+1 = 26 Step 2: Cl O O 14 20 028 electrons electrons remaining Step 4: O Step 3: Octet rule is satisfied 22 EVALUATING LEWIS STRUCTURES When evaluating Lewis structures for correctness, two points must be considered: 1. Are the correct number of electrons present in the structure? 2. Is octet rule satisfied for all elements? (Hydrogen is an exception) 23 Example 1: Determine if each of the following Lewis structures are correct or incorrect. If incorrect, rewrite the correct structure. 2(1)Octet + 4 + is 6 = 12 incomplete Octet is complete Doubletsisare Structure complete incorrect 24 Example 2: Determine if each of the following Lewis structures are correct or incorrect. If incorrect, rewrite the correct structure. Structure has 14 electrons Only+124(1) electrons 2(5) = 14 shown 2 4 2 2 2 Structure is incorrect Octets are complete 25 MOLECULAR SHAPES The A very three-dimensional simple model , VSEPR shape of(Valence the molecules Shell is an important Electron Pair Repulsion) feature inTheory, understanding has beentheir properties by developed andchemists interactions. to predict the shape of molecules basedhave on their Lewis large All binary molecules a linear shape since structures. they only contain two atoms. More complex molecules can have various shapes (linear, bent, etc.) and need to be predicted based on their Lewis structures. 26 MOLECULAR SHAPES Based on VSEPR, the electron pair groups in a molecule will repel one another and seek to minimize their repulsion by arranging themselves around the central atom as far apart as possible. Electron pair groups can be defined as any one of the following: bonding pairs non-bonding pairs multiple bonds 27 SUMMARY OF VSEPR SHAPES Number of electron pair groups around central atom Molecular Shape Bond Angle Example s Bonding Non-bonding 2 0 Linear 180 CO2 3 0 Trigonal planar 120 BF3 2 1 Bent 120 SO2 4 0 Tetrahedral 109.5 CH4 3 1 Pyramidal 109.5 NH3 2 2 Bent 109.5 H2O 28