Ch 8a
advertisement

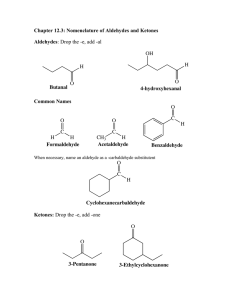
ALDEHYDES INTRODUCTION • Aldehydes are organic compounds which have a carbonyl functional group, C=O with at least one hydrogen 3,4- dimethyl pentanal 2- chloro propanal 2- ethyl pentanal 3- hydroxy butanal SYNTHESIS OF ALDEHYDES • Oxidation of Alcohols: Aldehydes can be prepared by oxidation of alcohols. O ǁ CH3 – CH2 – OH [O] CH3 – C – H • Acyl Chloride reduction: Aldehydes can be prepared by reduction of acyl chloride. O O ǁ ǁ [H O] R – C – Cl R–C–H 2 PHYSICAL PROPERTIES • Inter molecular forces in aldehydes are stronger than alkanes and ethers but weaker than alcohols. • Aldehydes do not have hydrogen bonding but can make hydrogen bonds with water. • Aldehydes with five or fewer carbon atoms are fairly soluble in water. Large aldehydes dissolve in non polar solvents. • The boiling points of aldehydes increases with increase in the molecular size due to increase in the inter molecular forces. REACTIONS OF ALDEHYDES (Chemical Properties) • OXIDATION: Aldehydes can be oxidized to carboxylic acids. • REDUCTION: Aldehydes can be reduced to primary alcohols. Uses Of aldehydes • Aldehydes are used for many things such as perfumes and flavorings. Other uses of Aldehydes include disinfectants and dying items. Aldehyde is also used in Bake Lite.