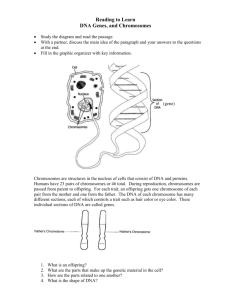
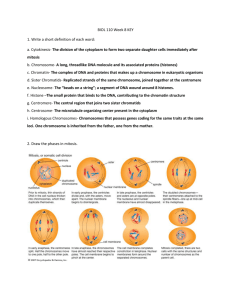
Cytological and molecular investigations in Lens and Pisum
advertisement

Cytological and molecular investigations in Lens and Pisum
by Jason Gordon Walling
A thesis submitted in partial fulfillment of the requirements for the degree of Master of Science in Plant
Sciences
Montana State University
© Copyright by Jason Gordon Walling (2003)
Abstract:
Three separate investigations involving genetic characterization of the pea (Pisum sativum) and/or
lentil (Lens culinaris) genome were performed.
The first experiment, evaluated the hypothesis of conserved gene synteny between lentil and pea
species. Sixty-two segregating P. sativum anchor markers (STS, isozyme, RFLP, and morphological)
and two lentil protein markers were tested within a lentil RIL population. Ten linkage groups were
assembled that span over 300 centimorgans. The majority of the loci evaluated share linkage synteny
with pea; however some notable genetic rearrangements were revealed. Supplementary to this study,
data supported the presence of two reciprocal translocations that differ in each parent. In addition,
approximately 25% displayed distorted segregation ratios.
The second experiment characterized and evaluated a lx Bacterial Artificial Chromosome (BAC)
library constructed from P. sativum. The results suggested that approximately 1.0% of the clones
contain chloroplast DNA sequence while approximately 0.35% would contain sequences homologous
to the 45s rDNA array. The presence of lower copy sequences, such as gene family constituents and
unique sequences were also evaluated. One clone, of the gene family small subunit of ribulose
bisphosphate carboxylase (Rubisco), was identified from the library. One low copy gene, glutamine
synthetase, was also identified within the BAC library. Inconsistent results obtained during the in-clone
verification process are discussed.
The final experiment used the two partially characterized BACs obtained from the library screening to
assess their use as in situ probes using Fluorescent In Situ Hybridization (FISH) on P. sativum
chromosomes. rDNA probes as well as repetitive DNA probes were integrated into the study. The
results verified the physical position of the NOR and 5s loci in both pea and lentil. Furthermore, a BAC
containing the sequence for the Rubisco small subunit hybridized to three regions. However, the BAC
containing the glutamine synthetase sequence did not hybridize to a specific location, possibly because
the C0t-1 blocking did not provide sufficient inhibition to cross-hybridization. Results indicated that
when using current protocols, positioning BACs on pea chromosomes is difficult, and improvements
are needed before such an approach can be routinely integrated into an experimental design. CYTOLOGICAL AND MOLECULAR INVESTIGATIONS IN LENS AND PISUM
by
Jason Gordon Walling
I
A thesis submitted in partial fulfillment
■of the requirements for the degree
^
of
Master of Science
in
Plant Sciences
MONTANA STATE UNIVERSITY
Bozeman, Montana
April 2003
N sie
Wiss
ii
APPROVAL
of a thesis submitted by
Jason Gordon Walling
This thesis has been read by each member of the thesis committee and has been
found to be satisfactory regarding content, English usage, format, citations, bibliographic
style, and consistency, and is ready for submission to the College of Graduate Studies.
Lfrlht
Dr. Norman F. Weeden
Signature)
Date
Approved for the Department of Plant Sciences and Plant Pathology
Dr. Norman F. Weeden
J((Signature)
Date
Approved for the College of Graduate Studies
s / ~ o 5
Dr. Bruce R. McLec
(Signature)
Date
STATEMENT OF PERMISSION TO USE
In presenting this thesis in partial fulfillment of the requirements for a master: s
degree at Montana State University, I agree that the Library shall make it available ,to,
borrowers under rules of the Library. . .
If I have indicated my intention to copyright this thesis by including a copyright
notice page, copying is allowable only for scholarly purposes, consistent with “fair use”
as prescribed in the U.S. Copyright Law. Requests for permission for extended quotation
from or reproduction of this thesis in whole or in parts may be .granted only by the
copyright holder.
Signature
Date
^
.
.iv
TABLE OF CONTENTS
1. THESIS INTRODUCTION.......... ................... ...........................................
2
Legumes.............. .........
2
■ p a w (Field pea) ................ ...... ..................; ;....... ...................................... 4
5
Zsms1(Lentil) .......................................
Literature Cited .............. !....... ................................
9
2. SCREENING OF A lx COVERAGE PISUMBACTERIAL ARTIFICIAL
CHROMOSOME (BAC) LIBRARY....:.............. ..... ................................. ....... 21
Literature review .......... ............. ..... ............................................
21
Introduction..................
21
DNA Libraries ............................... .:............ ............................................21
Bacterial Artificial Chromosomes .........................................
13
BAC Applications.....................................................................................
Materials and Methods..................................................,............................... 20
Probe Construction...,.........................
20
Probe Labeling and Hybridization..................... .............................. ;...... 24
Stringency Washes and Autoradiography of BAC Filters.......................26
Hybridization Signal Scoring.........;......................................................... 27
Clone Characterization and Sequence Analysis..... .:................................28
31
R esults ...............................................;......................................................'
Chloroplast Contamination Screen........................................................... 31
Ribosomal Screen..................... .............................................■.................32
Gene Family Screen....................
32
Low/Single Copy Screen..... .......................;............................................ 35
Discussion................. ......... :........ ;................ ................. ;....................... ......... 40
Literature cited ............'...,.... ...........'.............................................. ■
.................45
3. BAC FLUORESCENCE TASITU HYBRIDIZATION ON PISUM SA TIVUM
CHROMOSOMES....:..... ,............................................. ..................................... 49
■ Literature Review .......................... . .........................................;.......... ........... 49
Introduction-Cytology in Pisum.....................................
49
Cytological markers.............................
50
In Situ Sequence Detection:.........................
52
In Situ Sequence Detection in Pisum......................................... ........
57
BAC-FISH..........................................................................;................... '59.
TABLE OF CONTENTS -CONTINUED
Materials and Methods....•..............................................
Harvesting and Treatment of Pisum Root Tips for Cytological
Preparations.........................................................................
^2
Chromosome Pretreatment.............................................
54
Chromosome Isolation......................................................
Isolation and Nick Translation of BACs............................ .
67
Cyt-I Blocking DNA Isolation................................... ............................. gg
Fluorescence In Situ Hybridization.............................................. ;...
59
Secondary Antibody Detection.....................................;....;.................... j g
Fluorescence Microscopy.............. .................................
73
Results :...................... ................................................................. ’ ................... 75
Accumulation of Metaphase Chromosomes...............
....75
rDNA FISH on Pisum Metaphase Chromosomes....................................76
Repetitive Clone FISH................... ..................................
gg
BAC-FISH Gene Family....................
g2
BAC-FISH Single Copy...........................................................;...
g4
Discussion ................... .............. .................................................... .
.......; ‘‘§7
Literature Cited ..... ..................... ............................................
.
......95
4. COMPARATIVE MAPPING BETWEEN LENTIL (LENS SPP.)
AND PEA (PISUMSATIVUM)............................................................................ 99
Literature Review ................................................................ ........... .
. 99
Introduction.....................
99
Comparative Genetic Mapping.......................................
99
Sequence Tagged Sites (STS) Markers..................:............................... 102
Genetic Mapping in Lentil................................................... .................. iq 3
Linkage Conservation Among Temperate Legumes..... .......:.................105
M aterials and Methods ............. .................................... .............................. IQg
Mapping Population...................
IOg
Primer Characteristics................ ........... ............................................... 209
STS Analysis..............
ilO
Cleaved Amplified Polymorphic Sequence (CAPS)
Analysis.................. ................ .................-. ........................................
210
Genetic Mapping of Segregating Alleles.................... ........................... 111
Results ....................... ...................... ..................................................,
....113
D iscussion..................................................................................
"”223
Characterization of cross Lc #7 x Le #32 ....................... ...................... 223
Conserved Gene Synteny....................................................
225
Discrepancies in Lentil Genetic Mapping.........................
126
Literature Cited ...............................................................................
228
--------------------------------------
.
■ —
■
-
'
'
- I I - Ll
Ml
IM
vi
TABLE OF CONTENTS-CONTINUED
5. EXPERIMENTAL SUMMARY..............................;................. ..............
il31
APPENDICES.......... ...... r............ .........................................;.......................... I35
Appendix A: Raw BAC insert sequence ....................... .............................. 137
Appendix B: Raw marker segregation data from
CROSS LC #7 x L e #32......................................... ;........................ ..................
......I 40
1U
,l_,
;■
V ll
LIST OF TABLES
Tab,e
1. Characteristics of BAG Screening Probes Used to Screen
' Library..... ........................................ ......................................
Page
2]
2. Summary of BAC Screening Results.:.,............................................................... 34
3. STS PCR Primer and Product Characteristics....................... .................'...........114
■ 4. Segregation of Markers.From Cross
L. cuiinaris x L. ervoides...................... .............................................................j jg
5. Conserved Linkages Between Pea and Lentil..... ............. ........................ .
121
V lll
LIST OF FIGURES
piSure
Page
1. Phylogentic Relationship Among the Tribes Vicieae, Trifolieae,
and Cicereae.......................................................................... ;
3
2. Trnl-C and trnl-D Gradient PCR Product............................................................ 32
3. A) cpDNA BAC Filter Autoradiography........... ..... ............................................ 33
B) 2SsrDNA BAC Filter Autoradiography..................................................... ...33
4. A) RbcS BAC Filter Autoradiography
....................................................... .34
B) Lb BAC Filter Autoradiography........................................................ ;.......... 34
5. A) Drr49 BAC Filter Autoradiography.................. ..................................... .... ;.. 3g
B) Drr49 BAC Filter Autoradiography (shortened exposure).’....... ....... ............ 36
6. Pool #1 BAC Filter Autoradiography;.......... ................................................... ...,33
7. Merging Monochrome. Images into Colors Channels......................................... 74
8. A) Pisum sativum Metaphase Chromosomes ...................................................... 76
B) Pisum sativum Metaphase Chromosomes............. ........................................ 76
9. A) FISH using NOR probe (metaphase) ...................... ....................................... 77
B) FISH using NOR probe (metaphase)......... :.................................................... 77
C) FISH using NOR probe (metaphase)......................... ..................................... 77
D) FISH using NOR probe (prometphase)...... .... ................................................77
10. FISH on Interphase Nuclei using 5s and NOR rDNA Probes ..... ................ ;
79
11. A) FISH on Lens culinaris Chromosomes using 5s and NOR
Probes (60x)................................................... ...... ...................................;.......... 80.
B) FISH on Lens culinaris Chromosomes using 5s and NOR
Probes (IOOx)................................................................ ...................................... 80
12. A) FISH using Probe PisTR-B............. .................. .................................... ....... 81
B) FISH using Probe PisTR-B....................;............ ........................................... gl
13. A) FISH using BAC 103-Kl (RbcS), Low Blocking........................................... 83
B) FISH using BAC 103-KI (RbcS), High Blocking.......................... ................ 83
LIST OF FIGURES-continued
14. A) FISH using BAG 89-E10 (Gsc), Low blocking
(prometaphase)............ ................................................... ................................ §5
B) FISH using BAC 89-E10 (Gsc), Low blocking
(interphase).........................................................................................................
•C) FISH using BAC 89-E10 (Gsc), Low blocking
(metaphase)
■..................... ................ ...........' ........................ . g5
15. FISH using BAC 89-E10 (Gsc), High Blocking............... ............................ ...... 86
16. Linear Lentil Linlcage Groups.............................................................. ........... ...115
17. A) Interchange I/II Translocation Complex............................ ;..... ;............... .116
B) Interchange V/VI Translocation Complex................................... ...;....... . 116
18. PCR Products Generated using CipPor primers............................................... 119
19. Consensus Pea Map Displaying Areas of Conserved Synteny
with Lentil......................................................................................................... 12O
ABSTRACT
Three separate investigations involving genetic characterization of the pea (Pisum
sativum) and/or lentil (Lens culinaris) genome were performed.
The first experiment, evaluated the hypothesis of conserved gene synteny between
lentil and pea species. Sixty-two segregating P. sativum anchor markers (STS, isozyme,
RFLP, and morphological) and two lentil protein markers were tested within a lentil RIL
population. Ten linkage groups were assembled that span over 300 centimorgans. The
majority of the loci evaluated share linlcage synteny with pea; however some notable
genetic rearrangements were revealed. Supplementary to this study, data supported the
presence of two reciprocal translocations that differ in each parent. In addition,
approximately 25% displayed distorted segregation ratios.
The second experiment characterized and evaluated a lx Bacterial Artificial
Chromosome (BAC) library constructed from P. sativum. The results suggested that
approximately 1.0% of the clones contain chloroplast DNA sequence while
approximately 0.35% would contain sequences homologous to the.45s rDNA array. The
presence of lower copy sequences, such as gene family constituents and unique
sequences were also evaluated. One clone, of the gene family small subunit of ribulose
bisphosphate carboxylase (Rubisco), was identified from the library. One low copy gene,
glutamine synthetase, was also identified within the BAC library. Inconsistent results
obtained during the in-clone verification process are discussed.
The final experiment used the two partially characterized BACs obtained from the
library screening to assess their use as in situ probes using Fluorescent In Situ
Hybridization (FISH) on P. sativum chromosomes. rDNA probes as well as repetitive
DNA probes were integrated into the study. The results verified the physical position of
the NOR and 5s loci in both pea and lentil. Furthermore, a BAC containing the sequence
for the Rubisco small subunit hybridized to three regions. However, the BAC containing .
the glutamine synthetase sequence did not hybridize to a specific location, possibly
because the Cgt-I blocking did not provide sufficient inhibition to cross-hybridization.
Results indicated that when using current protocols, positioning BACs on pea
chromosomes is difficult, and improvements are needed before such an approach can be
routinely integrated into an experimental design.
I
CHAPTER I
THESIS INTRODUCTION
Legumes
The legume family (Fabaceae) is the third largest family of flowering plants next
to the orchid family (Orchidaceae) and the sunflower family (Asteraceae), respectively
(Stern, 1997). The Fabaceae contains approximately 350 genera composed of 18,000
species of considerable genetic diversity that vary in both climatic and geographical
distribution. Legumes are distributed throughout the world, from the tropics to arctic and
alpine habitats.
There are several common characteristics that support the family of legumes.
Most legumes possess the typical fruit morphology represented by a pod that encloses the
seed(s), which when dry will split along two sutures facilitating seed dispersal. The word
legume originates from the latin term legumen which literally means ‘anything that can
be gathered’, presumably referring to the grouping or gathering of seeds within the pod.
The general flower morphology has been described as being either radial or bilateral
(asymmetrical) (Stern, 1997). The asymmetrical flower, specifically the morphological
features of the banner, keel, and wing-petals, are arguably the most recognizable features
of the subfamily Papilionoideae. One common characteristic shared among most species
of legumes is the symbiotic relationship shared with the bacteria Rhizobium, which
facilitates the fixation of atmospheric N2 to ammonia (NHs) or other nitrogenous bases.
The ability of legumes to fix nitrogen promotes their use in crop rotations and as a green
manure (Campbell, 2002). The economic value represented by this family of legumes, is
2
surpassed only by that of the grasses (Poaceae) (Hymowitz, 1990) . However, it has been
argued that in terms of the number of species and various uses of legumes such as green
manure, fuel, forage and feed crops, cover crops, chemicals and timber; members of the
Fabaceae are probably more widely used by humans than any other family of plants
(Hymowitz, 1990).
Within the Fabaceae, members of the subfamily Papilionoideae, specifically the
tribes Vicieae (Pisum, Lens, Vicia) Phaseoleae (Phaseolus, Vigna), and Cicereae (Cicer)
represent what have been referred to as the primary grain legumes (Hymowitz, 1990).
The distinction between primary, secondary, and tertiary grain legumes is based, on
,
greater production numbers within the primary grain over constituents of the secondary
and tertiary grain legumes (Hymowitz, 1990). Other commonly !mown members of the
subfamily Papilioniodeae are the tribes lupin (Lupinus), clover (Trifolieae), and peanut
(Arachis).
Members of the tribe Viceae such as Pisum sativum L. (field pea), Lens culinaris
(lentil), Viciafaba (fababean), andLathyrus sativus (grass pea) are commonly labeled as.
cool season legumes, referring to the species preference of temperate/cool growth
conditions. This tribe is thought to have originated along the Fertile Crescent. The
domestication of the primary legumes was concurrent with the domestication of the
cereal grains (Zohary et ah, 1993). The phylogenetic relationship among these three
tribes within the subfamily Papilionoideae is described in Figure I .
3
100
Cicer canariense,
Qicermacraeanll
[icerpinn
uega on
y j tiy r u s a y ,^ ^
100
Gicereae
Steele et al. 2003
p M E E S " =iiI= >
IMthyrus Iingitanus
Pisiim satmiin
i,ens culinpns
. ^enf ervoides
^
100
;iSto yfIfosaensis (section Cracca
,'i.cifl ludoviciana*\
100
\tjflifl flSiciffala section Ervoules
YieiflMrsuta section Cracca
■89
ai&UumWtlK
99
100U K
100
North American
Trifoliiim
’ r^pfulm willdei
Trifplium worms,
too
100
99
Vicieae
j subgenus Mt/,
100
100
clade 2
'icip amencana*
100
riM iiimbi------------Piielliunum
rifplium cherangiense
n p liu m semipifosum
rifplium hybndum
rifplium spumoSpm
rifplium eaucasiciim
fifplm m Iflrtum
rifplium tnearnatum section Trifolium
,nfpliUm patulum
100
96
m fplium subterraneum
Trifpliumlupniaster .
TriMium diibiuin section Chwnosemmm
Wefflcafo ferac^crtrpo | sect*on Tunatae
Medipago hyppgaea
Medicago IalTugera
80
= I g C B I s e c t I o n f/orycaowe
88
•
•
■
Medipago heymana ,
Medicaga pdlymorpha
Medipago italica ,
Meclicagb truneatula
'
• Seiicafo orSlcularis
88
■Medipagb prostrata
■ Midipago
■ Mechpago
melieaginoides
monanmp
section Biiceras
■Meehpago monspehaca
■Meelipago pplyperatici
99
Trifolieae
'P
B lX m
Mdilotus mdiea
100
TngoneUa cretica
Tngonella coentla
Trigbnella fopnitm graeciim
rigonella gladiqta
rigonella cberulescens
Irigonella maerorrhvncna
jigonella qrqbiea '
rigonella balspnap
pigonella ceflheens
99
100
100
88
pigonella spruneriana
rigonella eornieulata
lnonis qrveiisis
\non.t,s biflopa
moms lieitnx____________
Figure I. (Top) Phylogenetic relationship between members of the tribes Vicieae and
Trifolieae based on plastid
sequence data (Steele et al. 2003).
4
Maize and beans were thought to have co-evolved in the New World, peas, lentils, and
chickpeas co-evolved with wheat and barley in the Fertile Crescent, and rice co-evolved
with soybeans in the Orient (Hymowitz, 1990).
Pisum (Field Peal
Pisum sativum L. or garden/field pea is an annual cool season legume that prefers
relatively humid conditions, ranging in temperature from 7 to 30°C (Muelhbauer, 1997).
P. sativum is a self-pollinating diploid, with a haploid chromosome number of seven
(n=7)- Compared to most plant species, Pisum sativum has a large genome comprised of
approximately 4,OOOMbp (Megabasepairsr Mb) (Arumuganathan et ah, 1991);
consequently, the size of each of the seven chromosomes represented by the standard
karyoptype is relatively large.
The morphology of the field pea has been described as bushy or climbing. The
stems grow 30-150cm in length and are relatively weak. The leaves are alternate,
pinnately compound and possess branched tendrils. Some cultivars have a mutation that
converts all laminae to tendrils and are referred to as ‘semileafless’. In addition to the
characteristics of legume flower previously stated, the pea inflorescence is a raceme and
the flowers range in color from white to purple. The fruit morphology has been described
as pods that are curved or straight, swollen or compressed, and having 2-10 seeds which
are dispersed by pod dehiscence along two sutures (Muehlbauer et ah, 1983).
Peas are grown primarily for their fresh or dried seeds, tender pods and foliage.
Peas are cultivated throughout the world’s temperate regions and represent an important .
5
export crop that comprises almost half the world trade in pulse crops. Most of the
commercially produced peas in the United States are grown in Wisconsin, Minnesota,
Washington, and Oregon (Muehlbauer, 1993). From 1993-1997 the number of harvested
acres of pea in the U.S . was estimated to be 250,000 acres, yielding 450,000 tons of
processed pea with a total value of approximately 140,000,000 dollars (Rhodes, 2002).
However in 1990 it was estimated that the total acreage of peas in Canada was three
times greater than that in the U.S. (Stinkard et ah, 1990).
The elucidation of the genetics behind a few morphological features o ff. sativum
was concurrent with the establishment and initial development of genetics as a science.
Specifically, the pioneering works of Gregor Mendel who chose the garden pea as the
primary system in which to test his hypotheses on inheritance (Mendel, 1866). The seven
contrasting pea morphological traits that Mendel analyzed in his famous study include
seed shape, cotyledon color, flower color, pod shape, pod color, flower position, and
plant stature. Mendel published his findings in scientific paper titled “Versuche liber
Pflanzen-Hybriden” or “Experiments on Plant Hybrids” in which he outlined his findings.
Although the nomenclature came later, the principles of segregation and independent
assortment that govern the science of genetics were based on Mendel’s conclusions.
Lens (LentiP)
1
.
Lens spp., like Pisum, are annual legumes that prefer temperate to warm season,
although they require less humidity than pea. Lentil also shares the same haploid
chromosome number and ploidy level as P. sativum-{n=l) and also is primarily a self-
6
pollinating species. The genus Lens is comprised of five species that can be placed into
two crossability groups (Ladizinsky et ah, 1984). Group one contains Z.
T.
onentalis, and L. odemensis and group two contains L. nigricans, L. ervoides. Studies
analyzing the variation of chloroplast DNA sequence among the five members revealed
their phylogenetic relationship. As expected, considering the results from Ladizinslcy’s
fertility studies, members of crossability group one had the closest sequence identity to
each other. L. culinaris was most closely related to L. orientalis followed by L.
odemensis. Furthermore, results from this experiment confirmed the hypothesis that L.
orientalis represents the progenitor species to L. culinaris. The members of the second
crossability group show the greatest amount of genetic variability at the plastid DNA
level, with some results suggesting that L. ervoides may be closer to L. culinaris than L.
nigricans (Muench et ah, 1991).
The morphology of A. culinaris Medik. has been described as a bushy, semi-erect
or erect with slender angular stems that reach a length of 18-36cm (Muehlbauer et ah,
1985). The leaves are pinnate and alternate and there may be ten to sixteen leaflets
attached to the rachis with simple tendrils extending from the top. The fruit consists of
pods that are oval shaped containing 1-2 seeds. One to four flowers can form from each
peduncle (2.5-5cm) and vary in color from white to purple. The inflorescence is a
raceme. The seeds of the lentil come in a variety of shades of yellow, red or orange and
have been described as lens shaped and small. The size of the seed varies in diameter
from 6 to 9mm in the New World (macrosperma) species and 2 to 6mm in the Near East
(microsperma) species (Muehlbauer et ah, 1985).
Although to some extent lentil is used as a green manure crop, the seed is
probably its greatest asset. The seed it typically eaten as dhal which is simply seed that
has been peeled and split. Lentils are primarily grown in Turkey, India, Syria, Nepal,
■
Canada, Ethiopia and Spain. In 1992 India lead the world in total lentil production with
750 tons produced followed by Turkey and Canada. In the United States the total amount
of acreage sown with lentil in 1996 was estimated to be 60,000ha. The total production
from 1984-1993 was approximately 65,000 tons (Muehlbauer, 1997).
The following three chapters will describe three separate investigations involving
Pisum and Lens. Each experiment was designed to utilize the most current technology
available to geneticists as well as to exploit and integrate information gained from past
investigations within these systems. The first two experiments described in chapters 2
and 3 involve an assessment of the logistics for integrating a large insert library into
Pisum genomics. The first experiment will evaluate the efficiency and accuracy of
locating genes, via hybridization, within a newly developed lx genome coverage, Pisinn
sativum Bacterial Artificial Chromosome (BAC) library (Coyne et ah, 2000). The second
experiment will use the BACs resulting from the first experiment to evaluate their
potential use in subsequent pea cytology experiments. Fluorescence In Situ
Hybridization (FISH) with Euorescently labeled BACs will be used to examine the
hybridization characteristics of the large insert clones. The information acquired from
these investigations will be valuable to future experiments directly involving the library
as well as subsequent use of the BACs identified from the library.
8
The final experiment described in chapter 4 will examine the hypothesis of
conserved gene synteny shared among members of the tribe Vicieae' A linkage map of
Lens was generated primarily using a novel set of sequence tagged sites .(STS) markers
with clear positions on the Pisnm linkage map (Brauner et ah, 2002). The STS marker
data was integrated with previously generated Isozymej protein and restriction fragment
length polymorphism (RFLP) data (Weeden et ah, 1992). Gaps between markers will be
closed using RAPD (Random Amplified Polymorphic DNA) markers. The resulting map
will be compared to the consensus map o ff. sativum (Weeden et ah, 199.8) and areas of
synteny will be revealed. Based on the results of this study, other members within the
subfamily Papilionideae could be easily tested using this approach. Thus, the efficiency
of generating linkage maps for each of the species will be discerned.
9
Literature Cited
Arumuganathan, K. E.D. Earle. (1991). Nuclear DNA content of some important plant
species. Plant Molecular Biology Reporter. 9:211-215.
Campbell N.A., J.B., Reece. (2002). Biology (6th ed). Benjamin Cummings, San
Francisco, CA.
Brauner S., R.L. Murphy, J.G. Walling, J. Przyborowski, N.F. Weeden. (2002). STS
Markers for Comparative Mapping in Legumes. J. Amer. Soc. Hort Sci. 127 :616-622.
Coyne, C. K. Melcsem, D.A. Lightfoot, K.E. Keller, R.R. Martin, M.T. McClendon,
D.A. Inglis, E.W. Storlie, K.E. McPhee. (2000) Construction of bacterial artificial
chromosome library for pea (Pisum sativum L.) Pisum Genetics 32: 23-26.
Hymowitz, T., (1990). Grain legumes. In: J. Janick and TE. Simon(eds.), Advances in
new crops. Timber press, Portland Oregon.
Ladizinsky, G., D. Braun, D. Goshen, F.J. Muehlbauer (1984). The biological species of
the genus Lens L. Bot. Gaz. 154:253-261.
Mendel. G. (1866). Versuche uber Pflazen-Hybriden. J. Heredity 42, 1951 p3-37.
(Reprint).
Muehlbauer, F.J., R.W. Short, J.M. Kraft. (1983). Description and culture of dry peas.
USDA-ARS Agricultural Reviews and Manuals, Western Series, No. 37. Western
Region, Oakland, California. P.92.
Muehlbauer, F.J., J.I. Cubero, R.J. Summerfield. (1985), Lentil (Lens culinaris Medik.)
P.266-311. In: R.J. Summerfield and E.H. Roberts (eds.) Grain Legume Crops. Collins 8
Grafton Street, London, UK.
Muehlbauer, F.J. (1993). Food and grain legumes, p.256-265. In: J. Janick and J.E.
Simon (eds.), New Crops. Wiley, New York.
Muehlbauer, F.J, (1997). Pisum sativum L. NewCRQP FactSHEET
Muehlbauer F.J., A. Tullu. (1997). Lens culinaris Medik. NewCROP FactSHEET.
Muench, D.G., A.E. Slinkard, G.J. Scoles. (1991) Determination of genetic variation
and taxonomy in lentil (Lens Miller) species by chloroplast DNA polymorphism.
10
Rliodes D. (2002). U.S. Processing Pea Production Statistics (1992-1993).
http://www.hort.purdue.edu/rhodcv/hort410/peas/pe00002.htm
Stinkard, A.E., R.S. Bhatty, B.N. Drew, R.A.A. Morrall. (1990) Dry pea and lentil as new
crops in Saskatchewan: A case study, p.159-163: In: J. Janick and J.E. Simon (eds.),
Advances in new crops. Timber Press, Portland OR.
Steele, K. P., and M. F. Wojciechowski. (2003). Phylogenetic Systematics of tribes
Trifolieae. and Vicieae (Fabaceae). Advances in Legume Systematics. (in press).
Stem, K.R. (1997). Introductory Plant Biology. Wm.C.Brown publishers. Dubuque,
IA.
Weeden, N.F., F.J. Muehlbauer, G. Ladizinsky. (1992) Extensive conservation of linkage
relationships between pea and lentil genetic maps. J. Hered. 83:123-129.
Weeden, N.F., T.H.N. Ellis, G.M. Timmerman-Vaughan, W.K. Swiecicki, SM. Rozov,
V.A. Berdnikov. (1998). A consensus linkage map for Pisum sativum. PiSum Genetics.
30:1-4. I
Zohary, D., M. Hopf (1993). Domestication of plants in the old World - The origin and
spread of wild cultivated plants in West Asia, Europe, and the Nile Valley. Clarendon
Press, Oxford
11
■ CHAPTER 2
CHARACTERIZATION OF PISUM BACTERIAL ARTIFICIAL CHROMOSOME
(BAC) LIBRARY '
Literature Review
Introduction
Since the introduction of molecular cloning techniques in the late 1970s, DNA
libraries have become a powerful means for accessing genetically important fragments of
DNA. The segmentation and subsequent subcloning of genomes allows researchers to
avoid some of the experimental limitations of manipulating complex genomes in vitro.
By means of displaying sequence information in an ordered array such as in microtitre
plates or on high-density filters, DNA libraries can facilitate the identification and
cloning of DNA sequences that may otherwise remain unresolved. DNA libraries, of one
form or another, have been constructed for nearly all agronomically and genetically
important species and have played a crucial role in furthering the status of both structural
and functional genomics in these species.
DNA Libraries
The extent to which DNA can be systematically analyzed has generally been
governed by the recombinant cloning vector used in the cloning experiment. In
particular, the size of the insert a vector can faithfully accept remains a limiting factor=
Consequently, a variety of libraries have been developed that range in both size and
complexity (Ausubel et ah, 1999). Libraries employ the ability of genetically engineered
12
cloning vectors to accept fragments of DNA. These ‘recombinant’ vectors are
subsequently transformed into a host cell to stabilize and propagate the vector. The
components of a cloning vector such as a selectable marker, a sequence for controlling
copy number, and multiple cloning sites allow the successful cloning of fragments to be
accomplished with little effort. The simplest cloning vector, the plasmid, is used
routinely in single event cloning experiments, such as the cloning of a PCR product.
Although some plasmids have been shown to accept fragments up to 2'0kb, routine
plasmid cloning is typically restricted to fragments that are between three and IOlcb
hindering their utility in constructing genomic libraries. Thus, plasmids generally are
only used for subcloning arrays of larger fragments.
Bacteriophage lambda (X) is a cloning vector that has the ability to package DNA
into the phage particle, thus providing a stable means of maintaining insert integrity.
Furthermore the high magnitude of phage multiplication provides-aceess to large amounts
of cloned DNA. Lambda cloning is also limited to relatively small fragments of DNA,
with 25kb being the upper size limit. Thus, lambda vectors are typically used in small,
targeted libraries. Cosmid cloning vectors are similar to plasmids but have the advantage
of being to able hold larger inserts. This ability is gained from the addition of cos sites
derived from the bacteriophage DNA. The cos sites facilitate proper packaging of the
DNA clone by first ligating itself to the insert and creating circular DNA that is suitable
to enter the capsids. Consequently cosmids can hold DNA fragments up to 45kb
(Fairbanks et al., 1999).
13
The utility of cDNA libraries provides a means for restricting the DNA fragments
examined to that of only actively transcribed genes. Copy DNA or cDNA libraries, made
from RNA templates, are typically assembled using lambda or plasmid cloning vectors.
mRNA are typically relatively short, thus, plasmid vectors are generally used (Ladish et
ah, 1999).
Of particular interest for this study are large insert libraries such as Yeast
Artificial Chromosomes (YACs) and Bacterial Artificial Chromosomes (BACs). These
libraries contain genomic fragments that can range from 0.1Mb to several Mb, and are
essential for characterizing genomes as a whole (Wang et ah, 1996). YACs have lost
popularity among those using libraries due to their tendency to form chimeras, instability
of clones, and difficulty in preparing DNA (Yu et ah, 2000).
'
Bacterial Artificial Chromosome
A Bacterial Artificial Chromosome (BAC) can be defined as a genetically
engineered F factor, isolated from E. coli, with the ability to hold fragments of DNA up
to 3OOkb. BAC components consist of regulatory elements that confer selectable
antibiotic resistance, regulate replication, and ensure low copy number within the host
cell (Fairbanks, 1999). BAC DNA is typically introduced into the host cell using
electroporation, a method that utilizes a brief but substantial electric pulse to open ‘pores’
in the cell membrane and permit insertion of large circular DNA.elements into E. coli
cells. The stability of BACs compared to YACs and the average insert size compared to
cosmids, both demonstrate the superiority of BACs as vectors for constructing and
1X
14
maintaining large insert libraries. Consequently, BAC libraries now exist in many
significant plants species including but not limited to Arabidopsis thaliana (Wang et al.,
1996), pearl millet (Allouis et al., 2001), potato (Song et al., 2000), sugarcane (Tomkins
et al., 1999), Medicago truncatula (Nam et al., 1999), sorghum (Woo et al., 1994), barley
(Yu et al., 2000) and pea (Coyne et al., 2000)..
The maximum allowed insert size of BACs is 3001cb. The large size promotes the
logistics of cloning the total genetic composition of a species; however the task itself
remains laborious. Briefly, total DNA is extracted in the case of pea, from seedlings that;
were grown in the dark for 3 days (Coyne et ah, 2000). The total extract is then partially
digested with six base pair restriction enzymes, and size selected twice, using pulsed field
gel electrophoresis (PFGE). The size selected DNA is gel purified, ligated into the
appropriate BAC cloning vector and electroporated into competent E. coli cells. The
■clones are then stored in 96 or 384 well microtitre plate's (Nam et ah, 1999; Coyne et ah,
2000). The library can be screened using robot-arrayed high-density filters and DNA
hybridization of radiolabeled probes or via PCR using pooled BACs (Nam et ah, 1999).
An initial characterization of the BAC library with respect to plastid DNA
contamination, average insert size, and the ability to locate specific sequences is
necessary to assess the quality of the library. The evaluation of the average size of
inserts within the library is typically accomplished by size fractionating random BAC
clones using PFGE (Allouis et al., 2001; Tomkins et ah, 2001; Song et ah, 2000). The
extent to which the library contains plastid DNA contamination can most easily be
estimated by hybridization of radiolabeled plastid sequences to BAC array filters. Extra
15
nuclear DNA is typically not desired in a DNA library, thus it is important to identify
clones containing such sequences. Certainly when dealing with a genome the size of pea,
the more ‘nuclear specific’ the inserts of the library are, the fewer clones it would take to
encompass a lx coverage of the genome. For instance results from screening the potato
BAC library (Song et ah, 2000) indicated an estimated ~3% of the library contained
BACs with organellar DNA inserts.. When this number is applied to a library constructed
from pea in which it is estimated that 250,000 clones would provide a 4-5x coverage
approximately 7500 clones would contain cpDNA inserts. Thus, the importance of
reducing the amount of. contamination, or at minimum, acknowledging the presence of
organellar DNA inserts is of importance to the project. The experiment demonstrates
how the percent contamination can act as one indicator of the overall quality of the BAC
library. It has been suggested that isolating tissue with relatively low amounts of
chloroplasts can reduce the amount of chloroplast contamination. For example, isolating '
genomic DNA from source tissues that have either been light deprived, or are located
within the innermost tissue of a particular plant, would decrease the amount of
chloroplasts and thus cpDNA within the genomic DNA extract (Tomldns et ah, 1999).
Although plastid DNA contamination is generally undesirable, some, (1-3%) is expected
./
(Yu et ah, 2000),
. Finally, the location of specific low copy sequences within the library can be .
determined in various ways. Anchored Sequence Tagged Sites (STS) and cDNA clones
have been used to identify such sequences. Allouis et ah (2001) demonstrated the utility
of using STS markers to locate, clone homologs by choosing STS markers that are
16
genetically distributed throughout the pearl millet linlcage map. Subsequent PCR
amplification of a pooled BAC library with STS specific primers verified the quality of a
pearl millet BAC library. Similarly, screening of pooled BACs allowed the
identification of clones containing ethylene response genes in Medicago while
establishing utility of the library (Nam et ah, 1999).
BAC Applications
Although the previously described methods for confirming the utility of the
library also can provide BAC insert data, such an approach is certainly not limited to just
library characterization. The ability to locate BAC clones using molecular mapping
markers, also Icnown as positional cloning, has become an important step in both physical
mapping and investigations of the sequences flanking the sequence of interest.
Resistance Gene Analogs (RGAs) provide a useful source of low copy sequences,
appropriate for assessing condition of the library. Yu et ah (2000) used a set of RGA
probes to screen BAC library filters of barley (Hordeum vulgare). One hundred twenty-.
.
x,
one clones were identified that contained positive sequence homology. In a similar
fashion, BACs containing sequences associated with cyst nematode resistance (CNR) in
soybean {Glycine max) were identified from a soybean BAC library using RFLP-based
markers tightly linked to the CNR locus (Danesh et ah, 1998). Zwick et ah (1998) used
six RFLR markers mapped to a linkage group in which liguleless is anchored, to screen a
BAC library of Sorghum.bicolor. Sixteen BACs were identified and physically mapped
along chromosome I of S. bicolor. Results indicated that the physical map reflected the
.
1 7
.
linkage order established by RFLP mapping and also confirmed previous data indicating
a conservation of the liguleless linkage group in rice and maize.
The large insert size in a BAC library allows the characterization of sequences
flanking the original sequence identified. Screening flanking sequences using short
anchor probes facilitates structural and evolutionary studies. Cregan et al. (1999)
designed a targeted approach to identify simple sequence repeats (SSR) linked to
previously established molecular markers in soybean. Two BACs identified by RFLP
based markers conferring soybean cyst nematode resistance, were used to construct a
subclone library. Hybridization screening of this subclone library with SSR probes
identified 14 unique SSRs all localized within the genetically defined cyst nematode
resistance locus.
BAC clones have been used to characterize complex areas of the genome. The
arrangement and copy number of centromeric &m3AI DNA fragments in sorghum was
’ determined using subclones of BACs (Miller et al., 1998). Sequence analysis and in situ
hybridization of the subclones indicated a complex arrangement of the SauSAl fragments
localized within the centromeres of sorghum. A method was devised to visually assess
the complexity of the DNA found within the BAG, which could then be correlated with
gene density within a particular portion of the genome. Using in situ hybridization of
BAC subcloned DNA on isolated intact BAC clones, Jackson et al, (1999) was able to
characterize areas within the BAC with respect to sequence composition and
arrangement.
18
The informative DNA content of BACs when used in interspecies comparative
studies can reveal mechanisms of gene or genome evolution. Areas of synteny between
tomato and Arabidopsis were investigated by comparing tomato (Lycopersicum
esculentum) BAG sequence, to Arabidopsis genomic sequence (Ku et ah, 2000). Results
obtained from comparing gene order and content, suggested that the driving factor
influencing species divergence is likely to have been a series of large-scale genome
duplications such as polyploidy rather than smaller rearrangements such as duplications
and translocations. In a similar study, a BAC contig from Arabidopsis was used.to
propose a mechanism of genome expansion in Brassica rapa (Jackson et ah, 2000).
Rather than relying on sequence data, this experiment used a macroscale approach of '
arraying fluorescently labeled BAC contigs on extended DNA fibers of Brassica. The
FISH signal was duplicated in the Brassica genome. However the orthologous loci
retained the same length as seen in Arabidopsis1 suggesting that large scale duplications,^
rather than an amassing of intergenie repeat sequences most likely contributed to the
genome expansion.
Recently a BAC library was constructed of Pisiim sativum'(germplasm line PI
269818) genomic DNA with.an estimated lx genome coverage. As a prelude to the
many applications this particular BAC library will contribute to, this study will assess the
overall quality of the library.' To test the utility of this BAC library, a series of ■
experiments will be implemented to assess the quality of the BAC library. Initially, the
amount of chlofoplast DNA contamination will be estimated by quantifying the number .
of positive BAC clones identified by hybridization of chloroplast sequences to the
19
library. Secondly, the quality of the arrayed nuclear DNA clones will be investigated by
hybridizing with.characterized repetitive clones (25s rDNA). Finally, the ability to locate
low copy, (gene family) and unique sequences (single copy), within the library will be
assessed using hybridization followed by a PCR verification, to find genetically anchored
and characterized loci in the lx P. sativum BAC library.
20
Materials and Methods
The BAC library, previously constructed by Coyne et al. (2000) of Pisum sativum
DNA using germplasm line PI269818 of the Western Regional Plant Introduction
(Pullman, WA.) had been prepared by cloning partially restricted DNA into vector
pCLD04541 (V41). BIBAC vector V41 is a binary vector developed by Dr. ID .G Jones
and is suitable for maintaining large inserts in E. coli isolates with the additional
capability of Agrobacterium-medidXed transformation of large inserts into plants.
Partially digested Hindlll fragments were ligated into the cloning vectors, yielding an
average insert size of I IOkb. Consisting of approximately 50,000 clones, this library
represents a lx coverage of the total pea genome ((5xl04)(l.IxlO5)= ~ 5000Mb).
Robotics (Genomic Solutions) were employed to array each individual colony twice on a
high density nylon membrane (Hybond N) (Coyne et al., 2000).
Dr. Clare Coyne, Washington State University- USDA NPGS, generously
donated three sets of BAC filters to our program. Immediately upon receiving the BAC
filters, they were stored at 4°C degrees until further use.
■Probe Construction
Probes appropriate for identifying single copy and multi-copy DNA sequences in
the Pisuni sativum genomic BAC library were generated from genomic Pisum DNA
(cv.'Sparkle1) via PCR using previously characterized primers developed for genetic
mapping in Pisum (see Tabled).
21
Table I . Characteristics of Probes used to Screen BAC library
Accession
Number
Gene
Symbol
Product
Length
Gene Organization
NA
cpDNA
51 Qbp
H igh co p y
25s rDNA
X52575
25s
400bp
H igh co p y
Cytosolic glutamine
sythetase
U 28925
Gsc
1 1 OObp
S in g le co py
Diminuto
' D 86494
Dimin
1200bp
S in g le c o p y
Ascorbate
peroxidase
M 93051
A pxl
1700bp
.. S in g le co p y
Sucrose synthase
AJ 0 12080
Ssyn
1850bp
S in g le co p y
P393
AA430912
P393
516bp
D u p lic a te co p ie s
P628
• AA430910
P 6 28
640bp
S in g le co p y
T protein glycine
decarboxylase
complex
A J 2 22771
Gdct
1360bp
S in g le co p y
PSL1 Lectin
IVM 8 1 6 0
Lectin
1OSObp
*G F
Leghemoglobin
AB009844
Lb
IOOObp
*G F
X 0 0 8 0 6 .1
RbcS
61 Obp
*G F
■M 6 4 6 1 9
Cab
660bp.
*G F
Disease resistance
response protein 49
U 31669
Drr49
Ribosomal protein
CL22
M 60951
Rpl22
900bp '
*G F
L08664 ■
Ftase
900bp
*G F
Gene Name
Chloroplast trnL-c &
trnL-d
/
Ribulose 1,5bisphosphate
carboxylase small
subunit
Chlorophyll a/bbinding protein
Farnesyltransferase
*GF=Gene family member
■ 6,OObp
■
*G F
22
PCR products were immediately labeled using random priming and used for probe
hybridization or cloned into plasmids for use at a later time. Genomic DNA extractions
using the CTAB method followed previously described protocols (Torres et ah, 1993):
Freshly harvested meristem leaf tissue (about I cm2) was placed into a small mortar. One
milliliter of Extraction Buffer (50 ml IM Tris-HCl, pH 7.5, 70 ml SM NaCl,
40 ml. 0.5M EDTA, 340ml ddH20, 1% mixed alkyltrimethylanimonium bromide,.and
0.4% P-mercaptoethanol) was layered on top of plant tissue. The tissue was ground with
a:pestal into a slurry. The slurry was incubated for 30 minutes at 65°C in the presence of
IOOpi of chloroform:isoamyl alcohol (24:1). The tube was to allowed cool to room
temperature, filled to the top with additional 24:1 chloroform:isoamyl alcohol and
vortexed to emulsify. The aqueous phase was separated from plant tissue and chloroform .
wash, using centrifugation in a fixed rotor centrifuge at 14g (Beckman Coulter
Microfuge® 18 Centrifuge) for 5 minutes and carefully pipetted off the top. DNA was
precipitated by adding 0.8-1.0 ml of ice cold 95% ETOH, which was initially layered on
top of the buffer solution and incubated at room temperature for 10-20 minutes to allow
precipitate to form at the solution interphase. Solutions were mixed by inversion to
complete the precipitation of DNA. The precipitate was pelleted using a centrifugation of
IOg for 5 minutes, and the extraction buffer and alcohol.was carefully decanted. The
pellet was washed once for ten minutes at room temperature with 1.0 ml of 0.2M sodium
acetate in 75% ETOH and was washed a second time in 0.01M ammonium acetate in
■
75% ETOH for five minutes. DNA was dissolved in appropriate amount of TE (IOmM
23
Tris, ImM EDTA), typically ranging between 0.1 and 0.15 mis. One microliter of
RNASE A was added to the samples to digest contaminating RNA.
PCR was performed in either an MJ Research® PTC-100 or PTC-200 “DNA
Engine” thermocycler. Promega Taq or Platinum Taq (Invitrogen) polymerase was used
in the PCR reaction for primer extension, Reaction preparation and solutions are as
follows:
Reagent
ul/reaction
ddH20
15.78
Stock Cone.
. Reaction Cone.
MgClz free buffer
2.5 -
IOlC
IX
MgCh
3.0
25mM
3mM
dNTP’s
0.87
IOmM each nucleotide
Taq polymerase
0.1
5 units/pi
0.5units
Forward Primer
0.75
20pM
0.6pM
Reverse Primer
' 0.75
20pM
0.6pM
DNA
0.34mM each
1.0
25.0 pi
*Water was sterile
One micrdliter of template DNA was pipetted into individual microcentrifuge
tubes. According to protocol above, a master mix was constructed by considering the
number of reactions required and multiplying each specific addition by the number of
reactions needed plus one or two extra reactions, to account for inherent pipetting errors.
Twenty-four microliters of the master mix was aliquotted into each reaction tube, which
already contained the one microliter of template DNA. If the PTC-100 was used, it was
24
necessary to top each tube with I -2 drops of sterile mineral oil to reduce evaporation.
The thermocycler was programmed to run according to the following protocol:
Cycling Parameters;
1-
94°C - 2.0 minutes
Initial DenatUration
2-
94°C - 1.0 minutes
Denafuration
3-
37-62°C - 2.0 minutes
Annealing
4-
72°C ^ 2.0 minutes
Extension
5-
35x to step #2
Cycling
6-
72°C - 8.0 minutes
Final Extension
7-
4°C - indefinitely
Holding Temperature
PCR products were separated on a 1% agarose gel to check the relative quality of
amplification as well as size of amplification product. To isolate the PCR product from
any unwanted contaminants such as unused primer and mis-primed products; the PCR
product that corresponds to the expected size was extracted from the agarose gel and
purified according to protocol supplied in “QIAquick Gel Extraction Kit’’ by Qiagen®.
DNA was eluted from columns with minimal elution buffer.
Gel extract product was quantified by resolving the product next to “DNA
Quantiladder” using agarose gel electrophoresis.
Concentration was adjusted with TE to
the standards required for the radiolabeling reaction;
Probe Labeling and Hybridization
Random priming reactions, using the Klenow fragment to fill gaps between
primed random IOmers, were used to incorporate 32P labeled dCTPs into the isolated
25
sequence. DECAprime™ II kits by Ambion® were used to facilitate the radiolabeling of
between I and 3 probe sequences per reaction.
Upon completion of the labeling reaction, the product was cleaned using
“Performa DTR Gel Filtration Systems” column by Edge Biosystems® according to
manufacturer's protocol. This step is necessary to filter out unincorporated labeled
nucleotides from previous reaction. Cherenkov counts were used to quantify the efficacy
of labeling reaction using a Bioscan QC 400 xer scintillation counter.
Hybridization of probe to filters containing BAC clone arrays was carried out as
follows. One or more filters was prehybridized in 20-30mls of “Prehyb solution” (5x
SSC, 5x Denhardts (lg. Ficoll 400, lg. PVP (Polyvinyl Pyrrolidone), lg. BSA (Bovine
Serum Albumin), 1% SDS (Sodium Dodecyl Sulfate)), using a rotisserie style,
hybridization oven, fitted for hybridization in tubes. The filter was prehybridized for 2030 minutes during which time the probe and blocking DNA was prepared. Blocking
DNA consisted of 20pl of sheared salmon sperm DNA (Eppendorf, l Omg/ml), that was
denatured by boiling for 5 minutes, followed by a rapid cooling in an ice bath. Denatured
salmon sperm DNA was added to the “Prehyb” within 5 min of the commencement of
prehybridization.
Concurrent to prehybridizing and preparation of blocking, the radiolabeled probe
was also denatured by incubating in boiling water for 10 minutes and likewise was
rapidly cooled in an ice bath to avoid re-annealing. The entire amount of denatured
probe was added directly to the prehybridization (now hybridization) solution and
allowed to hybridize, overnight at either 37° (initially) or 55°C.
26
Stringency Washes and Autoradiography of BAC Filters
Following the hybridization reaction, the filters were subjected to a series of
stringency washes. This step ensured that the filter was clean of hybridization solution
and free of residual, non-specifically bound probe. The following is the protocol
developed for utilizing stringency washes to prepare hybridized filters of the pea BAC
library for autoradiography.
The hybridization solution was decanted into a proper radioisotope disposal
container. The filter was rinsed of hybridization solution using a low stringency wash
(LS=2xSSC, 0.1% SDS) for 10 minutes at 37°C, under slight agitation. Before
administering an additional wash, the relative amount of isotope fixed to filter was
estimated by scanning the filter with a Geiger counter. For clear resolution of signal it
was empirically determined that the average intensity should be between 2000-3OOOcpm
on the Geiger counter. This objective was met by applying additional washes; however,
with each additional wash an increase in stringency and incubation temperature was used.
For example, a more aggressive medium stringency (MS= 0.2xSSC/0.1%SDS) a 42°C
incubation temperature was used. The MS wash was routinely followed by a high
stringency (HS= 0.1xSSC/0.1%SDS) wash in which a 65°C incubation temperature was
used. At times the filter needed only to be washed once at each stringency. However, on
some occasions multiple washes at high stringency, in addition to the three routine
washes were needed. Each wash was allowed to proceed for 15 minutes at which time a
reading was taken with the Geiger counter. The number and stringency of washes used
27
was dependent on probe concentmdon and efficiency of isotope incorporation and thus
had to be adjusted with each individual filter. A final 2x SSC rinse for 10 minutes at
room temperature was used to remove excess fSDS from washing steps. The filter was
wrapped in one layer of cellophane and exposed to X-ray film within 24 hours of
hybndization. It was beneficial to have a small amount of background, as it assisted in
aligning the autoradiography to the filter map.
The filter was exposed to X-ray film (Kodak X-OMAT® AR Scientific Imaging
Film) to visualize any hybridization signal. This exposure was carried out using a
developing cassette supplemented with an intensifying screen and stored for 24-30 hours
at -80°C. Subsequently, the cassette and film were allowed to thaw, and the film was
developed according to manufacturer's instructions.
Hybridization Signal Scorinp
The definition of legitimate clone was the presence of two probe signals within
one square patch with the idea that each colony is duplicated within one patch. This
system allows the scorer to differentiate between artifactual signal and a true
hybridization. The two signals may be oriented in one of four ways: vertical, horizontal,
diagonal right and diagonal left, with each arrangement representing the clone's plate of
origin. Superimposing a map of the library over the developed film allowed the
assignment of an identification number, based on the location and alignment of the
signal, which is associated with the clone's origin within the library. Accordingly, This
information was used to request the clone of interest from the library curator.
28
The library was initially screened for the presence of chloroplast DNA
contamination. This was done using an annealing temperature of 37°C. TrnL-c and trnLd primers, originally described by Taberlet et al. (1991), were used to generate the
chloroplast DNA specific probes. The number of positive signals was counted and used
to estimate the percent of clones with plastid inserts.
Clone Characterization and Sequence Analysis
A small aliquot of the freezer stock containing E.coli cloned BACs was streaked
out on LB (Luria-Bertini Medium, premanufactured LB agar EM Science, EM Industries
Inc. Gibbstown, NI.) agar plates containing an appropriate selective agent. Using Vector
PCLD045411 the selection was accomplished using ampicillin (20pg/ml) in the LB
media.
Preliminary confirmation of the identity of the clone insert was accomplished by
direct colony PCR. This method is similar to the described PCR reaction except that the
template was obtained directly from the E.coli host, rather than purified DNA. The
experiment was carried out by picking a small portion of an isolated colony, preferably a
satellite, from the LB plate and adding the cells to a prepared PCR master mix that
includes the primers suitable for amplifying the putative sequence from the insert. A
duplicate PCR of each colony lift was performed using primers specific to sequences
found on the vector as a positive check to confirm the polymerase chain reaction.
To further confirm the presence of the sequence of interest within the insert it was
necessary to sequence a portion of the insert DNA- not including the rDNA and cpDNA
29
clones. Sequence template was obtained from the direct colony PCR reaction, which was
either immediately sequenced or cloned into a vector and sequenced at a later time.
At times the sequencing reaction required a higher template yield than the direct
colony PCR provided. This requirement was overcome by inserting the PCR fragment
into a cloning vector suitable for sequencing. In the same manner as the previous gel
excision, the PCR product was separated from the agarose gel and cloned into a vector.'
The cloning reaction, facilitated with TOPOClonell™ by Invitrogen, ligates the PCR
clone into a vector containing T3/T7 priming sites, thereby p e r m it t in g subsequent
sequencing reactions.
The sequencing reaction was carried out using ABI Prism BigDye Terminator
v.2.,0 100RR cycle sequencing kit (Applied Biosystems, Foster City, CA), to end
sequence each PCR product. Sequences were generated in both forward and reverse
directions using the T3 and T7 priming sites located on the cloning vector. Reaction
conditions include mixing 0.5pl forward or reverse primer, 3.5pi PCR generated template
and 2pi BigDye reaction mix. Reactions were performed on a Perkin Elmer GENEAMP
PCR System 9600 with cycling parameters: 96°C for 10 seconds, 50°C for 5 seconds,
and 60°C for 4 minutes. Products of sequencing reaction were cleaned and concentrated
using a routine ethanol precipitation followed by re-dissolving in 0.8 pi of Blue Dextran
Dye. Sequencing products were applied to a paper comb and inserted into sequencing gel
according to manufacturer's protocol. The sequencing gel was run on an ABI Prism 377
automated DNA Sequencer (Applied Biosystems, Foster City, CA). Chromatograph files
were converted into forward and reverse sequences using Chromas™ version 1.45
30
software (Copyright Conor McCarthy, Griffith University, Southport, Queensland,
Australia; available as freeware at http://www.technelysium.com.au/chromas.htral).
Sequences were edited at the 5’ and 3’ ends for low quality sequence using Bioedit
Sequence Alignment Editor (copyright Tom Hall, North Carolina State University;
available as freeware at http://www.mbio.ncsu.edu/BioEdit/bioedit.html). Sequence
analysis using BLAST (www.ncbi.nih.org/blast) verified the presence or absence of the
putative gene sought within the BAC insert.
31
Results
C h lo r o p la s t D N A S c re e n
' The trnL~c and trnL-d PCR primers (see Table I Chap. 2 Materials and Methods)
designed specifically to amplify fragments of noncoding regions of chloroplast DNA
(Taberlet, 1991), were generously donatedtoour project by Dr. M. Lavin (Montana State
University). Initial results from an “annealing temperature gradient PCR” revealed a
'
PCR product of approximately 510 base pairs when an annealing temperature of between
50C and 65C was used (see Figure 2). When generating the fragment for use as a
hybridization probe, an annealing temp of 60C was used. This product was radiolabeled
and used to screen BAC filter SIU-WSU Pea #49-72 for the presence of chloroplast DNA
I
sequences. The autoradiography of the hybridized filter revealed 92 positive hits under
medium/high stringency (See Figure BA). Using the following Equation: (92
signals)/(9216 colonies per filter) = 0.998% or 1.0%; it was estimated that a minimum of
I
1.0,percent of the BACs within the library would have inserts containing chloroplast
sequence. However, it should be noted that this small probe represents only a portion of
the chloroplast genome, thus there should be more BACs with cpDNA inserts that
remained undetected. Furthermore, because the annealing temperature used to screen for
cpDNA (37°C) is considered low by most hybridization standards, the representation of
:
the cpDNA within the library should be confirmed using a higher annealing temperature
(>55°C).
)
:
'
-
:
32
NFWGradient Primers: Chloropiastic DNA(Lavin)
07,2501
..
Annealing Temps
SflC
SfldC
S iz e
52 SC
54 2C
% dC
58 SC
$ 1 OC
SZ TC
63 SC
64 TC
SSOC
F ig u r e 2 . G e l e le c tr o p h o r e s is o f P C R p ro d u c t a tta in e d fro m g r a d ie n t P C R
o i t r n L - c and
t r n L - d u s in g P is u m g e n o m ic D N A .
R ib o s o m a l D N A S c re e n
O n e p r i m e r s e t ( s e e T a b le I C h a p . 2 M a te r ia l a n d M e th o d s ) d e s ig n e d to a m p lify a
4 0 0 b p f r a g m e n t o f th e 2 5 s c o d in g re g io n r e p r e s e n tin g th e la rg e (4 5 s ) rD N A a r ra y w a s
u s e d to e s tim a te th e p e r c e n t o f in s e rts c o n ta in in g r D N A s e q u e n c e s . B A C f ilte r 3 P (9 7 120)3 w a s s c r e e n e d w ith th e r a d io - la b e le d 2 5 s rD N A P C R p r o d u c t u n d e r m e d iu m /h ig h
s trin g e n c y . T h e a u to r a d io g r a p h y o f th e h y b rid iz e d f ilte r d is p la y e d 33 s ig n a ls (S e e F ig u re
3 B ). T h u s , I w a s a b le to e s tim a te , u s in g E q u a tio n I, th a t a p p r o x im a te ly 0 .3 5 p e r c e n t o f
th e lib r a r y in s e rts c o n ta in rD N A .
G e n e f a m ily S c re e n
F o r a c o m p le te lis t o f p r o b e c h a ra c te ris tic s r e f e r to (T a b le I C h a p . 2 M a te ria ls a n d
M e th o d s ). A to ta l o f fiv e p r o b e s s p e c if ic fo r k n o w n g e n e f a m ilie s w e r e u s e d to sc re e n
th e B A C lib ra ry . H o w e v e r , d u e to th e lo w r e u s a b ility o f th e f ilte r s e ts , o n ly o n e o f th e
fiv e p r o b e s
{ L e c t i n ) w a s u s e d to s c re e n th e e n tire lib ra ry .
33
F ig u re 3. A ) H y b r id iz a tio n o f c h lo ro p la s t p ro b e to
o f 2 5 s rD N A p r o b e to P is u m B A C filte r.
P is u m B A C filte r. B )H y b rid iz a tio n
T h e o th e r f o u r p r o b e s w e r e u s e d to s c re e n at le a s t tw o o f th e fiv e filte rs . T h u s in so m e
c a s e s , le s s th a n 5 0 % o f th e p e a B A C lib ra ry w a s s c r e e n e d fo r s o m e o f th e d e s c rib e d g e n e
f a m ilie s . T h e s c r e e n in g o f th e fiv e p r e v io u s ly d e s c r ib e d p ro b e s d e te c te d a to ta l o f n in e
p o s itiv e s ig n a ls d e te c te d . O n ly o n e o f th e n in e p o s itiv e s ig n a ls w a s in itia lly c o n firm e d
u s in g b o th P C R a n d s e q u e n c e id e n tific a tio n (T a b le 2 ). T h e B A C c lo n e th a t w a s
s e q u e n c e d w a s v e r if ie d a s c o n ta in in g th e R u b is c o (s m a ll s u b u n it) g e n e w a s B A C 1 0 3 -K 1 .
(S e e F ig u r e 4 A ). A lth o u g h th e e x p e r im e n t p ro d u c e d o n e p o s itiv e ly id e n tifie d B A C
c lo n e , th e r e w e r e a ls o tw o c a s e s in w h ic h fa ls e p o s itiv e s w e r e p r o d u c e d th a t a re
n o te w o r th y .
I n itia lly th e l e g h e m o g lo b in p ro b e id e n tifie d m a n y B A C c lo n e s d is p la y in g
s ig n if ic a n t h y b r id iz a tio n s ig n a ls , in d ic a tin g in s e rt s e q u e n c e h o m o lo g y to le g h e m o g lo b in
(S e e F ig u r e 4 B ).
34
T a b le 2. S u m m a r y o f R e s u lts fr o m B A C L ib ra r y S c re e n in g
Probe Name
Multicoov:
cpD N A
2 5 s rD N A
# Filters
Probed
* # Positive
Hybridization
Signals
PCR
Verified
Sequence
Verified
BACs
Identified
1
1
92
33
NA
NA
NA
NA
NA
NA
5
5
5
5
5
5
5
2
O
O
7
Yes
NA
NA
“ Yes
Yes
NA
NA
NA
NA
Yes
NA
NA
NA
N e g a tiv e
NA
NA
NA
NA
89-E10
Yes
Yes
Yes
N e g a tiv e
N e g a tiv e
N e g a tiv e
N e g a tiv e
Yes
No
No
N on e
N on e
Low Copy:
G sc
Dimin
Apx 1
Ssyn
P393
P 6 28
Gdct
Rpl22
Ftase
1
1
1
O
O
O
O
N one
N on e
N one
N on e
N one
N one
N on e
N on e
Gene Family:
Lectin
Lb
R bcS
Cab
Drr49
5
5
2
2
3
1
4
2
I
1
103-K1
N one
N on e
‘ Number does not represent the total number of hits, rather the number of clones that were
subsequently PCR verified (Not including cpDNA and rDNA). NA=Not Applicable. “ False Positive
R b c s s p e c if ic p ro b e . B ) B A C f ilte r
L b s p e c if ic p ro b e . In se t: C lo s e -u p o f h y b r id iz a tio n s ig n a l.
F ig u r e 4 . A ) B A C f ilte r h y b r id iz e d w ith
h y b r id iz e d w ith
35
Furthermore, the majority of those clones identified from the screening tested positive '
using PCR to verify the presence of the sequence. When sequenced, however, the results
indicated that insert Contained either small portions of the original transcribed gene
(pseudogene) or repetitive sequences sharing enough homology to allow hybridization.
and PCR amplified (data not shown).
Screening the library using probes corresponding to Drr49 produced a single
intense hybridization signal under normal conditions. Resolving the orientation of the
signal required additional high stringency washes and a shortened re-exposure time
(Figure 5 A and B). BAC clone 2 1-F18 was identified as containing sequences
homologous to the Drr49 sequence. A Southern blot was performed (Master candidate
Susan Hance) using the DrMP probe on BAC 21-F18 to examine the cause of the failed
PCR in light of the robust filter screening signal (see Table 2). The results of the
Southern blot may indicate whether enough sequence homology was present to allow
.hybridization of the probe (data not shown) but not PCR. ■
Low/Single Copy Gene Screen
. Nine single/low. copy markers (see Table I in BAC Material and Methods for a
complete list), genetically anchored to Piswn linkage groups IV and VIl were used to
screen the lx Pisum BAC library. The hybridization reactions were initially carried put
under low stringency conditions, i.e low annealing temp of 37C, with the intent of
maximizing number of hybridization points .
36
B
.
i.
" •
-
' T
I
v '
is- >'
T .
th . % -
, ,
% # a-
■X V T«' 'I
1
f fr
r
J i ,
■
;
SH
f
e
.
-
/
; > -
,
■
/
/
o ------- kjl i s , i t ? p i u u c iu r is u m d / \ v iiu r a r y . a ) S n o rte n e d r e ­
e x p o s u r e tim e o f h y b r id iz a tio n s e e n in ‘A ) ’. In se t: C lo s e - u p o f h y b r id iz a tio n sig n a l.
P rio r to p o o lin g , th e p r o b e s w e r e q u a n tifie d w ith re s p e c t to m o le c u la r w e ig h t u s in g
e th id iu m b r o m id e g e l a n a ly s is ru n n e x t to s iz e /w e ig h t s ta n d a r d D N A la d d e r s ; th e p ro b e
c o n c e n tr a tio n s w e r e b a la n c e d a n d la b e le d w ith 32P.
T h e lo w s tr in g e n c y o f th e
h y b i id iz a tio n in th e firs t fe w a u to r a d io g r a p h y s r e s u lte d in m o re h y b r id iz a tio n s ig n a ls th a n
w o u ld b e e x p e c te d u n d e r s u c h c ir c u m s ta n c e s i.e. a lo w c o p y B A C lib ra ry c o u p le d w ith
s in g le c o p y g e n e p r o b e ( d a ta n o t s h o w n ). A c c o r d in g ly , th e s tr in g e n c y o f th e re a c tio n w a s
in c r e a s e d to a h y b r id iz a tio n te m p e r a tu r e o f 5 5 °C to d e c r e a s e n o n - s p e c if ic b in d in g .
T h e p o o ls w e r e c o n s tr u c te d a s fo llo w s : P o o l I ) G sC ,
P628,
A p x l , D im in , Pool 2) P393,
G d c t, S s y n , a n d P o o l 3 ) s a m e as p o o l tw o , w ith o u t S s y n . E a c h o f th e n in e p ro b e s
w a s u s e d to s c r e e n th e c o m p le te l x lib ra ry w ith th e e x c e p tio n o f p r o b e
F ta s e and R p l2 2
w h ic h w e r e u s e d o n ly o n o n e filte r. T h u s e a c h p ro b e u s e d h a d a n e q u a l o p p o rtu n ity to
lo c a te th e s p e c if ic g e n e w ith in th e B A C lib ra ry . T h e fo llo w in g in f o r m a tio n c a n b e fo u n d
37
summarized in Table 2. The.following information can be found summarized in Table 2.
Seven probes revealed a total of 23 signals of convincing intensity. It should be noted
that this total number of hybridizations are not indicated on the Table 2 as the
hybridization signal received from the pools could not be associated with a particular
probe without PCR verification. Eight signals were received from Pool I, eleven froni
Pool 2 and two from Pool 3. PCR-based verification reduced the number of positive
BAC clones from twenty-three to eleven. Clones 23-P17 and SShElO both had
amplification products using the Gsc primers, clone 13-P3 using the P393 primers and '
clones 13-P3, 13-P12, 19-G10, 104-018, 49-L19, 64-C5 and 68"-J4 using Ssyn primers.,
As unique sequences should only be present in one clone from a lx library, the suspect
results attained from the PCR of the Ssyn primer set was further examined. Results
suggested that the direct colony PCR method of initially checking the clones was
responsible for the false positives. This conclusion was confirmed by demonstrating that
the Ssyn primers could amplify an identical product when tested on “empty” E. coli
clones (data not shown). Thus, it was determined that the Ssyn probe had to be discarded
from pool, and the library screening of Poof 2 was repeated using Pool 3, I was them able
to eliminate the clone identified by Ssyn which decreased the number of PCR verified
BACs containing a single copy genes, from eleven to three.
Sequence results of the PCR products from the three clones positively identified '
using probes Gsc and P393 indicated that only clone 89-E10 Contained sequences
homologous to Gsc or GS3A (Walker et. al. 1995) (See Figure 6). The sequence results
38
iio m th e o th e r tw o P C R p r o d u c ts fro m th e c lo n e s a m p lifie d w ith e i th e r
in d ic a te d th a t th e p r o d u c t c o n s is te d m o s tly o f r e p e titiv e
G s c o r P393
P is u m D N A .
F ig u re 6. P i s u m B A C f ilte r h y b r id iz e d w ith p o o l I d is p la y in g h y b r id iz a tio n sig n a l
fro m p r o b e G s c . In s e t: C lo s e - u p o f h y b rid iz a tio n s ig n a l.
T h e B A C lib r a r y r e s u lts c a n b e s u m m a riz e d a s fo llo w s :
1) A p p io x im a te ly 1 .0 % o f th e B A C s w ith in th e l x lib ra ry c o n ta in in s e r ts w ith s e q u e n c e
id e n tity h o m o lo g o u s to c h lo r o p la s tic D N A .
2 ) A p p r o x im a te ly 0 .3 5 % o f th e B A C s w ith in th e l x lib ra ry c o n ta in in s e r ts w ith s e q u e n c e
id e n tity h o m o lo g o u s to 2 5 s r ib o s o m a l D N A
3)
O n e B A C , 1 0 3 -K 1 w a s id e n tif ie d fro m th e l x lib ra ry , a s c o n ta in in g s e q u e n c e s
39
homologous to the Icnown gene family of Rubisco (small subunit).
I
^
89-El Ocontaining a single copy gene,
BAC library.
was identified from die lx
40
Discussion
The intent of this study was to identify clones containing different types of
nuclear and cytoplasmic DNA within a lx coverage Pisum BAC library. The results
would provide a direct indication of the quality .of the BAC library and reveal the
logistics of locating a unique sequence within this library. To initially,assay the library,three features of the BAC library were examined: I) contaminating chloroplast DNA 2)
abundance of rDNA sequences and 3) ability to locate clones containing both low copy
(gene family) and single copy sequences within the library. The three experiments to
address these three objectives were designed as follows. Chloroplast specific probes
were used in hybridization experiments to estimate the abundance of one component of
extra nuclear DNA within the BAC library, A rDNA probe was used in a similar
experiment, again to estimate the abundance of rDNA sequences within the library.
Probes derived from five known gene families and nine single copy genes were used to
screen for BAC clones with homologous sequences.
The chloroplast screening resulted in an estimate of 1.0% of the BAC clones
containing chloroplast sequences. Simple comparisons of the percent cpDNA
contamination determined for other BAC libraries constructed from agronomic crops
suggest that our library is of average chloroplast content. Reports of chloroplast content
are as follows: Solanum tuberosum 2.4% (Song et ah, 2000), Hordeum vulgare 1.5% (Yu
c
et ah, 2000), Medicago iruncatula 1.4% (Nam et ah, 1999), >1% for both Saccharum
Officinarum (Tomkins et ah, 1999) arid Pennisetum glaucum (Allouis et ah, 2001), These
preliminary results indicate that with respect to cpDNA contamination, the Pisum BAC ■
41
libraiy does not deviate significantly from other plant species. However, this estimate Pf
1.0% should be further confirmed using the following two experiments. The first
experiment should duplicate the previous screen at a higher annealing temperature to
reduce the potential of false positives. The second experiment should address the
possibility of any. chloroplast sequence remaining undetected as the trnl does not
iepiesent the entire cpDNA genome. Thus, a new probe should be designed from a
location opposite the trnl sequences within the circular chloroplast genome. A complete
assessment of extra nuclear or organellar DNA would also require the quantification of
DNA that also has been donated to the BAG library from mitochondria. This experiment
would be relatively simple and identical to the methods described for estimating
chloroplast content of the BAG library, with the exception of using a probe derived from
mitochondrial DNA.
An experiment comparable to that of the chloroplast screen was set up to quantify
the representation of rDNA sequences within the Pisum BAG library. It has been shown
that the 45s array has a haploid copy number of approximately 4,000 genes which
accounts for approximately 0.8% of the pea genome. (Jorgenson et ah, 1987). My results
indicate that approximately 0.35% of the BAG clones contain sequences homologous to
the 45 s array. A lx library would be difficult to compare the expected amount of rDNA
to the amount determined experimentally. Although in some cases statistics can be used
to correct for differing coverage between libraries, a lx library deviates significantly
from the coverage of a 4-5x coverage library and comparisons would be of limited value.
The results obtained from this experiment provided the first indication of the presence
and availability of nuclear D N A within the library
Upon the completion of the physical evaluation of the BAG library, that is, the
characteristics of the library directly affected by the construction of the library i.e. extra
nuclear DNA contamination and condition of the filters themselves, the library was
assessed for the representation of low and single copy geries. This evaluation is of
particular importance as the library will be used for positional/map-based cloning of both
single copy genes and gene family constituents such as anchor probes and disease
resistance homologs.
Of the gene families inspected only one clone was found that could be positively
identified through sequencing to contain a member of the Rubisco gene family. Previous
RFLP results using the Rubisco small subunit probe, indicated the presence of multiple
bands, most of which appeared to co-segregate to a single locus (Polans et ah, 1985).
More recent results suggested the presence of a duplicate genetic locus in a separate
linkage group (Ellis et al. 1992), however, these results have yet to be confirmed.
Moreover, the structure of the gene family, that is tandemly oriented pseudogenes and
transcribed genes, are generally represented within a single locus (Sugita et ah, 1991).
This arrangement indicates that the Rubisco gene family probes provide a well suited
intermediary between middle-high copy gene such,rDNA and the low copy gene such as
glutamine sythetase (Gsc). Thus, one can expect, even with low coverage libraries, to
detect the presence of one of the gene family members. As a tool for testing BAG library
■
'
43
I
quality, gene family probes can be used in the initial stages of construction, especially
with low coverage libraries where the likelihood of detecting a single copy gene is low.
An unexpected result of the BAC screening with gene family members was the
relatively large amount of false positives. Even a clone that was identified by a strong
hybridization signal when screening under stringent conditions failed PCR verification
(see Figure SB Chap. 2 Results section). It can be hypothesized that the reason for this
lies either within the acquirement of clones or the PCR verification procedure itself. The
obvious conclusion that could be made was that the wrong BAC was sent. This
deduction is unlikely, as there were two clones (89-El 0 and 103-K1) that were identified,
ordeied, and sent tested positive on all accounts. A more likely solution may be found in
the PCR verification procedure. A positive PCR result indicates that not only some
sequence homology exists between the probe and the BAC insert, but also verifies the
presence of uncompr.omised priming sites. Pseudogenes may produce hybridization
signal with intensities roughly equal to signal expected from the transcribed gene from
which the probe was derived. Pseudogenes are often associated with sequence
divergence from the original gene that may or may not include one or both of the priming
sites, thus leading to a failed PCR reaction. As an alternative to the PCR verification, one
could assemble a Southern blot of a digested BAC containing the putative gene family
insert and identify the band using a probe generated via PCR from genomic DNA as was
described in the results section. Additionally, well-supported labs could sequence the
entire BAC and unambiguously verify sequence homology to the probe sequence.
44
However, at this time the cost of sequencing and entire BAC is approximately $7000.00,
which is neither feasible nor worthwhile for most labs.
To assess the quality of the library for use in positional or map-based cloning it
was imperative to demonstrate that a single copy gene could be identified from the
library. The probability (P) of detecting a unique sequence within the lx coverage Piswn
BAC library was calculated to be approximately 74.8% using the following equation:
N = In (I - P ) / In (I - I / CS). This equation takes into account the size of the genome
(Pea GS = - 4000Mbp (Arumuganathan et ah, 1991)), the number of clones within the
■library (N = 50,000 (Coyne et ah, 2000)), and the average insert size within each clone (I
= ~110,OOObp (Coyne et al., 2000)) ( I Williams’ Group, Rbsilin Institute, Edinburgh)
(www.labs.roslin.as.ulc/jwilliam./bacequa.html).
A total of eight probes containing low copy sequences were used to identify
BACs. Only one probe identified BAC that was subsequently verified using PCR. This
BAC was clone 89-E10 which sequence results confirmed the presence of the Gsc
(cytolosolic glutamine synthetase) gene. Unfortunately, at the time of the single copy
screening my intentions were to locate genes anchored to linkage groups IV and VII from
the P. sativum consensus linkage map (Braunef et ah, 2002). Certainly there were many
more gene markers that could have been screened from outside these two linkage groups.
Interestingly, when screening for low copy genes the problem of finding falsd positives
was not seen to the extent that it was with the gene families. This is not to say that there
were no false positives, yet the ones that did turn out to be false did not display the signal
intensity under stringent conditions that was seen in the gene family screen. The false
45
positives that were observed were generally attributed to the low stringency used when
screening with low copy probes. However, a probe containing marker P393 produced a '
signal from the BAC library of convincing intensity and even tested positive for the
sequence presence via PCR. When sequenced, the results were similar to leghemoglobin,
in that the insert amplified by P393 showed sequence identity to a previously
characterized repetitive element from the pea genome. This result was not surprising and
represents a significant setback when screening the pea BAC library via filter
hybridization. The pea genome is extremely large-4000Mbp with estimates of
interspersed repetitive DNA exceeding 95% (Murray et ah, 1981). The hybridization of
clones does not seem to be able to discriminate between pseudogenes, short homologous
sequences and the true transcribed sequences of interest. Many researchers have gone to
BAC pooling systems (Nam et al, 1999) in which a three-dimensional configuration of
the microtitre plates containing the BAC clones are constructed. Using the architectural
arrangement of the plates, a systematic set of pools can be developed that would allow
the identification of a specific clone using PCR to screen the large pools. The specificity
of PCR would eliminate the clones identified from the nonhomologous filter
hybridization experiments, such as the resulting clones identified by the leghemoglobin
probe, and also permit the screening of the entire library within a few days. Conversely,
depending of the position of the priming sites, some of the clones that one would expect
to identify when screening the library may remain unidentified. Thus, if this approach
were adapted for routine use, it would be of importance to know the priming sites with
respect to intron and exon position, and proximity to the transcribed gene boundaries.
46
Literature cited
Allouis5S. X. Qi5 S. Lindup5 M.D. Gale5 K.M. Devos. (2001). Constuction of a BAC
library of pearl millet, Pennisetum glaucum. Theor. Appl. Genet. 102:1200-1205.
Ausubel5F.M, R. Brent5R.E. Kingston5D.D. Moore5LG. Seidman5J.A. Smith5K.
Slruhl. (1999) “Short Protocols in Molecular Biology” John Wiley and Sons Inc. New
Yorlc5New York.
Brauner S., R.L. Murphy5 LG. Walling, J. Przyborowski5 N.F. Weeden. (2002). STS
Markers for Comparative Mapping in Legumes. J. Amer. Soc. Hort Sci. 127 :616-622..
Cregan5P.B., J. Mudge5E.W. Fickus5L.F. Marek5D. Danesh5R. Denny5R.C.
Shoemaker5B.F. Matthews5T. Jarvik5N.D. Young. (1999). Targeted insolation of
simple sequence repeat markers through the use of bacterial artificial chromosomes.
Theor. Appl. Genet. 98: 919-928.
Coyne5C. K. Meksem5 D.A. Lightfoot5 K.E. Keller, K R 1Martin5 M.T. McClendon,
D. A. Inglis5 E.W. Storlie5 K.E. McPhee. (2000) Construction of bacterial artificial
chromosome library for pea (Pisum sativum L.) Ezram Genetics 32: 23-26.
Ellis5T.H.N., L. Tumer5KP. Hellens5D. Lee5C.L. Harker5C. Enard5C. Domoney5and
D.R. Davies. (1992). Linkage maps in pea. Genetics 130:649-663.
Fairbanks5DJ.., W.R. Anderson. (1999). “Genetics: The Continuity of Life”: Cole
Publishing Co. 1999. New York5New York.
DaneshD., S. Penuela5J. Mudge5R.L. Denny5H. Nordstrom5J.P. Martinez5N.D. Young.
(1998). A bacterial artificial chromosome library for soybean and identification of clones
near a major cyst nematode resistance gene. Theor. Appl. Genet. 96:196-202. ■
Jackson S.A., F. Dong5J. Jiang. (1999). Digital mapping of bacterial artificial
chromosomes by fluorescence in situ hybridization. Plant J. 17:581-587.
Jackson S.A., Z. Cheng5 M.L. Wang5 H.M. Goodman5 J. Jiang. (2000). Comparative
fluorescence in situ hybridization mapping of a 431-kb Arabidopsis thaliana bacterial
artificial chromosomes contig reveals the role of chromosomal duplications in the
expansion of the Brassica rapa genome. Genetics 156:833-838.
Jorgensen5RA., R.E. Cuellar, W.F. Thompson5TA. Kavanagh. (1987). Structure and
variation in ribosomal RNA genes of pea. Plant Molec. Biol. 8:3-12.
47
Ku, H.M., T. Vision, J. Liu, S.D. Tanksley. (2000). Comparing sequenced segments of
tomato and Arabisopsis genomes: Large-scale duplication followed by selective gene loss
creates a network of synteny. PNAS. 97:9121-9126.
Ladish, H., A. Berk, S.L. Zipursky, P. Matsudaira,.D. Baltimore, J. Darnell. “ Molecular
Cell Biology, 411 ed.” W.H. Freeman and Company 1999. New York, New York.
Miller, LT., S.A. Jackson, S. Nasuda, B.S. Gill, RA: Wangr Ji Jiang. (1998). Cloning
and characterization of a centromere-specific repetitive' DNA element from Sorghum
bicolor. Theor Appl. Genet. 96:832-839.
MuiTay, M., D. Peters, W.F. Thompson. (1981). Ancient repested sequences in the pea
and mung bean genomes and implications for genome evolution. J.Mol. EvoL 17:31-42
■ Nam Y.-W., R.V. Penmetsa, G. Endre, P. Uribe, D. Kim, D.R. Cook. (1999).
Construction of bacterial artificial chromosome library of Medicago truncatula and
identification of clones containing ethylene response genes. TheOr Appl. Genet. 98:638646.
.Polans N.O., N.F. Weeden, W.F. Thompson. (1985). Inheritance, organization, and
mapping of rbcs and cab multigene families in pea. PNAS 82:5083-5087.
Song, J., G. Dong, J. Jiang. (2000). Construction of a bacterial artificial chromosome
(BAC) library for potato molecular cytogenetics research. Genome. 43 :199-204.
Sugita, M., T: Manzara, E. Pichersku, A.R. Cashrnore, W. Gruissem. (1991). Genome
organization, sequence analysis, and expression of all five genes encoding the small
subunit of ribulose-1-5-bisphosphate carboxylase from tomato. Mol. Gen. Genet
209:247-256.
,
.
Taberlet, P., G. Ludovic, G. Pautou, J.Bouvet. (1991). Universal primers'for
amplification of three non-coding regions of chloroplast DNA. Plant Mol. Bio. 17' 11051109.
■
Tomldns5LP., Y. Yu, H. Miller-Smith, iD.A. Frisch, S.S. Woo, RA. Wing. (1999). A
bacterial artificial chromosome library for sugarcane.. Theor Appl Genet. 99:419-424.
Torres, A.M., N.F. Weeden, Ai Martin. (1993). Linkage among isozyme, RFLP, and
RAPE).markers in Vicia faba. Theor. Appl. Genet. 85:937-945.
Wang, G.L., W.Y. Song, L i . Chen, R. Rtian, S. Sideris, P C. Ronald. (1996). Construction of an Arabidopsis bacterial artificial chromosome library and isolation of
48
clones hybridizing with disease resistance gene-like sequences. Plant Molec. Biol.
Reporter. 14:107-114
Walker E. L., N.F. Weeden, C.B. Taylor, P. Green, G. M. Coruuzi. (1995). Molecular
evolution of duplicate copies of genes encoding cytosolic glutamine synthetase in Pisum
sativum. PlantMoL Bio. 29:1111-1125.
Woo, S.S., J. Jiang, B.S. Gill, A.H. Paterson, RA. Wing.. (1994): Construction and
characterization of a bacterial artificial chromosome library of Sorghum bicolor. NucL
Acids Res. 22:4922-4931.
Yu. Y., J.P. Tomkins, R. Waugh, DA. Frisch, D. Kundra, A. Kleinhofs, R.S.
Brueggman, G.J. Muehlbauer, R.P. Wise, RA. Wing. (2000). A bacterial artificial
chromosome libraiy for barley (Hordeum vulgare L.) and the identification of clones
containing putative resistance genes. Theor. AppL Genet. 101:1093-1099.
Zwick, M.S., MNL Islam'-Faradi, D.G. Czechin, Jr., RA, Wing. G E. Hart et al (1998).
Physical mapping of the liguleless linlcage group in Sorghum bicolor using rice selected
RFLP Sorghum BACs. Genetics 148:1983-1992.
CHAPTER 3
BAC FLUORESCENCE IN SITU HYBRIDIZATION ON PISUM SATIVUM
CHROMOSOMES
Literature Review
Intoduction-Cvtology in Pisum
Cytological investigations into the karyotype of Pisum sativum (2n=14) were
initiated in the early 1930s with the morphological characterization of the haploid set of
seven pea chromosomes (Lewitsky, 1931). The methodology used in this study quickly
became obsolete and improvements on chromosome isolation were made that were based
primarily on the use of oxyquinoline derivatives for chromosome preparations. These
treatments provided.a repeatable means superior to previous treatments (Caroli 1952).
Furthermore, a standard means for measuring chromosome structure with respect to total.
chromosome length, and arm ratios was developed. The intent of the experimental series
was to establish a standard karyotype which would represent a consensus “normal
karyotype” to which other pea karyotypes could be cytologically compared (Caroli and
Blixt, 1953 , 1954, 1955, and Blixt 1955). The results of these experiments indicated the
line originally deemed the standard karyotype, Kloster pea, was not representative of the
normal karyotype. Thus, a systematic examination of chromosome measuring techniques
and refinement of such techniques again improved the reliability of results from
chromosome characterization studies (Blixt, 1958a). An additional outcome of these
experiments provided evidence to designate Pisum line LI 10 as the ‘standard’ karyotype
(Blixt, 1958b). The Pisum chromosome karyotype underwent several numerical changes
50
throughout the duration of these experiments; however, a comparison of five interchange
lines to the standard karyotype allowed the assignment of a numerical designator to each
chromosome. The chromosome numbers were based on linkage groups
o r i g i n a l ly
described by Lamprecht (Blixt, ,1959). Chromosomes were numerically identified with
respect to total chromosome length, arm ratios and location of primary and secondary
constrictions! This chromosome numbering system provided a standard to which all subsequent karyotype studies could be compared.
.Cytological Markers .
The assignment of numerical designators to chromosomes provides a standard
means for presenting data and results based on karyotypic studies. However, to fully
exploit the power of cytology in generic studies, chromosome markers were introduced to
provide a logistical means of identifying both whole and portions of chromosomes.
Cytological markers can be used alone to identify a particular chromosome or used in
conjunction with translocation stocks to map chromosome content and rearrangements
(Hall et ah, 1997b; Lamm et ah, 1958).
The first and arguably most accessible cytological markers are .chromosome
morphological markers. These consist of variations in chromosome size, number, and
location of primary (centromeres) and/or secondary constrictions (satellites). The
karyotype of P. sativum was compared to that of P.fulvum using morphological markers.
This comparison was facilitated using a chromosome image analysis system (Errico et ah,
1990). The authors were able to capitalize on the resolution and efficiency by which
51
karyotypic data could be generated using the image analysis system and detected
numerous novel polymorphism's between chromosome morphological markers in i3.
Julvum and P. sativum. An additional chromosome, satellite or secondary constriction
was found on chromosome 6 in P. fulvum that is not present in P. sativum. Chromosome
7 was satellited in both karyotypes, however, P. fulvum was shown to have a satellite
significantly larger that that found in P. sativum. Thus, the authors were able to
positively confirm, using chromosome morphological markers, the proposed hypothesis
suggesting a karyotypic dissimilarity between the two species.
Chromosome morphological markers are of limited value and exist only in
species that have physically distinguishable chromosomes; consequently, a method was
developed that could distinguish chromosomes based on the genetic content rather than
the structure. Chromosome banding was developed to allow positive identification of
chromosomes based on the staining pattern displayed by each chromosome. C-banding
with Geisma stains differentially bands areas of the chromosomes permitting
.
identification of individual chromosomes, chromosome parts, and breakpoints of
chromosomal rearrangements. Lamm (1981) describes the BSG (Geisma) C-band and
silver staining of chromosomes from Pisum both in meiotic and mitotically-derived
chromosomes. Chromosomes were characterized based on their C-banding pattern and
accumulation of silver staining. Interestingly, karyotypic results differed from those
previously compiled by Blixt (1972) with respect to amrleg ratios of the non-satellited
chromosomes, indicating discrepancies in the characterization of chromosome
morphology. The banding characterization of the seven chromosomes in this experiment
52
indicated that the resolution of C-banding and silver staining of Pisum was insufficient to
provide usable markers for mapping, and thus the discrepancies could not be elucidated.
.
In Situ Sequence Detection
The eytological markers of chromosome morphology and banding patterns can
reveal information based chromosome differentiation and content, However, with
advancements in DNA technology such as genetic cloning and DNAiDNA hybridization
experiments, a more targeted approach to eytological markers became feasible. Gall and
Pardue (1969) were able to detect RNA sequences on intact eytological preparations of
Xenopus using radiolabeled ribosomal RNA sequences as probes for in situ, hybridization'
(ISH). The oocytes of Xenopus were isolated in an agar matrix and the extranuclear
rDNA in meiotic nuclei was targeted with rRNA probes. Autoradiographs of the .
radioactive signal were superimposed over images of stained nuclei,, thus revealing the
chromosomal positions of extranuclear ribosomal DNA. Furthermore, the authors were
able to detect changes in the abundance and position of rDNA throughout the stages of
meiosis, indicating variability in the transcription of ribosomal genes during meiosis.
Intuitively, hybridization of homologous sequences is not limited to RNA/DNA
heteroduplexes, rather DNA/DNA heteroduplex formation is used experimentally quite
routinely (Southern, 1975). Shubert et al. (1985) used in situ hybridization of DNA
probes to target homologous DNA targets in situ. Radiolabeled rDNA probes were
hybridized to prepared metaphase chromosomes of Allium. The nuclear organizer region
(NOR) was investigated for transposition using rDNA probes to localize the NOR to
53
specific chromosomal regions in several species of Allium. These results corroborated
previous silver staining results that indicated that the NOR region may possess mobile
characteristics. While silver staining would only tag actively transcribed NORs, ISH ■
detects transcriptionally active and inactive rDNA. The results clearly substantiated the
hypothesis of the mobility of NORs in Allium sp. and further promoted the use of ISH to
effectively target intact nuclear sequences. In situ hybridization or ISH dramatically
increased the power of cytology and arguably was the factor that has rejuvenated
cytology as an integral tool in many genetic studies.
Although the preliminary studies involving ISH showed great potential for this
technique, sequence detection was limited by the resolution and monochromatic nature of
sequences labeled with radioactivity. The development of fluorescence as a method for
sequence detection greatly improved the ability to detect and resolve hybridization
patterns (Heslop-Harrison et ah, 1991). Like ISH, Fluorescence In Situ Hybridization
(FISH) can be used to .detect sequences on cytological preparations by hybridizing a
labeled probe sequence to target DNA on the chromosome. FISH employs probe
sequences that are either initially labeled with fluorescence and detected directly using
fluorescence microscopy or are visualized indirectly by first labeling the probe with an
antibody and detecting hybridization with the fluorescently labeled antibody conjugate.
. Abbo et ah (1994) used FISH to reveal the locations and quantity of rDNA arrays
of both wild and cultivated Species of lentil (Lens) and chickpea (Cicer). Wheat probes
containing the 18s, 5.8s, and 26s were labeled with digoxigennin-11-dUTP and
hybridized to metaphase chromosomes preparation. Two species of lentil, (L. culinaris
54
and L. orientalis), displayed two signals indicating one 45s locus. The cultivated species
of chickpea (C arietinum) had three pairs of rDNA sites; however, only two were
detectable in the wild species (C. reticulum), indicating a karyotypic rearrangement
between the two species.
Data generated using FISH of repetitive sequences-such as the ribosomal repeats
can be used to investigate the evolution of species and species divergence. Krishnan et
al. (2001) addressed the possibility of an ancient polyploidization event occurring in wild
species of Glycine. The cultivated species of Glycine (G. max) has been postulated to
have undergone an allotetraploidization event followed by diploidization; however, wild
species had not been investigated, cytologically to confirm such a phenomenon. By using
FISH to physically map the 5s and l8s-28s rDNA loci in each species, the authors were
able to compare the number of signals received when each species was tested with the
rDNA probes. Variability of hybridization patterns in different wild Glycine sp. indicated
a loss of a 5s rDNA locus in the diploid species and a loss of two in the tetraploids. In .
addition, the 18s-28s rDNA displayed two signals per haploid genome rather than the
expected four. These results indicate that the formation of the cultivated tetraploid did
involve a conventional double diploidization model, but must have occurred through a
more complicated process.
Probe sequence characteristics, (i.e. length, copy number and flanking sequences),
can have profound implications on the types of experiment conducted as well as the
logistics of accomplishing such as task. The reviews of FISH experiments discussed until
now have used rDNA or rRNA probes to detect homologous sequences on isolated
55
chromosomes. Early DNA denaturation/reannealing experiments have shown that the
,
DNA content of chromosomes can be arranged into three groups: highly repetitive,moderately repetitive and low/s'ingle copy elements (Murray et ah, 1978). The nature of
visualizing sequences on Condensed chromosomes favor sequences that exist in higher
copy numbers rather than low copy numbers. The greater number of target loci and the
more localized the hybridization targets are, the more intense the signal will be. In
systems such as Pisum, however, this notion can be misleading given that the many
highly repetitive elements are interspersed throughout the genome, rather than focused
within specific regions of the genome. Elements that are known.to exist in tandem
(rDNA) or defined locations (centromcric DNA) are generally considered the easiest
FISH targets. The use of rDNA probes is of limited value given that there are only a few
loci in the genome. Considerably greater coverage could be achieved by applying this
technique to lower copy gene sequences.
The use of low copy sequences to physically map seed protein genes displayed
the limitations of such an approach (Fuchs et ah, 1995). Three separate members of the
legumin gene family (Legumin A, B3, and B4) and a seed non-storage protein (USP)
were used in FISH experiments on metaphase chromosomes in Vicia faba. The seed
protein genes can be characterized as gene sub-family constituents, meaning that they
share sequence; identity with each other and typically are limited to fewer than ten copies
at a single locus. Thus, these sequences should provide superior FISH probes. FISH
probes of the legumin and USP genes were generated from either cDNA or genomic
clones with sizesjanging from 1.7kb to 3.5kb. All three seed storage protein genes were
56
localized to separate chromosomes, indicating that the progenitor legumin gene had been
duplicated and rearranged within the genome. However, the USP genomic DNA probe
(3.5kb), produced multiple dispersed hybridization signals presumably due to a 2.8kb
sequence of repetitive DNA flanking the gene. Furthermore, xFl a pseudogene of.
legumin B4, sharing 97% sequence identity to the probe sequence could not be detected
due to deletions that reduced the target size to less than I kb. Both results indicate that
ihsert/target size and composition of the insert play a crucial role for sequence detection
in situ.
■ An additional factor that must be considered for in the pursuit of in situ sequence
detection is the condition of the chromatin. The dynamics of chromatin during somatic
cell division govern the state of chromatin condensation. Condensed chromatin, while
providing a superior karyotypic template, can physically hinder probe access to target
DNA. Consequently, treatments that control the condition of the chromatin during the
chromosome preparation must be refined to allow probe access to the target sequence,
while retaining, chromosome characteristics required for karyotyping. Jiang et al. (1996)
devised a method for detecting sequence signal on interphase chromatin. Less
compaction of chromatin permits a resolving power at least two magnitudes higher than
that of condensed metaphase chromosomes. Obviously, this approach cannot be directly
applied to studies that intend to place a gene on specific chromosome; however, the
number and position of the signals visualized can confirm presence and copy number of a
particular sequence. In addition, the authors were able to quantitatively correlate the .
57
■ physical interphase distance with that of linear distance as long as the probe sequences
were within IMb of each other.
Alternatively, the attributes of meiotically-derived chromosomes at pachytene
stage have been exploited in high resolution in situ mapping (Cheng et ah, 2001).
Significantly, meiotic chromosomes are often ten to forty times longer than mitotic
chromosomes. They also are grouped into the haploid number of bivalents. Thus meiotic
■preparations often provide superior FISH targets. Genetically anchored BAC clones
along chromosome 10 office (Oryza sativd) were used to examine the distribution of
recombination along this chromosome. The restricted chromosome number and the high
resolving power allowed the investigators to localize several genetically anchored
markers on pachytene chromosomes, spreads. This resolution allowed the genetic and
physical maps to be fully integrated and correlated.
In Situ Sequence Detection in Pisum
'
The shear size and complexity of the pea genome pose a challenge for detecting.
low copy sequences on isolated chromosomes. Simpson et al. (1988) established
procedure for FISH in Pisum by refining the FISH technique, including the use of
protoplasts for isolating chromosome. This result led to the verification of a single
position of a 13.5kb legumin genomic clone on metaphase pea chromosomes.
Subsequently, a similar FISH method was used to identify a reeipfocal translocation in
Pisum using biotin labeled tandemly repeated probe sequences (Simpson et al., 1990).
The addition of a signal site associated with a particular chromosome indicated a
translocation differing from the standard karyotype. These results provided
supplementary cytological markers that could be assigned to some of the chromosomes
FISH with rDNA (5s and 25s), multicopy (&%%/%;%and wczfm) and simple repeat
(AG12, AC12, GAA12, GATA10) probes allowed the discrimination of most of the pea
chromosomes (Fuchs et ah, 1998). Furthermore, the rDNA and gene family sequences
had been mapped to specific linkage groups allowing the assignment of five out of seven
of the linkage groups to specific chromosomes, with chromosome 4 and 7 and linkage
groups IV and VII remaining unassociated.
Neuman et al. (2001) developed a series of chromosome identifying probes by
screening for unique hybridization signals from FISH experiments using isolated
repetitive sequences from Pisum Cyt fractions. The authors used probe PisTR-B in
conjunction with a probe indicating the position of the 5s rDNA array to identify each
chromosome under fluorescence microscopy based on the unique signal pattern bestowed
by the co-hybridization of the two probes .
In what turned into an elegant series of experiments leading to the definitive
association of each linkage group with each pea chromosome, Neuman and colleagues
later used pea interchange lines in conjunction with the chromosome identifier probe and
Fluorescence Activated Cell Sofrer (FACS) to nncover the linkage group identity o f each
chromosome. The chromosomes of interchange lines were hybridized with one of the
chromosome identifier probes and subject to FACS. PCR amplification o f chromosome
pools using genetically anchored probes, allowed the assignment of linkage groups to all
seven chromosomes (Neuman et al., 2002):
BAC-FISH
Physical mapping of chromosomes has become a routine method for fine mapping
genes to chromosomes. In addition, physical maps are blind to obstacles inherent to
genetic mapping, such as attempting to fine map areas that .display suppression of
recombination or in experiments using small sample sizes. One popular method for
physically mapping a region is by piecing together large cloned portions ofDNA. Large
insert libraries can be screened for adjacent and overlapping inserts, in effect creating a
contiguous chromosome or contig (Mozo et ah, 1999). Chromosome walking through
contig assembly is typically considered too difficult if only smaller insert, clones such as
cosmid and plasmid libraries are available. The small insert size of the cosmid and
plasmid clones would cause great difficulty in attempting span, or ‘walk across’ large
areas of repetitive DNA that are devoid of anchor markers. In addition, when using these
clones as FISH probes, it can.be difficult to resolve low copy (<5) or single-copy genes
on condensed chromosomes with reproducible results.. Large insert libraries such as
BAC and YAC libraries have been developed in many species (See Chapter 2 Literature
Review). Ultimately, the assembly of a contig that spans an entire chromosome would
definitively establish both the sequence and genetic arrangement of the chromosome.
Mozo et al. (1999) illustrate the utility of using large insert clones as FISH probes to
distribute physical markers along a chromosome. The results made it possible to
estimate physical distance and close gaps between clones as well as to physically localize
clones on chroriiosomes.
60
Jackson et al (1998) demonstrated the applicability of using. BAC-FISH to
estimate contig and gap size between contigs on physical maps. Localizing fluorescently
IabeledBAC contigs on extended DNA Gbefs (Fiber-FISH) of
permitted micro measurements of hybridization signal that could be correlated with
sequence length. One inherent difficulty in physical mapping through contig construction
can be closing gaps between contigs, thus the ability to quantify such a distance would
provide valuable logistical data. BACs from contigs bordering gaps were isolated and
hybridized to extended DNA fibers. Micro-measurements of the area between the
hybridization signals indicated the distance of the gaps validating BAC-FISH as a <
powerful tool in genome studies.
Kulilova et al. (2001) constructed a cytological map of Medicctgd truncaiulci
■
based on chromosome length, centromere position and FISH signals of rDNA probes.
Subsequently, this cytological map was integrated with the genetic map using additional
FISH experiments by associating 2-5 single copy BACs per chromosome with the
previously described cytological markers/ The distance between FISH signals was
estimated by comparing microscopic distance (micrometers) to contig length (basepairs).
•
- Clearly, detecting sequences in situ via the application of FISH using large insert
'
probes provides a powerful tool to explore genome organization. Recently, a BAC
library has been developed in Pisum, representing a lx coverage of the Pdsum genome
(See Chapter 2). However, prior to instituting large-scale experiments using BAC-FISH,
the feasibility of such an approach on the large and complex genome o f Pisum must be
assessed. Of particular interest is the utility of large insert probes in FISH experiments
61
especially considering the excessive amount of dispersed repetitive sequence within the
pea genome. BAC probes identified 6om the BAC library as containing either single
copy oi gene family (multicopy) were used to probe metaphase chromosome spreads.
Additionally, small insert probes containing rDNA repetitive elements previously
■
localized were used as positive checks. It is hypothesized that FISH will allow us to
localize BAC probes, containing either single copy or gene family sequences, on
metaphase chromosome spreads of Pisum.
62
Material and Methods
Chromosomes were isolated from P. sativum cv. ‘Sparkle’, which.possesses the
Standard karyotype for the species as originally described by Blixt (1953). The lentil
chromosomes were isolated from L. culinaris as described by Tadmor et al. (1987) and
Abbo et al. (1995). The objectives when isolating chromosomes for use as FISH targets
are distinct from those corresponding to karyotypic studies. A morphology exhibiting a ;
complete condensation of chromatin is necessary for karyotyping chromosomes based on
physical characteristics; consequently, treatments were employed that maximize the ■
chromosome condensation. However, highly condensed chromosomes may be a
hindrance to the FISH reaction by physically sequestering sites.. Thus, treatments must
be altered to control the state of chromosome condensation. Furthermore, cytoplasmic
cellular debris, although aesthetically unfavorable, typically is not a hindrance to
karyotypic studies. For FISH analysis, cellular debris in close proximately to the
hybridization target will produce background and hinder the probe’s access to target and
must therefore be minimized. The following is the protocol developed specifically for
FISH on mitotic chromosome preparations of P. sativum.
Harvesting and Treatment of Pisum Root Tips for.Cvtological Preparations
Pea seeds {P. sativum cv. ‘Sparkle’) were germinated in an artificial soil medium
in 6” (15.2cm.) pots (3 seeds/pot) for approximately one week. The germination time was
optimized to allow sufficient growth of secondary roots but not long enough to produce
63
pot bound roots. It was determined that emergence of the first set of leaflets signaled
time of root tip harvest.
To empty the soil plug from the pot, the pot was inverted and tapped smartly on
the bottom. The root system from each plant was carefully dissected away from the
potting mix. Both primary and secondary roots were sampled.
The root tips were excised approximately 2-3 cm distal to the root tip with a
scalpel. The root tips could be excised closer to the end; however, this unused portion,
acts as a buffer zone to subsequent manipulations of the root tip that may harm the
meristematic tissue.
The excised root tips were placed in an ice-cold ddH20 bath in a petti dish until
they could be processed further (up to 24 hours). The root tips were rinsed 2x for 5
minutes in water to wash off extraneous soil debris. A spindle-fiber inhibitor was
employed to facilitate the accumulation of metaphase chromosomes located in the
meristematic portion of the root tip. Many spindle fiber inhibitors exist: cold treatment,
colchicine, hydroxyquinoline, and all have shown success in accumulating metaphase
chromosomes in different species. I found that 2mM 8-hydroxyquinoline produced a
high quality chromosome morphology without the over-condensing of chromatin.
Empirical evidence generated with time course experiments suggests that these results
can be produced if the root tips are treated in the 2mM hydroxyquinoline for
approximately 4 hours in the dark.
.
Spindle-fiber treatment was rinsed off by washing the root tips 2 x 5min in
ddH20. To preserve the chromosome morphology provided by the previous treatments,.
64
it was necessary to fix the cells. Fixation was accomplished by immersing the root tips in
freshly made Carnoy’s solution (3:1 absolute ethanohglacial acetic acid) and incubating
for I week at 37°C to allow sufficient time for fixation. -Fixed root tips can be stored up
to one year in this solution.
Chromosome Pretreatment.
:
.
■
The fixative must be removed prior to cytological work. For this reason the root
tips were washed 2x for 5 minutes each in ddFFO. The presence of fixative still on the
roots is evident by the movement of the root tips in the water caused by the hydrophobic
interactions between the fixed root tips and the water. Movement subsides when the
fixative is completely removed. Rinsed root tips were placed in a microfuge tube
containing a 5OOpl aliquot of a cell wall digestion cocktail consisting of 1% eellulase/1%
pectinase in water and incubated for 35 minutes at 37°C. Time of this digestion is truly
species and root type specific. If the species has tips that are large and fibrous it may be
necessary to treat longer; however, there are many species that require no digestion.
Again the tips were rinsed 2x for 5 minutes each in ddH20 to remove digestion
cocktail. Clearing root tips in 45% acetic acid (v/v) for approximately 8-10 minutes
turned most cell debris translucent and consequently reduced the appearance of artifact
cell material debris under microscopy.. To assist in visualizing cytological preparations,
the nuclear material was stained with either aceto-carmine (Carolina Biological Supply
Company) or aceto-orcein for 10-3 Omin, depending on the level of staining desired.
When a quality phase-contrast scope was used to inspect preparations, the use of a
65
chromosome stain such as aceto-carmine was not necessary. My findings suggest that
aceto-orcein may have auto fluorescence properties, which may affect the level
background noise in FISH.
Chromosome Isolation .
Root tips were removed individually from the staining solution and placed on a
clean slide under a dissecting microscope. To rinse the root of excess stain and prevent
drying, I drop of 45% acetic acid was applied to the foot tip on the slide and incubated
for I minute. The location of the meristematic tissue was obvious from the previous
staining treatment as it stained a darker red than the rest of the root material. The root
cap was first excised from the specimen. The root cap appears as the minute portion at
the apex of the root tip, just distal the meristematic tissue. A small cross section of the
root tip containing the meristematic tissue was isolated from the rest of the root material
using a razor blade and placed on a clean microscope slide. An additional drop of 45%
acetic acid was applied to further rinse the specimen. Superior spreading of the
chromosomes was achieved when the smallest possible slice was used. When possible
the rest of the root was saved, as it is feasible to make many slides from one root as long
small pieces of meristematic tissue are used. Excess liquid was removed using Kimwipes'
and a final drop of 45% acetic acid was applied to the sample in preparation for squash,
A double-edged razor blade was used to separate one comer of the cdverslip from
the slide. By tightly pressing a finger down on one comer (opposite to the one with the
razorblade) and not letting the cover slip move, a flex is created within the coverslip that
66
in concert with light tapping with a blunt instrument efficiently spreads the cell material
throughout the coverslip area. It was imperative to minimize any movement of the cover
slip during the squash as chromosomes will ‘roll’, ruining chromosome morphology.
The objective was to spread out the cell; however, care was taken not to push the cell
material out from under the cover slip. The absence of visible red' (although a pink haze
is acceptable) material is indicative of a quality spread. When the spread was deemed
complete, the razor blade was removed, and an additional five to ten taps on top of the
coverslip completed the spread.. The strength of the tap was not as important as the
complete coverage of the slide. Tapping commenced in the middle of the slide and in a
I
circular motion, progressed toward the outer edges of the cover slip.
.
The slide was warmed over a small alcohol lamp until it felt uncomfortably
warm, and hand pressure was applied to the coverslip over a piece a Whatman paper
effectively removing liquid from under the coverslip as well as affixing the coverslip to
the slide. It is important not to ‘boil’ the slide. The slide was briefly scanned under
phase contrast using a 40x objective, for presence.of chromosomes to determine the
quality and quantity of the spread. A good slide would consist of many chromosome
spreads with good morphology, free of cell debris. Typically, depending on the roots tips
and species, anywhere from 50-75% of the slides produced were discarded simply
because they were not high enough quality for FISH. Slides were stored at -SO0C until
used for FISH.
67
Isolation and Nick Translation of BACs
Pisum BAC clones,-previously identified from a lx library, were selected for
FISH on the basis of copy number and availability (See Chapter 2 Table 2). BAC clones
were allowed to grow for 24 hours at 37°C on LB agar plates supplemented with
tetracycline (20pg/ml). Individual colonies were selected and used to inoculate a liquid
LB culture, the volume of which was dependent on the vector and the method of
extraction used. BAC or plasmid DNA was extracted and purified using either Qiagen
“Large Construct Kit”™ or “Miniprep Kit”™, respectively and according to
manufacturers instructions. The concentration of the purified BAC was determined by
creating a serial dilution series of extract product i.e. lx, O.lx, 0.0 lx, 0.00 lx and run next
to either lambda DNA of Icnown concentrations or “EZ Load™ Precision Molecular
Mass Standard” (BIORAD). One microgram of plasmid/BAC DNA was required for one
nick translation reaction.
The following protocol (Jiang et ah, 1996) was adapted for use in nick translating
BACs with labeled nucleotides. Incorporation of fluorescently labeled nucleotides is
done using the DNA nicking ability of DNase I coupled with the gap filling capability of
DNA polymerase I. It was imperative to initially determine the activity of the DNase in
our hands, before a nick translation could be accomplished. The DNase activity was . ■
determined by digesting Ipg lambda DNA with a serial dilution of DNase I (Promega­
same dilutions as above) at 15°C for 2 hours in nick translation buffer. The reaction was
68
stopped with the addition of 5pi 0.2M EDTA (pH 8.0). Reaction products were analyzed
using gel electrophoresis, and a dilution was selected that yields DNA fragments of
between 300-600bp. The nick translation reaction was set-up as follows:
MasterMix (pl/rxn)
DNase
I
Polymerase I Buff.
5
Polymerase I
I
0.5mM ACG nucs.
5
d-UTP/TTP(dig/bio.)1 5
DNA
OS to 5Oul w/ddH20
x(Ipg)
x
Total: 5Opl
Reactions were cleaned of unincorporated nucleotides using a Qiagen “PCR spin
column” according to manufacturer’s procedure and eluted in minimal buffer. Labeled
products were stored at'-20PC until further use.
C flt-1 Blocking DNA Isolation
Concentration of genomic DNA from P. .sativum was determined Using a
spectrophotometer (BioRad MicroSpec®) by taking absorbency readings at 260 and
280nm. Genomic DNA from P. sativum was then sheared to an average size of 3OObp. ■
The shearing was accomplished using a sonicatdr by applying the genomic DNA to the
sonicating probe for 45 seconds to I minute. Gel electrophoresis was.used to determine
the amount of shearing that occurred. Shearing was deemed complete when gel
69
electrophoresis displayed a smear o'fDNA, spanning from IOObp to 600bp with the
majority residing around the 3OObp mark (data not shown).
Annealing time to allow for a Cot of I was determined using the following
equation developed by (Jiang et ah, 1996).
Time to anneal: 5.92/conc.(mg/ml)=time in minutes.
Annealing to Q t-I was accomplished by placing sheared DNA in a 65°C water bath to
equilibrate. A IOmM NaCl solution, prewarmed to 65°C was then added to a final
concentration of 0.3M and allowed to incubate for the duration of the calculated
reannealing time. To digest the residual single stranded DNA, SI nuclease (lunit/mg
starting material) and I volume of 2x SI nuclease buffer (0.5M NaCl, 2mM ZnSO4,
0.06M NaAc (piI 4.2), 10%glycerol) were added to the solution and incubated at 37°C
for 30 minutes. Reannealed products were phenol/chloroform extracted and recovery
was estimated again using the spectrophotometer. As experiments require differing ,
amounts of blocking DNA, the samples were prepared for a: specific reaction by drying
down the required amount of blocking DNA in a lyophilizer.
Fluorescence In Situ Hybridization (FISH)
The protocol originally described by Timing Jiang (1996) has been adapted for use
in P. sativum to allow detection of labeled BAC via fluorescence in situ hybridization to
metaphase chromosomes.
>
Cytological preparations were stored at -SO0C and removed just prior to
hybridization experiments. The coverslips were removed from the slides by setting the
.
70
prepared slide on a piece of dry ice until the liquid turned opaque indicating the 45%
acetic acid solution had frozen. Immediately the coverslip was flicked off by inserting a
single edged razorblade under a comer of the coverslip and rapidly lifting straight up. To
.maintain the appearance of the chromosomes it was important to prevent the liquid from
thawing and subsequent rehydration of chromosomes. Thus, the slides were subjected to
an ethanol dehydration series by placing slides in a Coplin jar containing 70% ETOH at
room temperature for 5 minutes. The first wash was followed by a 90% ETOH rinse at
room temperature for 5 minutes and then a 100% ETOH rinse at room temperature for a
final 5 minutes. Slides were air dried on a bench top. The hybridization reaction was set­
up as follows:
Final Vol./Conc.
MASTER MIX (units= pl/slide)
50%
Formamide
5,(high quality deionized is preferred)
10% ■
Dextran Sulfate
2
20xSSC
I
2x
Salmon Sperrn
■~17ng
Probe DNA
Total: IOpl
. I (Sheared and denatured)
I (If Cyt-I blocking is needed, add
to probe DNA and dry down to I pi)
The probe DNA as well as the target DNA fixed on the slide were denatured
separately. .
Slide: IOOpl of 70% formamide in 2xSSC was placed over the chromosome
spread and covered with a 22x40mm cover slip. Each slide was incubated on a hot plate
at 90°C for exactly 90 seconds. The slides were immediately immersed in 70% ethanol
(prechilled to -20°C) for 5 minutes allowing, the cover slip to fall off in the ethanol bath.
71
In the same fashion as the previous ethanol series, this step was followed by 90% ethanol,
and 100% ethanol washes, but at -20° C. Then a final rinse 100% ethanol at room
temperature for 5 minutes. The slides were allowed to air dry at room temperature on a
paper towel.
Probe: The probe DNA was denatured by incubating the sample at 90°C for 10
minutes followed by immediate submersion in ice bath preventing reannealing.
Ten microliters of MasterMix was applied to the area of the slide containing the
chromosome spread and covered with an 18x18mm cover slip. Edges were sealed with
rubber cement to prevent evaporation. Hybridization conditions consisted of incubation
at 37° for 6 hours to overnight in a moist chamber. No apparent difference in
hybridization was noticed with increased hybridization times.
Slides were removed from the moist chamber and without moving cover slip the
line of rubber cement was peeled away. Slides were then washed in a series of stringency
washes consisting of a 5 minute incubation at room temperature in 2x SSC in which the
coverslips were allowed to fall off. The washes were followed by three additional
washes in 2x SSC. However, the second wash employed an increase in temperature to
42°C and an increase in incubation time to 10 minutes. The third wash was identical to
the first wash. The final wash was in lx Phosphate Buffered Saline (PBS).
Secondary Antibody Detection
72
An appropriate fluoresceritly-labeled secondary antibody was chosen that would
form a conjugate with the biotin- or digoxigenin-labeled probe. Typically, to detect
biotin-labeled probes, strepavidin conjugated with FITC was used. To detect
digoxigenin, a mouse anti-digoxigenin conjugated to rhodomine was Used. Intuitively,
when detecting two differently labeled probes on same set of chromosomes, it was
necessary to choose fluorescence that had different emission wavelengths with respect to
each other to permit differentiation of the separate probes. Secondary antibodies were
allowed to form conjugates according to the following protocol:
MASTERMIX (units=pl/slide):
5x Buffer
20 (stock:5xPBS,25mMEDTA,5%BSA)
ddH20
80
2° Antibody
1-2 !manufacture’s directions)
Total:« lOOul/slidc
One hundred microliters of the antibody cocktail was placed on each slide, above
the qhromosome spread and covered with a 22x40mm coverslip. The slides were
incubated in a moist chamber in the dark for 30 minutes at 37°C. An initial wash in lx
PBS was administered and the coverslip was allowed to float off. The slides were '
washed 3 minutes at room temperature in lx PBS. Washes were performed in a dark
chamber as the fluorescence will fade over time when exposed to light. Excess PBS was
removed by shaking the slides in the air, but the slides were not allowed to dry. One drop
of Veotashield™ antifade/mounting solution containing either propidium iodide (red) or
DAPI (blue) was applied to each slide to protect the fluorescence signal from fading, and
to counterstain the chromatin a different color than the probc(s).
’
73
An 18x18mm coverslip was placed on the slides and excess Vectashield was
removed with Whatman paper. Slides were visualized immediately under fluorescence
microscopy or stored at -20C.
Fluorescence Microscopy
;
.
.
■
Hybridization was detected visually using a fluorescent microscope (Olympus BX
.
51 Microscope). Typically a 40x objective was used to scan for the presence of
chromosomes under normal phase contrast. Fluorescence was detected and images'were
captured with a 60x objective using a mercury bulb as the light source coupled with a
selectable wavelength filter adapted to the wavelength fluorescence of desired. Each of
the fluorochromes was captured in monochrome using a Charged Coupled Device (CCD)
camera (Olympus MagnaFire CCD), using MagnaFIRE (v.2.1 Universal Imaging '
Corporation) real time imaging program. Images of both probe and counterstain signal
were imported into Adobe Photoshop 6.0 where the images were adjusted with respect to
field of view and contrast. The images that coincide with each other were each assigned
to a color channel within Adobe Photoshop! The color channels from each FISH reaction
were merged to form a consensus image (See Figure 7 A, B and C).
C
Figure 7. A) Monochrome image of probe signal. B) Monochome image of chromosome
counter-stain signal. C) Images A and B merged through color channels (green and blue
respectively.
75
Results
Accumulation of Metaphase Chromosomes
Time course experiments utilizing two different spindle fiber inhibitors were used
to test for the production of an optimal accumulation of chromatin in the form of
individual condensed metaphase chromosomes. As briefly described in the FISH
materials and methods section, it was empirically determined that 8-hydroxyquinoline
treatments as opposed to treatments using colchicine or cold incubations were superior
for producing chromosomes that were morphologically identifiable, appropriately
condensed, and easily separated from each other. Examples of chromosome spreads
typically attained when the protocol described in the Pisum FISH material and methods
section was followed can be seen in Figure 8 (A & B). These chromosomes display the
distinguishing features of differing arm length ratios and easily identifiable NOR II0
restrictions on chromosomes 4 and 7. Four of the diploid fourteen chromosomes appear
submetacentric (chromosomes I & 2 (Neuman et ah, 2002) ) and the other ten are
acrocentric. Of the acrocentric chromosomes four contain secondary restriction points
(chromosomes 4 & 7). The time course experiment was repeated using colchicine as the
spindle fiber inhibitor. It was found that colchicine produced a general metaphase
morphology that was overly condensed and produced results with greater inconsistency
than those received from the hydroxyquinolone treatment (data not shown).
76
Figure 8. Images A and B represent the Piswn standard chromosome morphology as
seen under phase contrast microscopy. 60x
rDNA FISH on Pisum Metaphase Chromosomes
rDNA probes were used in the initial FISFI experiment to test the reproducibility
of the in situ hybridization reaction on a number of chromosome spreads. Results
received from these hybridization experiments using NOR probes labeled with biotin,
revealed either four or eight strong signals. The number of signals was dependent on the
state of chromatin condensation. More specifically, when the resolution of the
chromosomes allows for individual sister chromatids to be visualized, two signals from
each chromosome can be seen. However, when the targeted chromatin is in a state of
mitosis other than condensed metaphase, these two signals cannot be distinguished from
one another. Thus only one signal can be seen from each pair of chromosomes. In
Figure 9 (A & B), one distinct signal can be seen that originates from each of the sister
chromatids of a single chromosome or dyad. In contrast, Figure 9 (C & D) shows
77
chromatin in opposing states of chromatin dynamics with image C representing a highly
condensed form and image D representing a more decondensed form. The latter
effectively reduces the ability to resolve subtle differences in morphology, such as
identifying sister chromatids. Hence only four signals can be resolved. Nevertheless, in
each the pictures the probe signals remain localized to the II0 constrictions of
chromosomes 4 and 7.
Figure 9. Images A, B, & C- FISH using biotin (green) labeled NOR probe on early
metaphase pea chromosomes. Image D- FISH using biotin (green) labeled NOR probe
on prometaphase pea chromosomes. All chromosomes counterstained with DAPI
(A,C,D=red, B=blue). 60x
78
In addition to the NOR (45s subunit), the 5s ribosomal array was also tested for
'
reproducible localization. Figure 10 is a picture of a somatic chromatin isolated at
interphase of mitosis that was co-hybridized with an NOR (red) and a 5s (green)
ribosomal probe. The NOR was labeled with digoxigenin and the 5S with biotin. When
chromosome spreads hybridized with a probe containing the 5s array, six signals are
observed. Four of the six 5s rDNA signals are distinctly stronger than the other two.
Again, two loci containing the NOR subunit can be seen as four distinct signals.
Additional evidence of this signal localization can be seen in Figure 14C in which the 5s
.
.
|
probe is used in conjunction with a labeled BAG probe where the 5s probe represents a
positive check for the FISH reaction. The conventional chromosome morphology is
represented in this picture and again for the 5s rDNA there are four strong signals in
addition to the two secondary signals. All four of the strong signals are found on
5■
submetacentric chromosomes with two signals localized toward the telomere region on
!;
the long arm. The other two signals remain closer to the centromere on what appears to
'
be the largest of the submetacentric chromosomes, presumably chromosome 5. The
■
secondary signals are located toward the telomeric region on a set of chromosomes that
:
appear nearly metacentric.
:i
A final test for the reproducibility of the FISH experiment was to repeat the
;
experiment on chromosome spreads of Lens culinaris (2n=14). When probed with the
:
'
.
'
•
.
;
NOR and 5s probes, two distinct sets of signals are seen. The NOR and 5s loci reside on
separate chromosomes and appear equally intense. The NOR probe signal originates
from the terminal 11° constriction point of the homologous satellite chromosomes.
: I:
‘
;j
79
Figure 10. FISH on interphase Pisum nuclei, co-hybridized with both 5s (biotingreen) and NOR (digoxigenin-red) probes and counterstained with DAPI (blue). 60x
The 5s array probe is localized in a close proximity to the centromere on two metacentric
chromosomes. Unfortunately, overlapping chromosomes and relatively poor resolution
of the chromosome spread encumbers any further karyotypic characterization of the 5s
signal. In addition, probe signals were also detected from interphase chromatin, located
immediately next to the chromosome spread in this image (see Figure 11A). Again there
appear to be two strong signals from each of the NOR and 5s that have hybridized to four
loci within this diploid somatic nucleus.
80
Figure 11. Images A & B. FISH on Lens culinaris chromosomes co-hybridized with 5s
(biotin-green) and NOR (digoxigenin-blue) using multicolor-FISH. Chromosomes were
counterstained using DAPI (red). Image A 60x. Image B IOOx.
In figure 11A the signal from the 5s appears to be stronger than that of the NOR probe;
however, this is the only observed case of this signal difference.
Repetitive Clone FISH
Probe PisTR-B was previously shown to distinguish three of the seven
chromosome pairs through its hybridization pattern (Neuman et al., 2001). When used in
conjunction with the 5s rDNA probe, each of the seven chromosome pairs could be
identified cytologically. Probe PisTR-B was tested for use as a chromosome-identifying
tool with intentions of utilizing the probe to distinguish between morphologically
indistinguishable chromosomes for further hybridization experiments such as BACFISH Figure 12 (A and B) displays the general hybridization pattern received from the
hybridization experiments. Signal can be seen on nine of the fourteen chromosomes, of
81
which the majority appears to be localized toward the telometric regions of the
chromosomes. A better representation of the hybridization patterns can be seen in Figure
12b, although in this particular case only ten of the fourteen chromosomes have remained
within the field of view. A complete set of chromosomes displaying a signal from the
PisTR-B probe was not found.
Figure 12. Images A and B. FISH on Pisum chromosome, hybridized with biotin
labeled probe PisTR-B, and counterstained with DAPI. 60x.
The PisTR-B probe was tested in a number of experiments in which it was used in
conjunction with a BAC probe in multicolor FISH and/or blocking DNA (Cot -I). In
each of these experiments signal could not be detected when either condensed
chromosomes or interphase nuclei were examined (data not shown).
82
BAC-FISH Gene Family
{
BAC clone 103-K1 was used in an in situ hybridization experiments on
metaphase and prometaphase Pisum chromosome spreads. As indicated in the Results
section of Chapter 2, this particular clone contains sequences corresponding to the
Rubisco small subunit and is expected to be localized to two positions within the Pisum
karyotype. Each experiment was repeated with increasingly greater amounts of Cyt-I
blocking DNA, ranging from Ong to 4000ng (4pg). The best representation of a typical
hybridization pattern received from this particular BAC when used in conjunction with
low amounts (<1000ng) of blocking DNA can be seen in figure 13A. In this image,
prometaphase chromosomes display signal from biotin labeled BAC probe 103-Ki using
SOOng blocking Cyt-I DNA. The signal appears evenly dispersed throughout the semi-'
condensed chromosomes. A subtle accumulation of signal probe located telometrically
on an extended chromosome can be seen in the upper comer on the chromosome spread
(See arrow). However, this result was not reproducible under similar FISH conditions,
i.e. premetaphase chromosome arrangement under low blocking conditions.
Figure 13B shows a hybridization pattern received when the probe was used in
conjunction with relatively large amounts blocking Cyt-I blocking DNA, in this case
'
4000ng was used. In this experiment, the dispersal of signal received from the labeled
BAC probe has been noticeably reduced. It should be noted that within this particular
chromosome spread, only ten of the fourteen chromosomes are within the field of few.
Probe signal appears localized to six points on. six of the fourteen chromosomes. Four
signals appear to reside on homologous chromosomes.
'
■
■
83
Figure 13. Image A- FISH on premetaphase Pisum chromosomes hybridized with biotin
labeled BAC 103-K1 (green) and counterstained with DAPI (blue). 5OOng blocking DNA.
60x. Image B- FISH on Metaphase Pisum chromosomes hybridized with biotin (green)
labeled BAC 103-K1 and counterstained with DAPI (red). 4000ng blocking DNA 60x.
Two of the signals are localized to the short arms of acrocentric chromosomes (Arrows
A) and the other two signals appear adjacent to the centromere on chromosomes that are
nearly metacentric (Arrows B). Of the remaining two signals, one is localized to what
appears to be satellite that may have been separated from its respective chromosome.
The homologous signal is located telomerically on a separate submetacentric
chromosome (Arrows C). This result could be interpreted as a signal received from one
of the two pairs of homologous satellited chromosomes, both of which may have been
separated from their respective satellite during the squashing procedure. The putative
body of one of these chromosomes has been labeled distally and adjacent to the point
were the secondary constriction would normally be viewed. The signal from the other
chromosome was labeled on the satellite itself, presumably within the region that would
otherwise contain the secondary restriction connecting it to the satellite’s respective
84
chromosome. This result would suggest the existence of a third locus. This experiment
was not repeated and the possibility of artifactual signal could not be ruled out.
BAC FISH Single Copy
■Clone 89-E10 Was previously described in chapter 2 as containing sequence that
corresponds to one of the two Gsc (cytosolic glutamine synthetase) gene “twins”, both of
which have been characterized a single copy genes (Walker et ah, 1995). Sequence
results indicated a closer sequence identity to that of GS3A rather than GS3B. This
particular marker has been genetically anchored to linkage group VII (Brauner et ah,
.
2002). Probe 89-E10 was used in FISH experiments with increasing amounts of C gt-I
blocking DNA. Initially no blocking DNA was added to the reaction mix with
subsequent reactions incorporating an increasing amount of Cgt-I in increments of
'
500|ig. Figure 14 (A and B) has representative images of either interphase nuclei or
condensed metaphase chromosomes that have been hybridized with biotin labeled BAC
89E-10 using 500ng of blocking DNA. These images are indicative of the results
typically seen when clone 89E-10 was hybridized using lower amounts of C g t-I blocking
DNA. The results received are also similar to the type of hybridization pattern seen when
BACs containing Imown gene family members were used. The signals are evenly
dispersed throughout the chromosomes. Figure 14C displays an image of an additional
reaction in which the 5s rDNA probe that was described previously was labeled with
digoxigenin and used as a positive check on the hybridization reaction.. All other reaction
85
conditions, including labeling and amount of blocking Cyt-I DNA (500ng) were identical
to the reaction conditions used to produce Figure 14 (A and B).
Figure 14. Images A & B) FISH on prometaphase and interphase Pisum chromosomes
(respectively) hybridized with BAC 89-E10 (biotin-green) and counterstained with DAPI
(A=red, B=blue) using FISH. 500ng Cyt-I DNA 60x.
Image C) Metaphase Pisum chromosomes co-hybridized with Ss (digoxigenin-red) and
BAC 89-E10 (biotin-green) and counterstained with DAPI (blue). SOOng Cyt-I DNA 60x.
86
In contrast to the gene family FISH experiments, the results shown here suggest
that the increase of blocking DNA used in each reaction did not significantly sequester
non-homologous, probe binding sites. Unfortunately, probe signal was only detected
from one of the slides used in this particular experiment. Figure 15 contains a
chromosome spread displaying a hybridized biotin labeled with probe 89-E10 and
blocked with 3OOOng of Cot-1 blocking DNA. The hybridization signal appears in an
abundance that is similar to that observed in the BAC-gene family hybridization when
low amounts of blocking DNA were used. Although individual chromosomes cannot be
distinguished in this particular image, the results clearly indicate that that nonhomologous probe signal is not suppressed and no evidence of a probe localization
indicating a single copy gene is readily observed.
f ..
ST
Figure 15. FISH on Pisum metaphase chromosomes hybridized with biotin labeled
BAC 89-E10 (green) and counterstained with DAPI (red). 3OOOng blocking DNA
60x.
87
.
Discussion
The intent of this investigation was to assess the feasibility of detecting sequences
of relatively low copy within a large insert clones in this case a Bacterial Artificial
Chromosome (BAG), in situ on chromosomes spreads o f f . sativum. The utility and
reproducibility of this approach has been demonstrated many times as indicated in the
literature review. What differentiates this approach from the previously established ■
experiments is the size and organizational complexity of this particular genome. When
compared to other systems in which BAC FISH is done routinely, the size of the pea
genome dwarfs the others. For instance Arabidopsis (145 Mbp), Brassica sp. (400-700
Mbp), Sorghum (748 Mbp), Medicago (500 Mbp), Oryza (450 Mbp) all have genomes
under 1000 Mbp, whereas the genome size of the garden pea has been estimated to range
between 3947-4397Mbp. (Arumugahathan et ah, 1991). The genetic content amongst
different plant species, that is the number of the transcribed genes, remains relatively the
same. However, it is the repetitive “filler” DNA that is responsible for the vast size
differentiation among plants. This phenomenon is commonly referred to as the “C-value
paradox” (Fairbanks et ah, 1999), which simply states that there is substantial variability
among plant genome sizes but little variability in the number of transcribed genes .
Herein lies the experimental question: considering the vast amount of repetitive DNA
responsible for pea’s large genome and the fact that this repetitive DNA is dispersed
relatively evenly throughout (Murray et ah, 1981): will FISH be able to unambiguously
detect localized BAC probe signal given the abundance and distribution of repeats within
the pea genome?
''
88
In order to develop a reproducible experimental design for in situ sequence .
detection on P. sativum chromosome spreads, rDNA probes were employed because the
utility and characteristics of the hybridization pattern has been well established (Neuman
et ah, 2001; Fuchs et al., 1998; and Jorgensen et ah, 1987).. As indicated in these studies,
the large rDNA clusters (45s or NOR) reside on the two satellited chromosomes,
specifically within the secondary constrictions of IV and VII. Our placement of the
hybridization signal on the secondary restriction of the chromosomes IV and VII are in ■
agreement with the previous characterization of the 45 s array, on pea chromosomes.
The 5s hybridization signal was initially documented on interphase nuclei (Figure
9). The number and signal intensity of 5s loci observed are in agreement with previous
reports (Neuman et al., 2002). Specifically, the 5s arrays are localized to three loci, with
two of the signals displaying greater intensity than the third. Figure 9 reflects this
description. The 5s rDNA cluster has been shown to be localized to chromosomes 5, 3,
and I or 2 with recent results favoring chromosome I (Neuman et al., 2002). The 5s
probe was later used in conjunction with a BAC probe as a positive check in a FISH
experiments. In reference to Figure 13, six 5s signals originating from three loci can be
seen. One pair of signals appears localized interstitially on the short arm of the largest of
the acrocentric chromosomes, which is in agreement with the previous chromosomal
placement of chromosome 3. The second pair is located toward the telomere on the short'
arm of a submetacentric, presumably chromosome 5. The third signal appears to be
located on the long arm of a nearly metacentric chromosome, presumably chromosome I .
Both of these results corroborate the previous placement of the other two 5s signal on
89
chromosomes. 3 and I, respectively (Fuchs et ah, 1990 and Neuman et ah, 2002), A small
side experiment involving FISH using the same 5s probe on chromosomes ofZ. culinaris
provided an interesting result. To the best of our knowledge, this is the first
documentation of the placement of the 5s rDNA within the lentil karyotype. In contrast
to the three loci seen within pea, the lentil displays signal from only one locus.
According to the results presented in Figure 10, it appears that the 5s rDNA locus is .
Iocalizedjust outside the centromeric region of a large, nearly metacentric chromosome
and the NOR is located terminally on a.different chromosome. When compared to the
signal received-from the 5s rDNA in Pisum, the FISH signal closely most resembles the
signal from pea chromosome 5. In further support of this hypothesis, Chapter 5 describes
the relationship, between the pea and lentil linkage maps in which Conserved gene synteny
is observed between pea and lentil with respect to pea linkage group III (Chromosome 5).
The locus number and position of the 5s rDNA locus varies among and within species
(Cerbah et ah, 2003 and Raina et ah, 2001). Thus it would require further
experimentation using species positioned basal to lentil and pea within the tribe Vicieae
to determine if the difference in locus number was due to a selective loss of locus within
lentil, or due to gene duplications within pea. The 45 s data suggests that this locus has
been duplicated in pea, which is supported by the fact that Medicago truncatula, Vicia sp,
Lens sp. and Cicersp all contain one 45 s array. With the exception of Lens sp.., the other
three species are positioned more basal within the Vicioid clade (Steele et ah, 2003),
suggesting that the duplication was independent of species divergence.
90
Results obtained from FISH experiments using the PisTR-B clone donated to our
program from Dr. Pavel Neuman indicate that the probe is of limited value when used in
conjunction with BAC probes. Signal from this particular probe could not be detected
when co-hybridized with a BAC. The results suggest three possible hypotheses that
would explain the lack of signal. First, the vector of the probe may be cross-hybridizing
with the BAC vector, in effect sequestering it from accessing the targeted chromosomes.
This hypothesis is supported by results received from screening the BAC library. Probes
of the plasmid clones that were used to screen the library sequences strongly Crosshybridized to all the BACs on the filters at high stringency thus indicating a close
sequence homology amongst the two vectors (data not shown). The second hypothesis is
that the insert sequences themselves are cross hybridizing. Pea BACs undoubtedly
contain large quantities of repetitive sequence and clone PisTR-B is repeat sequence, as it
was isolated from Q t fractions (Neuman et al.,-2001). Finally, the use of blocking DNA
may be affecting the availability of the probe to find target sites on the chromosomes. In
this case Cyt-I DNA isolated from genomic pea DNA may find homologous sites on the
probe in excess of those available on the target DNA and effectively sequester the
binding availability of the PisTR-B. A clear demonstration of any of these candidates
may not be possible as it quite possible that all three factors may contribute to the lack of
signal obtained when using this particular probe in conjunction with BAC probes.
PisTR-B probe was used in a few ‘single’ probe FISH experiments.resulting in FISH
patterns that closely resemble those described by Dr. Neuman, although a complete
corroboration of results was not obtained. The results were presumably due to poof
91
quality of the slides used with this probe. Howeveqj the few slides that were examined
using the PisTR-B clone alone .did suggest the utility of this clone for identifying specific
chromosomes. I expect that more experiments would have revealed a more complete
chromosome set thus ,allowing validation of Dr. Neuman’s published results.
The rDNA FISH established the repeatability of the FISH experiment on pea
chromosomes. BAC clones were then tested for the ability to remain localized to a
specific sequence in situ. Rather than moving from repetitive sequence FISH (rDNA)
directly to unique.sequence BAC FISH, the structure of a gene family was exploited for
its tandemly oriented gene and pseudogene repeat structure (Sugita et ah, 1987). Thus,
one would expect a strong signal localized to only a few loci within the genome. In this
case a BAC containing the Rubisco subunit sequence was used for FISH. Initially, and
not unexpectedly, the use of low amounts of blocking Q t DNA produced a signal that
was dispersed throughout the chromosome spreads. This result indicates that there must
be enough repetitive sequences dispersed throughout the BAC insert sequence to crosshybridize to many sites within the pea genome. When blocking DNA was increased the
signal was restricted to three loci. It should be noted that the dispersed signal seen in
Figure 12 from an interphase chromosome cannot be directly compared to the signal
intensity and dispersion that would be seen if the identical experiment was reproduced on
condensed metaphase chromosomes. It has been suggested that interphase chromosomes
are much more sensitive-to DNA/probe heteroduplex formation, particularly in regard to
complex genomes (Jiang et ah, 1996). The Rubisco small subunit has been genetically
mapped in pea to two distinct loci, one on linkage group V and one on VI (chromosomes
92
3 and I respectively) (Polans et al., 1985; Hall et al., 1997). Two of the loci that were ,
detected using FISH appear to be in agreement with the genetic mapping. This ' .
•Conclusion is supported by the fact that one signal is localized on a submetacentric
chromosome presumably chromosome I and the other on a acrocentric chromosome
which may be chromosome 3. An easy way of verifying this result would be to use this
BAG containing the Rubisco small subunit in conjunction with the 5s rDNA in a FISH
experiment. Confirmation would be a co-localization of the 5s array and the BACon
both chromosomes I and 3. The detection of a third locus using BAG FISH cannot easily
be explained. According to RFLP data the Rubisco probe did produce a number of bands
indicating the possibility of multiple loci; however, none of the results, suggested the
presence of a third locus (Polans et ah, 1985; Hall et al., 1997). As indicated in the
Results section, the present work is the only documentation of localized hybridization
patterns from the FISH experiments using the tRubisco BAG’, thus artifactual signal
cannot be ruled out.
.
. 1
BAG clone 89-El Owas used to test the ability of FISH to detect localized signal
from a single copy gene of on pea chromosome spreads. Unfortunately, this experiment
was encumbered by problems outside those that could be attributed to the experiment
itself. Specifically at the time of the experiment the fluorescently labeled antibody
conjugate failed to produce signal, even on positive checks. Hence, I was not able to
obtain any data from numerous slides that were used in the FISH experiment. Many
initial undocumented experiments using this BAG provided evidence that the insert
sequence produced a dispersed hybridization pattern similar to that seen on the initial
93
‘gene family’ BAC FISH experiments. In contrast to these former experiments, the
blocking'of orthologons binding sites was not as effective as that seen with the gene
family BAC FISH. Suppression of background signal that would be expected when
increased amounts of blocking DNA was used was not found, at least not sufficient to '
permit signal localization. These results, however, are not completely unexpected and
reflect the reasoning for initially using a BAC containing a gene family. The BAC inserts
from the pea BAC library have an average insert size of 11Okb, thus 89-El0 containing a
single copy of a Ikb sequence homologous to Gsc (see Materials and Methods), would
potentially have 109kb of unknown insert sequence which may very well contain
repetitive sequences. The BAC containing the Rubisco small subunit conversely should
have several kilobases taken up by multiple versions of Rubisco sequence, in both coding
and non-coding (pseudogene) forms, thus limiting the amount of insert space available to
alternate repeat sequences. These results both indicate that a more thorough
characterization of the BAC insert used for FISH experiments is needed to determine the
feasibility of such ah approach.
To address the question of future experimentation using this approach in Pisum,
two experimental possibilities are suggested. First, screen for BACs in which the insert
sequence originates from areas of the pea genome that have been characterized as gene
rich areas. If a subset of BACs were identified that had potential for FISH experiments
one could screen these clones via dot blot hybridization experiments using C0t fractions
with the intent of identifying BACs that contain minimal amounts of repetitive DNA
(Zwick et ah, 1997).' Second, an alternative to characterizing the BACs themselves, one
94
could improve upon the genomic coverage of the blocking DNA. Different fractions of
Cot DNA could be tested for the ability to block repetitive sites. It has been suggested
that blocking only the highest of the repetitive sequences, that is those isolated at a Q t-1,
would not provide sufficient coverage of the repetitive sequences and the FISH would
still be contaminated by moderately repetitive sequence (Hans de Jong, personal
communication). To test this hypothesis, different fractions of repetitive DNA could be
used including those isolated at a Q t-100. Certainly, one could envision other
approaches to improve BAC FISH, however, the data obtained from my experiments
suggest the described experiments would be of value.
Although the results of these initial experiments are lacking in reproducibility and
quantity, they do indicate that the use of BAC-FISH on the pea genome will be anything
but trivial. Certainly, a more robust experimental design will be required to produce
conclusive results in order to localize single copy genes from large insert clones on
chromosome spreads ofP. sativum.-
'
.
95
Literature Cited
Abbo S. T.E. Miller, S.M. Reader, R.P. Dunford, T. Shimada.'(1995). Detection of
ribosomal DNA sites in lentil and chickpea by fluorescent in situ hybridization. Genome
37:713-716.
Ammuganathan, K. E.D. Earle. (1991). Nuclear DNA content of some important plant
species. Plant Molecular Biology Reporter. 9:211-215.
Blixt S. (1955). Cytological investigation of line no. 21 of Pisiim. Agri Hort Genet.
13:207-213.
Blixt, S. (1958a). Cytology of Pisum I. A Methodical Investigation. Agri Hort. Genet.
16:66-77.
'
■
Blixt, S. (1958b). Cytology of Pisum II. The normal karyotype. Agri Hort. Genet.
16:221-237.
Blixt S. (1959). Cytology of Pisum III. Investigation of five interchange lines and
coordination of linakge groups with chromosomes. Agri Hort. Genet. 17:47-75.
Brauner S., R.L. Murphy, J.G. Walling, J. Przyborowski, N.F. Weeden. (2002). STS
markers for comparative mapping in legumes. J. Amer. Soc. Hort. Sci. 127 :616-622.
Carol! G. (1952), The effect of some oxyquinoline derivates on pea chromosomes. Agri
Hort. Genet. 10:152-157.
Carol!, G., Blixt S. (1953). The morphology of the chromosomes of-Pisum. Agri Hort.
Genet. 11:133-140.
Caroli, G., Blixt S. (1954). Cytological verification of the genanalytically proved
interchange between chromosomes III and V of Pisum. Agri Hort. Genet. ' 12:107-114.
Caroli, G., Blixt S'. (1955). A cytologieal investigation for verifying the genanalytical
results regarding line no. 680 o f Pisum. Agri Hort. Genet. 13:95-102.
Cerbah, M.., Z. Kelvei, S. Siljak-Yakovlev, E. Kondbrosi, A. Rone, T. Hanh-Trinh.
(1999). FISH chromosome mapping allowing karyotypic analysis in Medicago
truncantula lines Jemelong and R-108-1. MPML 12:947-950.
Cheng, Z., G.G-Presting, C.R. Buell, R.A.Wing, J. Jiang. (2001). High resolution
pachytene chromosome mapping of bacterial artificial chromosomes anchored by genetic
96
markets reveals the centromere location and the distribution of genetic recombination
along chromosome 10 of rice. Genetics 157:1749-1757.
Coyne, C. K. Meksem, D.A. Lightfoot,' K.E. Keller, R.R. Martin, M.T. McClendon,
D.A. Inglis, E.W. Storlie, K.E. McPhee. (2000) Construction of bacterial artuficial
chromosome library for pea (Pisum sativum L.) Pisum Genetics 32: 23-26.
Enrico, A., C. Conicella, G. Venora. (1991). Karyotype studies on Pisum fulvum and
Psium sativum, using a chromsome image analysis system. Genome. 34:105-108. .
Fuchs, I , M. Kuhne, I, Shubert. (1998). Assignment of linkage groups to pea
chromosomes after karyotyping and gene mapping by fluorescent in situ hybridization.
Chromosoma. 107:272-276.
FuchsJ., I. Shubert. (1995). Localization of seed protein genes on metaphase
chromosomes of Vicia faba via fluorescence in situ hybridization. Chrom. Res. 3:94100.
Gall J.G., M.L. Pardue, (1969). Formation and detection of RNA-DNA hybrid molecules
in cytological preparations. Prdc Natl Acad Sci U S A , 63, 378-383.
Hall KJ. J.S. Parker, T.H.N. Ellis. (1997a). The relationship between genetic and
cytogenetic maps of pea. I. Standard and translocation karyotypes. Genome 40:744-754.
Hall K.J. J.S. Parker, T.H.N. Ellis, L, Turner, M.R. Turner, J.M.I. Kofer, J.Lu, C.
Ferrandiz, P.J. Hunter, J.D. Taylor, K. Baird. (1997b). The relationship between
genetic and cytogenetic maps of pea. II. Physical maps of linkage mapping populations.
Genome 40:755-769.
v
...
Heslop-Harrison, J.S,, T. Shwarzacher,. J. Malusynska, K. Anamthawat-Jonsson, M. Shi,
A.R. Leitch, I.J. Leitch. (1991). In situ hybridization with automated chromosome
denaturation. Technique 3: 109-115.
.
■
Jackson S.A., M.L. Wang, H.M. Goodman, J. Jiang. (1998). Application of fiber-FISH
In physical mapping o f Arabidopsis thaliana. Genome 41:566-572.
Jiang, J. S.H; Hulbert, B.S. Gill, D.C. Ward. (1996). Interphase fluorescence in situ
hybridization mapping: A physical mapping Strategy for plant species with large
complex genomes. Molecular and General Genetics. 525:497-502.
Jorgensen, RA., R.E. Cuellar, W.F. Thompson, TA. Kavanagh. (1987). Structure and
variation in ribosomal RNA genes of pea. Plant Molec. Biol. 8:3-12.
97
Krishnan, P., V.T. Sapra, K.M. Soliman, A. Zipf. (2001). FISH mapping of the 5s and
18s-28s rDNA loci in different species of Glycine. J. Heredity 92:265-300.
Kulikova O., G. Gualteri, R. Geurts, GJ. Kim, D. Cook, T. Huguet, H. de Jong, P.F.
Fransz, T. Hisseling. (2001). Integration of FISH pachytene and genetics maps of
medicago truncatula. Plant J. 27;49-58.
Lamm, R (1981). Giemsa C banding and silver-staining for Cytological studies in Piswn..
Hereditas 94:45-52.
LammR., R.J. Miravelle. (1958). A translocator tester set in Pisum. FIereditas.
46:737-744.
Lewitsky, G.A. (1931). Investigation on the morphology of chromosomes. Bulk Appl.
Bot. Genet. Plant Breed. Leningrad 27.
MozoT., K. Dewer, P. Dunn, J.R. Ecker, S. Fischer, S. Kolska, H. Lehrach, M. Mama, R.
Martienssen, S. Meier-Ewert, T. Altmann, (1999). A complete BAC-based physical map
of
Arabidopsis thaliana gtnome. Nat. Genet. 22:271-275.
Murray, M., R.E. Cuellar, W.F. Thompson. (1978). DNA sequence organization in the
pea genome. Biochemistry. 17:5781-5790
Murray, M., D. Peters, W.F. Thompson; (1981). Ancientrepested sequences in the pea
and mung bean genomes and implications for genome evolution. J.Mol. Evok 17:31-42
Neumann, P., M. Nouzova, J. Macas. (2001). Molecular and cytogenetic analysis of
repetitive DNA in pea (Pisum sativum). Genome 44:716-728.
Neuman P., D. Pozarkova, J. Vrana, J. Dolezel, J. Macas. (2002). Chromosomes
sorting and PCR-based physical mapping in pea. {Pisum sativum L.). Chromosome Res.
10:63-71.
Raina, S.N., Y. Mukai, K Kawaguchi, S. Goel, A. Jain. (2001). Physical mapping of the
18S-5.8S-26S and 5S ribosomal RNA gene families in three.important vetches {Vicia
species) and their allied taxa constituting three species complexes. Theor. Appk Genet.
103:839-845.
Simpson, P.R., M.A. Newman, D.R. Davies. (1988). Detection of legumin gene DNA
sequences in pea by in situ hybridization. Chromosoma 96:454-458.
Simpson, P.R., M.A. Newman, D.R. Davies, T.H.N. Ellis, P.M. Matthews. (1990).
Identification of translocation in pea by in situ hybridization with chromosome specific
DNAprobes. Genome. 33:745-749.
98
Shubert I., U. Wobus. (1985). In situ hybridization confirms jumping nucleolus
organizing regions in Allium. Chromosoma. 92:143-148.
Southern,. E.M. (1975). Detection of specific sequences among DNA fragments of
separated gel electrophoresis. J. Mol. Biol. 98:503.
Steele, K. P., and M. F. Wojcieehowski. (2003). Phylogenetic systematics of tribes
Trifolieae and Vieieae (Fabaceae). Advances in Legume Systematics. (in press).
Sugita5M., T. Manzara, E. Pichersku, A.R. Cashmore, W. Gruissem. (1991). Genome
organization, sequence analysis, and expression of all five genes encoding the small
subunit of ribulose-1-5-bisphosphate carboxylase from tomato. Mol. Gen. Genet.
209:247-256.
Tadmor Y., D. Zamir, G. Ladizinsky. (1987) Genetic mapping of an ancient translocation
in the genus Lens Theor. Appk Genet. 73:883-892.
Walker E. Li, N.F. Weeden, C.B. Taylor, P. Green, G. M. Corruzzi. (1995). Molecular
evolution of duplicate copies of genes encoding cytosolic glutamine synthetase in Pisum
sativum. Plant Mol. Bio. 29:1111-1125..
Zwick M.S., TL E. Hanson, T.D. McKnight, M.H. Islam-Faridi, D.M. Stelly, RA.
Wing, H J Price. (1997). A rapid procedure for isolating Cot-1 DNA from plants.
Genome. 40:138-142.
99
CHAPTER 4
COMPARATIVE GENETIC MAPPING BETWEEN LENTIL (LENS SPP.) AND PEA
Literature Review
Introduction
Throughout the past century, a primary focus among' geneticists has been
identifying genetic linkages within species. Initial efforts were placed oh mapping the
genetic traits of a few individual species, some of which are now referred to as model
species. As the linkage .maps of these systems grew, researchers undertook an effort to
develop efficient methods for generating linkage maps among genetically
underrepresented systems. Comparative genetic mapping or the evaluation of eolinear
gene arrangements among closely related species proved such a method for efficiently
developing knowledge for expanding linkage maps in species that had not been
intensively studied. The utility of exploiting linkage maps that have been developed
among popular genetic systems for mapping in other species has been suggested within
the disciplines of both comparative and functional genomics (Hart et ah, 1977; Tanksley
et ah, 1988; Mysore et ah, 2001).
Comparative Genetic Mapping
.
Arabidopsis thaliana has been, completely sequenced and has a saturated linkage
map, representing what is probably the most widely used system for comparative studies
(Hauge et ah, 1993; Hall et ah, 2002).
Kowalski et ah (1994) investigated the
100
hypothesis of conserved sequence arrangement between Bmssica oleracea and
Arabidopsis thaliana Using comparative analysis of linkage marker order. Although both
of these species vary karyotypically, this approach revealed eleven regions of conserved
synteny that span approximately 160cM (centimorgans) (24.6%) and 245cM (29.9%) of
the A. thaliana and B. oleracea genomes, respectively.
Evidence of conserved gene synteny has also been supported through
investigations not involving Arabidopsis. Early studies comparing wheat (Trilicum
aestivum) to its wild relatives were one of the first cases in which established linkages
were exploited to intensify other maps (Hart, 1979). Marker conservation.in the
Triticeae was later extended to encompass members of Poaceae in which conserved gene
synteny among these taxa.was established (Ahn et ah, 1993; Sherman et ah, 1995).
Efforts have been made to investigate such phenomena within the family
Solanaceae. The closely related species of potato and tomato were investigated for
conserved gene synteny. Evidence indicated that potato and tomato display nearly
perfect preservation of syntenic linkage between the two divergant taxa (Bonierbale et ah,
1988; Tanksley et ah, 1992). Tanksley et ah (1988) used a common set of cDNA RELP
markers to construct linkage maps for both tomato and pepper {Capsicum spp.). The
results obtained from mapping an identical pool of cDNA clones on both, species
suggested that the presence of these genetic markers was preserved through species
divergence. However the arrangements of genes in both species appeared significantly
different and in some cases complete linkage groups appeared restructured. Further
t
Studies on comparative mapping were reported by Prince et ah (1992) who constructed a
101
molecular linlcage map of pepper {Capsicum spp.) based on restriction fragment length
polymorphisms (RFLPs) and isozymes. The synteny of genetic markers ordered on the
pepper linkage map was compared to that of.established tomato map. The results
indicated that a minimum of 31% of the current pepper linkage map was thought to
display conserved gene synteny with respect to tomato.
Within the family Fabaceae (Leguminosae) examples of conserved gene synteny
have been reported. Boutin et al. (1995) used a set of 219 DNA clones to establish the
colinearity of genetic markers shared among three genera including Vigna radiaia and V.
unguiculata, Phaseolus vulgaris, and Glycine max. The results indicated.that the
members of the subtribe Phaseolinae (V. radiaia, V. unguiculata and P. vulgaris)
displayed considerable linkage co-linearity while G. max appeared more divergent.
Intertribal comparisons between Cicereae (chickpea (Cicer arieritinum)) and Vicieae (pea
I
{Pisum sativum) arid lentil {Lens culinaris)) as well as intratribal comparisons between P.
sativum and Lens spp.; have both reported evidence indicating considerable conservation
of gene synteny among these species (Simon et ah, 1997; .Weeden et al., 1992.). Torres
et al. (1991) produced, a linkage map of Vicia faba that consisted of 66 segregating
markers (isozyme, RFLP, and RAPD) which were then assembled into 11 independently
assorting groups. Although this report did not indicate areas of possible shared synteny
with related species, the findings will be informative to .future examinations of colinearity
among temperate legumes. Clearly, the ability to utilize known marker linkages for
species that might share .some genetic similarities establishes the power of using
comparative mapping to both generate and saturate linkage maps within related species.
102
■Sequence Tagged Site (STS) Markers
Geneticists are continually looking for genetic markers that allow a quick and
efficient means of constructing and intensifying linkage maps. Many genetic markers
exist including but not limited to: Restriction Fragment Length Polymorphism (RFLP),
Amplified Fragment Length Polymorphism (AFLP), Random Amplified Polymorphic
DNA (RAPD), Simple Sequence Repeat (SSR), isozyme, and morphological markers,
However, the utility of these markers is governed both by the efficiency at which
individual loci can be mapped and how informative such markers would be in future
applications. STS markers.are constructed by designing PCR primers from a Icnown
sequence that designates a particular locus within the genome. The characteristics of STS
markers suggest the value of such markers to a mapping program. STS markers can be
generated via PCR; thus the requirements of radioactive labeling agents, prolonged
experiment length, and high technical proficiency that are typically experienced with
markers such as RFLP and AFLP, are alleviated (Taylor et ah, 2001). Moreover the
amount of template DNA required for a PCR reaction is significantly less than that
required for RFLP analysis. STS markers are predominantly expressed in a co-dominaht
fashion, allowing the assessment of heterozygosity within a population. Currently, a
major effort in genomics is the sequencing of genomes thereby, generating markers that
reflect sequence data in contrast to markers such as isozymes and morphological
variation; are of particular interest. As the name implies, STS markers can be directly
associated with a coding sequence that allows complete correlation of marker sequence
103
data to genomic database information such as that found in BLAST, EST and BAC
databases. Primer sets for STS markers can be designed to target exons within
transcribed genes, allowing for cross-species applicability. The sequence that is
amplified using STS' primers often spans one or more introns, thereby greatly improving
the probability of detecting sequence polymorphism at a particular locus among
segregating lines. As introns are known .to accumulate substantially greater amounts
polymorphisms over exons (Holland et ah, 2001; Small et.ab, 2000) the inclusions of
introns is important for the detection of variability among orthologous loci.
The
usefulness of using STS marker in comparative studies is evident, and their utility has
been previously demonstrated (Taylor et ah, 1992; Iwata et ah, 2001; Tsunlura et ah, ■
1999).
Genetic Mapping In Lentil
Lentil {Lens culinaris. 2n=14) has not been subjected to the type of intense
linkage studies that have been applied to relatives such as soybean and pea.
Consequently, the number of linkage groups currently established in lentil is greater than
the haploid chromosome number. In Lens, hybrid studies between L. culinaris and L. ;
orientalis (Ladizinsky, 1979a & 1979b) allowed the first characterization of the cytology
of lentil and the genetics behind many of the morphological traits. The first reports of
gene linkages in lentil however, were not reported until several years later. Eight ■
isozyme markers were tested on the previously described cross to generate two linkage
groups composed of five loci (Zamir and Ladizinsky, 1984). In order to maximize the
104
genetic diversity within a potential mapping population, five species of lentil were
investigated for crossability and karyotypic arrangement (Ladizinsky et ah, 1984). The
genus could be divided into two general groups: I) L. culinaris, L. orientalis, and L. ■
odemensis, and 2) L. nigricans and L. ervoides. Ladizinsky et ah. (1985) used embryo
rescue to facilitate the recovery of viable progeny from the original cross between L.
culinaris and the wild species L. ervoides, both of which are situated in separate
crossability groups.
Mapping populations derived from the L. culinaris x L. ervoides cross were first
used in a mapping study by Tadmor et al. (1987). The investigation used four mapping
populations from six parents' to test 18 segregating markers. The marker sets used in this
study consisted of both isozyme and morphological markers and were assembled into five
•
■
.
i
linkage clusters that were tested using both F2 and F3 mapping populations. The study
also characterized, both genetically and cytologically, a reciprocal translocation involving
two chromosomes. Comparative analysis of the markers with their syntenic position in
Pisum suggested that the chromosomes involved would be similar to what we now refer
to as linkage groups I and II, in pea.
Muehlbauer et al. (1989) used a sets of progeny derived from seven crosses
between L.culinaris and L.orientalis to construct a lentil linkage map. The study also
tested the relationship among assembled linkages from previous lentil studies. Fourteen ;
isozyme and two morphological markers were assembled into six linkage groups
;
spanning 86 discontinuous centimorgans. This study also investigated the subcellular
compartmentalization of these isozymes in which confirmed several of the linkages that
105
were found. The lentil linkage map was further saturated with the inclusion of DNA
markers that were tested using the same L. culinaris x L. orientalis cross (Havey and
Muehlbauer, 1989). Nine partial linkage groups were assembled that consisted of 20
RFLP, eight isozyme and six morphological markers.
A mapping population produced from an interspecific cross between parental
lines L. culinaris #7 (Ladizinsky #7) and L. ervoides #32 (Ladizinsky #32) was used by
Weeden et al. (1992) to produce the most complete linkage map of lentil to date. In this
study, 64 markers consisting of isozyme, RFLP, and morphology were assembled into 11
partial linkage groups spanning 560cM.
Linkage Conservation Among Temperate Legumes
. Numerous studies have revealed conserved gene synteny among closely related
species. The intent of this investigation was to test the hypothesis of complete shared
synteny among temperate legumes (see Thesis Introduction) as demonstrated by the
following. First, the objective of the lentil investigation as previously described by
Weeden et al. (1992) was to compare the linkage conservation among the genes mapped
between two species within the tribe Vicieae, lentil and pea. The results of comparative
mapping between Pisum sp. and Lens sp. indicated a minimum of 40% linkage group
conservation which is in agreement with the proposed macrosyntenic gene correlation
among legumes (Weeden et al., 1992; Ellis et al. 2002). This same investigation,
however detected inconsistencies among linkage blocks indicating rearrangements not in
agreement with complete shared synteny. In particular, the placement of the 45s
106
ribosomal array in lentil deviated from both the arrays in pea and contradicted previous
placement in lentil..Intertribal linkage comparisons between P. sativum (Vicieae) and
Medicago truncatula (Trifolieae) revealed islands of gene synteny have been exposed
•>
between both, with some results indicating conservation of entire chromosomes (Vincent
.
et ah, 2000). Further comparative studies between these two.taxa have revealed a greater
amount of conserved gene synteny beyond that previously reported. Dr. N. Ellis reported.
that with the exception of pea linkage group VII; each of the six linlcage groups of pea
show a macrosyntenic relationship with the linlcage. groups reported in M. .truncatula.
(Ellis et al., 2002), Thus, one would expect an even greater extent of conserved gene
synteny shared between the two closely related taxa of P. sativum and L. cidinaris, than
between the slightly more divergent taxa of P. sativum and M. truncatula,
Morphological markers linked to molecular anchor markers have been shown to
share similar arrangements in both pea and lentil. Walling and Weeden (2002)
demonstrated the homology of two similar morphological characters D (Pisum) and Gs
(Lens), by establishing linkage between both Cop I,.a single copy gene in pea, and D in
pea and Gs in lentil. In addition the study conversely suggests the convenience of using
previously anchored morphological markers to supplement those markers currently being
tested.
Recently, a number of STS markers have been developed and anchored to the P.
sativum linkage map (Brauner et al., 2002). The mapping of this STS marker set within
I
pea RILs placed two STS markers on each of linkage groups I and II, and at least five
markers on each of the other five linkage groups. The wide coverage of these markers
107
across the pea map implies the value of such a marker set for comparative mapping
studies within legumes.
Lentil and pea are both placed, in the tribe Vicieae within the family FabaCeae
(Leguminosae). As indicated in the Introduction, the economic importance of these
species within the family underscores the significance of understanding the genetic
relationships between species. The following investigation will apply the previously'
described set of Pisum STS markers to further develop a lentil linkage map using the
Lc#7 x Le#32 Lens Recombinant Inbred Line (RIL) population, originating from the
previously described cross L. culinaris xL. erviodes (Ladizinsky et ah, 1985; Tadtnor et
ah, 1987). The linkages revealed on this map will be compared to those found on the
consensus P. sativum linkage map. Furthermore, the detected linkages will be integrated
with previously anchored isozyme, morphological, RFLP and protein markers in order to
maximize the density of informative markers within the linkage groups. RAPD markers ■
will be subsequently added to close gaps between loosely linked markers. It is
hypothesized that, by testing the relationship of genetic linkages within a lentil RIL
population using markers genetically anchored to Pisum, areas of conserved gene synteny
between lentil and pea will be revealed.
. 108
Materials and Methods
Mapping Population
50 Fg RILs derived from the original cross of L. culinaris #7 and L. ervoides #32
previously described by Tadmor et al. (1987), were used in this mapping project. Earlier
studies using this population, suggest that the parents of this wide cross, display
considerable genetic divergence, a characteristic desired in any mapping population. A
single reciprocal translocation involving Pisum linkage groups I and II and another
chromosome has been previously characterized and must be accounted for when mapping
within this region (Tadmor et ah, 1987; Weeden et al., 1992). Although observed in
some lines, semi-sterility is not typically problematic.
Both progeny and parents were grown under standard greenhouse conditions (i.e.
supplemental lighting, extended day length, and daily waterings) in 4-inch (10.2cm.) pots
until the plants were approximately 1.0cm. tall. DNA was extracted from fresh leaf
tissue using CTAB method described in Chapter 2 and/or by (Torres et ah, 1993). DNA
was dissolved using IOOpl of TE (I OmM Tris, ImM EDTA). RNA contamination was ■
eliminated via RNase treatment as previously described (Chapter 2 Material and
Methods).
.
The presence of genomic DNA from the extraction was checked using gel
electrophoresis of a 5pi aliquot of extract. Concentrated genomic DNA was stored at
-20°C in TE. The genomic DNA stocks were further prepared for enzymatic
109
polymerization reactions (PCR), by diluting the template by 50-75% in OdH2O. These
working stocks were kept for up to a month at 4°C.
Primer Characteristics
Primers sets designed to amplify anchored STS markers in Pisum (Brauner et ah,
2002) were used to target homologous loci within the lentil genome. These primers were
designed to hybridize to conserved exon sequences within the gene and amplify through
at least one intrbn. Improvements to primer design were carried out using BLAST
(www.ncbi.nih.org/blast). to detect areas of sequence conservation. Primerfmder v0.07
(http://eatworms.swmed.edu/~tim/primerfmder) was used to generate a list of priming
sites located within the conserved sequence with regard to primer size, annealing
temperature, and likelihood of self-annealing. Primers deemed suitable for mapping
retained minimal hairpin tendency, an annealing temperature close to 60°C, a GC ratio of.
greater than 50%, and a length of between 18 and 24 nucleotides. A complete list of STS
primers used from Brauner et al. (2002), amplification conditions, and methods used to
detect polymorphism, can be found in Table 3 A&C (see Results section). In addition,
rDNA primers designed to amplify the NTS between 18/5.8/25s arrays and two novel
markers: PsPk-2- (P. sativum Protein Kinase) and Pin-1 (Protein Kinase Pinoid),
.
generously donated to our program from Dr. D. DeMason (UC-Riverside) were
developed from Pisum and Arabiclopsis sequences respectively. The primer sequences of
P M , PsPk-2, and NTS :(18-5.8-25s) can be found in Table 3B (see Results section).
HO
STS Analysis
PCR conditions were carried out using reaction mix and cycling conditions
similar to those described in Chapter 2 (Materials and Methods). Parents were
investigated for length polymorphism by amplifying each allele in a 25 pi reaction which
allows enough product for electrophoretic analysis and, if necessary, a subsequent CAPS
(Cleaved Amplified Polymorphic Sequence) analysis. Expected fragment size of the
amplification using the lentil parents could be predicted based on size of the band that
was generated when primers were applied to Pisum templates (Brauner et al., 2002).
Five microliters of PCR product was applied to a 1% agarose gel and size evaluated using
electrophoresis. Positive length polymorphism indicated that the segregation of the
progeny at the respective locus could be revealed directly, using the STS primer set on
‘
each progeny line. However the majority of markers tested required an additional CAPS
analysis to reveal sequence polymorphism among progeny.
Cleaved Amplified Polymorphic Sequence (CAPS) Analysis
Five microliters of the PCR product was digested using a series of restriction
endonucleases that recognize-a four-nucleotide sequence. Such sequences occur, on
average, every 256 basepairs and these restriction enzymes are referred to as common
cutters. Refer to Table BA (see results section) for the specific enzyme that revealed the
polymoiphic sequence within each amplified STS sequence. Up to five CAPs analysis
can be assembled from the original 25pl parental amplifications. Reactions were set up .
as follows:
Ill
MasterMix (nl/rxn)
+PCR Product
5-10
+IOx PCR buffer
2.5
+ Enzyme
’
0.15
+ddH2Q__________ 12.5-17:5
Total: 25,Lil
■ Twenty-five microliter reactions were allowed to digest for one hour at 37°C.
Restriction fragments were applied to a 2% agarose gel and size fractionated using gel
electrophoresis. The progeny were all restricted with same endonucleases that revealed
sequence polymorphisms in the parents.
-
Genetic Mapping of Segregating Alleles
Progeny were scored based on allelic segregation patterns displayed at a particular
locus within the RIL population. STS markers nearly always exhibit co-dommate
characteristics; thus any remaining heterozygosity in the RILs should be revealed.
Segregation data was entered in a mapping program QUIKMAP EXCEL macro (N.
Weeden and J. Barnard, unpublished) as a character (F or S) representative of the allqle
inherited from Lc#7 or Le#32 parent, or in some cases, as heterozygote (H). The parental
segregating data confirmed both the size of STS/CAPS fragments produced by the
progeny, as well as defined the phase of the two loci displaying linkage. QUIKMAP
EXCEL arrayed the segregation data a linear fashion that facilitated the visual grouping
of linkage clusters . The program could also be used to predict relative gene order within
a linkage cluster by allowing one to manually arrange the data sets in such a way that
both minimizes the recombination between markers and reduces the number of double
crossover events among linked markers. The ability to re-arrange the data sets is of
,
112
particular importance when mapping within the vicinity of a translocation. Data from
groups of loci that are within each of the reciprocally translocated regions should display
two distinct linear marker arrangements that do not appear to assort independently. Thus,
most mapping programs will erroneously arrange most loci within the vicinity of a ■
translocated region, into a single linear linkage group. QUIKMAP facilitates the
assemblage of multiple linking arrangements allowing the investigator to propose a
marker arrangement for loci within the interchange complex. Previously generated Fg
morphological, isozyme (some F2) and RFLP.data, generated from the same lines were
integrated to both intensify marker density and confirm integrity of mapping our
population. RAPD markers generated by using the Operon® Primer series A,B,C,D
(Operon Technologies) facilitated the closure of gaps between linkage blocks. GMENDEL 3.0 (available as freeware www.css.orst.edu/G-mendel) confirmed the locus
ordering that was assembled using QUIKMAP EXCEL and more importantly generated
map distances between markers using recombination frequencies among linked loci. The
linkages that could be detected were only accepted under a LOD score of >2.5 and a
maximum recombination value of 0.20. G-MENDEL was not used for loci linked to
translocations or in regions exhibiting distorted segregation ratios.
113
~
:
Results
Thirty-three Pisum STS anchor markers (see Table 3) as described by Brauner et
al. (2002) were tested for linkage within lentil RIL mapping population (Lc#7xLe#32)
derived from L. cuUnaris x L. ervoides cross. Of the 33 STS markers initially tested, 20
-
markers displayed segregation among progeny. The segregation data were integrated
with 12 isozyme (Aatp, Ante, E stl, Est2, His I, Lap I, Lap2, Pepl, Pgdp, Pgmp Prxl,
Skdh), two RFLP (Lb, RbcS), two protein markers, (K9 and PT), and two morphological
(Tc and Gs) markers. All these latter markers with the exception of K9 and Pi, had been
previously anchored in pea as well as mapped within this lentil population (Weeden et al.,
1992). Twenty-six RAPD markers generated from progeny testing of the Lc#7xLe#32
cross, were used to Supplement the named markers with the intent of closing gaps
. . .
between distantly linked .markers. The complete integrated compilation Of the described
i
•
genetic markers resulted in a total of 77 markers that were tested for linkage within this
study.
;
Linkages were detected among 64 of the segregating markers. The markers were
.
assembled into 10 separate, multilocus groups; eight linear (Figure 16) and two complex
(Figure 17A and B), which span an estimated 300 discontinuous centimorgans. Thirteen
!
pea anchor STS markers could not be mapped within the population. Five of these STS
markers did not produce sufficient amplification products, and eight markers remained
monomorphic.even after CAPS analysis (see Table 3C).
,
114
■Table 3. STS PCR Primer and Product Characteristics
Gene
Sym bols
Apy
Apx 1
PAmyI
. PFruct
CipPor
Dimin
Enol
Gpi
Lectin*
Lip (CopI)
MnSod*
P628
P393
PhyApo
Pin 1
PsPk2
18s/25srDNA
Rpl22*
Sahh*
TubAI
Pisum Linkage
Group
V
■ ■V ll
VII
III
III
VllV
Vl
V ll
I
III
IV
IV ■
Il
■ Vl
V
IV & V ll
Vl
V ll
III
Annealing CAPS
Temp (C) Enzyme
59
60
58
60
58
59
60
60
60
60
58
55
55
59
57
58
60
58
58
60
Alu I
Hae 111
Alut
Rsat
Haettl
Ddet
. Haettt
Cfol
•
C fol
M bol
A fo l
N/A
C fol
C fo l
Dde I
H a e lll
N/A
N/A
A fo l
C fo l
Polymorphic
Fragment(s) Size(bp)
600/500
1200/1350
250/350
600/300
1100/550
500/400
1100/825
850/800
400/500
1000/450
800/700
670/550/350
450/380
900/600
1000/700
1200/1000
4000/3800
1400/900
1100/650
■ 1100/900
N/A- PCR Products exhibit polymorphism without additional CAPS analysis
*Unable to confirm homology of segregating product to designated gene_________
B.
Primer Sequ en ces
Pin I
Forward 5' A T G A TG G G TG G G TG G A A G A A A
Reverse 5’ G C TTG A A TA A G TG TTTG G G TTT
PsPk2-4
Forward 5' TG ATC G G TG TTC TG G G A TG A
Reverse 5’ G C C AAG TTAAC C C AAC AAA
25s-18s NTS 18s Reverse 5TG TC A C TA C C TC C C G G TG
_________________ 25s Forward 5’G TTTAG A C C G TC G TG A G A________________
C.
Pea STS Anchor Markers Not Mapped in Lentil
Primer Designation
Reason
M o n o m o rp h ic "
Fbpp
M onom orphic**
Pepcn
No A m plification
Paal 1,2
M onom orphic**
Rcpme
No A m plification
Gsc
M onom orphic**
Hopl
M onom orphic**
Pyrols
M onom orphic**
Vic J/K
No A m plification
Drr49
M onom orphic**
Enod 40
M onom orphic**
Gib2H
No A m plification
Ivdh.
No A m plification
Gdct
M onom orphic**
VR***
________________________________________________
M onom orphic**________
MtST-1***
**Monomorphic characterisics maintained through CAPS analysis
***STS anchor markers from M e d ic a g o tru n c a tu la _________________
_____
Linear Lentil Linkage Groups
-fDimin
Centimorgans
o-
wEnol
-S-O P D ielll
5_
10-
-OPA2*
-OPA9,
-Lapl
15_
-O P B lSlloc
20_
25.
1 -O P B 1 7 „
-Apxl
p1
&
-OPDS0
-p628a
-OPA2
-Hisl
35-
45-
L
-OPD5 1100
30_
40.
-OPA7 300
-*-OPD7,
-OPD5.
-p393
-OPA2,
X-Apy
50_
-#-OPB8 1000
VII
V
VII
VII
Figure 16. Lentil Linkage Groups. Color of bar and roman numerals indicate syntenic Pisum linkage group, (see Figure
19) Bold = STS marker. Areas of unknown synteny represented by black bars. Dotted lines indicate probable linkages
116
A) VH Interchange.
Copl —
—
C opl
L. culinaris
L. ervoides
Linkage Group I
l b __
Linkage Group I
— Lb
Gs —
— Gs
A a tp
A aIp
Yc
E st2
Pgmp
Yc
E st2
Pgmp
rDNA
rDNA
L. culinaris
L. ervoides
Linkage Group 2
Linkage Group 2
P hyA po
BI V/VI Interchange.
P hyA po
—
o p d i 6 1800_
— o P D ie 1
O P D ie 930-
— O P D ie 9
O P B l330
- O P B l 330
O P B l300
----O P B l300
O P B 18
O P B 1 8 i300
^^P xA
P inl
P inl
OPDT730-
"O P D T 730
O PD T113-
"O PD T 1130
PvPA2 -
-PsPk2
O P A lO 930
i30— I
fiFrucl
GP'
RbcS
Gpi
I— O
OlP A lO 930
CipPor
CipPor
Rpl22 =
RbcS
J
t
Rpl22
-fiFruct
Figure 17 A) Translocation quadrivalent involving linkage groups I/II. B) Translocation
quadrivalent involving linkage groups V/VI. Color indicates syntenic Pisum linkage group
(see Figure 19). Black=Unknown synteny. Bold=STS marker. Dashed Iine=Probable
linkage.
I
.
117
Three markers (Sahh, MriSod, Lap-2) did display segregation among progeny, however
could not be linked to any of the assembled linkage clusters under the described mapping
conditions. PFruct and Enol showed linkage to the cluster indicated in Figure 17B and
Figure 16 respectively, however only under very low stringencies (LOD<2.0 and max
recombination value 0.20), which is why they only are indicated as probable linkages.
Z
Within the multilocus linkages, 32 homologs, consisting of STS, RFLP, and isozyme
markers, were identified that exist between Pisum and Lens which allows a comparison
of their positions on the lentil linlcage map.
Two reciprocal translocations (I/II, V/VI) appear to be present within the cross,
accounting for two of the ten assembled linlcage groups. The loci intimately involved
within the reciprocal translocations, particularly the V/VI interchange, exhibited distorted
segregation that skewed away from the expected Fg of 1:1 (See Table 4 and Appendix B ).'
The segregation heavily favored the L. culinaris allele (see Figure 18). When mapping
within the distorted areas, the data had to be treated differently than standard segregation
data when using conventional methods of joint segregation analysis. By comparing only
the data points that exhibit the underrepresented allele, the markers could be assigned
relative positions within the linkage cluster that would minimize double crossover events
(Livingstone et ah, 2000). Although ordering of the loci was possible within the distorted
regions, estimating genetic distances between such loci, based on recombination
frequencies was impracticable and would have been grossly inaccurate if standard
equations were used.
118
Table 4. Segregation of markers from cross L. culinaris x L. ervoides
Marker
STS
N Segregation 3 G Valueb
Marker
RFLP
Apy
46
3 6:1 0
15.6***
Apxl
PAmyI
36
16:20
0.44
Lb
RbcS
49
27:2 2
0.51
Protein
PFrue t
40
30:20
2.01
CipPor
. Dimin
48
4 0 :8
36
Enol
Gpi
Lectin
Lip (Cop1j
N Segregation 3 G valueb
8
5:3
0.51
49
36:1 3
11.32**
K9
42
2 5 :1 7
1.53
2 3.2 8***
P1
42
19:23
0.38
13:23
2.81
Morphological
37
45
26:11
31:14
6.26*
6.58*
Yc
Gs
50
50
.26:22
0.35
3.03
37
2 1:1 6
0.67
38
2 0:1 8
6.11
Mnsod
40
3 2:1 8
P628a
47
18:30
RAPD
-
O P A 2 550
46
2 2:2 4
0.08
3.97*
OP A2y4Q
46
25:21
0.34
21:2 6
0.53
OPA2goo
46
2 0 :2 6
0.78
P393
49
23:26
0.18
OPA73oo
46
PhyApo
48
28:2 0
1.34
O PAO 290
46
Pin 1
46
39:7
24.5 3***
O P A l Ogg0
31:15
5.68*
25:21
0.34
45
36:9
17.34***
.
Ps P k2
46
4 4 :2
0.18
O P B I 350
46
4 1 :5
32.14***
1 8s/2 5srD N A
45
29:1 6
3.81
O P B I 500
46
39:7
24.53***
Rpl 22
39
33:6
2 0.5 7***
O P B 155Q
46
26:2 0
0.78
Sahh
31
22:9
5.62*
O R B I850
46
26:2 0
0.78
TubA 1
37
11:26
6.26*
OPB755q
45
23:2 2
. 0.02
OPBS-I0QQ
46
2 9 :1 7
3.16
Isozyme
Aatp
44
21:2 3
0.09
O P B S 900
46
2 4 :2 2
0.08
Aatc
46
20:2 6
■ 0.78
OPB17780
46
32:1 4
7.23**
Es tl
40
21:2 9
1.28
O P B V 900
45
25:2 0
0.55
E st 2
46
23:2 3
0
O P B 1S-I100
44
22:22
0
His 1
45
32:13
8.27**
O P B 1S150Q
46
39:9
16.41***
Lapl
46
35:11
13.16***
O P D S 1100
42
2 3 :1 9
Lap2(F 2)
20
9:11
0.2
O PDS^00
42
21:21
0.38
b
PepI (F2)
32
16:16
0
O P D S 950
42
27:1 5
3.47
Pgdp
45
18:27
1.81
O P D 7 1150
46
37:9
24.53**
Pgmp
25
11:14
• 0.63
O P D 7 550
46
26:2 0
0.78
,
Px1
44
35:9
16.41***
OPD775q
46
4 2 :4
36.58***
Skdh
45
15:30
5.09*
O P D 1 S1300
46
34:12
10.96***
O P D 1S300
46
2 7 :1 9
1.39
O P D 1 S950
46
38:8
21.26***
aExpected segregation for all markers: 1:1 bG value is the 'GMENDEL' version of %2
analysis (see appendix for formula). *P < .05; **P < .01; ***P < .001.
119
Farenti
^tc7Lei)2 Progeny-
I
1
.
Ww
#
F ig u r e 18. C i p P o r P C R a m p lif ic a tio n p ro d u c ts d ig e s te d w ith H a e \ \ \ d is p la y in g d is to rte d
s e g r e g a tio n a m o n g p r o g e n y . P a re n ts L c # 7 a n d L e # 3 2 o n le ft w ith R IL s a r ra y e d to th e
rig h t.
T h e n u m b e r o f lin k a g e s th a t d is p la y s e g r e g a tio n d is to r tio n w ith in th is c r o s s s u g g e s ts th a t
a p p r o x im a te ly 2 5 % o f th e le n til g e n o m e w o u ld b e a ffe c te d .
N in e m a r k e r s d is p la y e d lin k a g e to th e tr a n s lo c a tio n I/I I q u a d r iv a le n t, w h ic h
a llo w e d th e r e s o lu tio n o f m a r k e r p o s itio n s o n e a c h o f th e fo u r a rm s . B y c o m p a r in g
lin k a g e s to p r e v io u s m a p p in g re s u lts w ith in th is p o p u la tio n (W e e d e n e t a l., 1 9 9 2 ), th e s e
d a ta a llo w e d th e p r o p o s a l o f p a r e n ta l c h r o m o s o m e s , a s w e ll a s a s s ig n m e n t o f a lin k a g e
g ro u p to e a c h o f th e f o u r a r m s w ith in th e q u a d r iv a le n t (S e e fig u re 1 6 A ). S ix te e n m a rk e rs
d is p la y e d lin k a g e to th e p u ta tiv e V /V I tra n s lo c a tio n c o m p le x . H o w e v e r th e s e 16 m a rk e rs
c lu s te r e d in to th r e e lin k a g e b lo c k s , th u s o n ly th re e o u t o f th e f o u r a r m s o f th e
q u a d r iv a le n t c o u ld b e r e s o lv e d (s e e fig u re 1 6 B ).
M o s t m a rk e rs w ith in th e q u a d r iv a le n t
a p p e a r r e a r r a n g e d w ith r e s p e c t to p e a a n d h a v e y e t to b e r e p o r te d in le n til, c o n s e q u e n tly
th e p a r e n ta l d o n o r s a n d s y n te n ic p e a lin k a g e n u m b e r s c o u ld n o t b e a s s ig n e d a n y o f th e
a rm s
T h e c o lo r - c o d e d lin k a g e g r o u p s o f le n til c o r r e s p o n d to a r e a s o f s h a r e d c o n s e rv e d
g e n e s y n te n y w ith r e s p e c t to th e
P is u m lin k a g e m a p , a llo w in g lin k a g e c lu s te r s fro m len til
to b e a lig n e d to a r e g io n o f a p e a lin k a g e g ro u p (s e e F ig u r e 19). E a c h p e a lin k a g e g ro u p
120
with the exception of linkage group VI has at least one region displaying synteny to
lentil.
C onsensus L inkage M ap o f
I
Il
III
IV
Pisum sativum
V
r
Vl
Vll
Figure 19. Consensus linkage map of P. sativum (Brauner et al., 2003). Areas syntenic in
lentil are denoted with colored bars. Hashed bars indicate probable linkage in lentil.
Brackets are regions that displayed considerable segregation distortion
When the lentil linkage clusters are ordered similar to that in pea, we observe a
span of at least 50cM per pea linkage group, although estimates of linkage coverage
within the translocation complexes cannot be verified. The lentil linkage blocks that
121
were generated by this study and display conserved gene synteny to pea linkages, are
summarized on Table 5.
Table 5. Conserved Linlcages Between Lentil and Pea
Linkage
Groups
I
Jl
III
IV
V
Vl
Vll
P is u m
Syntenic Linkages Detected In Lentil
C o p l-L b -G s
P hyA po-A atp
T u b A I-A a tc
C ip P o r--^ F ru c t
P 6 2 8 -P 3 9 3
A p y -H is t- -E noI
P xA -R b cS
N fA
D im in -A p x t -(IAm yI
S k d h -E s tt
*Double dashes indicate probable linkages.
P g d p -P e p t
Absence of synteny was observed as a number of single and multilocus
rearrangements.. The 45 S ribosomal cluster, which is anchored on both linkage groups IV
and VII in pea, is linked to the Yc:Est2 cluster within the lentil LGI/II translocation
complex. Two loci (Gpi and CipPor) displaying linlcage to the translocation V/VI
complex appear to possess different positions relative to the consensus pea linlcage map.
The Gpi locus did not show a clear linkage to either Pinl or PsPk2 as indicated on the
pea map, but could be placed within.the same linlcage block via the PxA-RbcS linkage
(See Figure 17B). The CipPor locus, anchored to linlcage group III in pea, shows linlcage
to the linkage group V/VI complex in lentil. In addition to the rearrangements at the
single locus level, several linkage groups IcnOwn to be syntenic in pea could not be
connected in lentil (see Figure 19). Specifically the TubAl, Lapl, and CipPor groups (LG
122'
III in pea), Apy and PxA groups (LG V in pea) and the Pdgp, Skdh and Apxl groups (LG
VII in pea), each syntenic in pea, could not be j oined in lentil.
123
Discussion
The intent of this Study was to evaluate the hypothesis of complete shared synteny
among closely related members of the tribe Vicieae. The project adopted the
phylogenetic relationship between Pisum spp. and Lens spp. (Steele et ah, 2003) to
investigate STS marker linkage conservation between the two divergent species. To
support the placement of the STS markers as well as increase and intensify linkages that
were detected, a number of isozyme, RAPD, protein and morphological markers were
integrated with the assembled data sets. The majority of these markers could be
assembled into 10 linkage groups that span over 300 discontinuous centimorgans. The
linkage distances within the assembled linkage groups imply a moderate coverage of the
lentil genome; however, five markers failed to reveal linlcage when tested under low
stringencies (i.e. LOD<2.0 and maximum recombination value of 0.20). This result
suggests that a minimum of I OOcM of the lentil genome remain untagged; thus,
additional regions need to be characterized before a complete linkage map can be
assembled. However considerable coverage was attained and areas of the generated
linlcages groups displaying both conserved gene synteny and absence of gene synteny
between Pisum and Lens were revealed.
Characterization of Cross Lc#7 x Le #32 '
The results generated from mapping within this intefspecies cross, originally
described by Ladizinsky, strongly suggest the presence of an additional translocation
(V/VI) involving different linkages than the translocation that was originally described by
124 :
Tadmor et al. (1987) and corroborated by Weeden et al. (1992). Tadmor et al. (1987)
characterized the Lc#7xLe#32 cross both cytologically and genetically as containing a
reciprocal translocation involving linkage clusters that correspond to Pisum linkage
groups I and II. The presence of the translocation (I/II) within the cross was confirmed
based on my data. However, the additional translocation was detected within linkage
clusters that display severe segregation distortion, and it was only after careful'
examination of these data that an additional translocation could be postulated.
Distorted segregation patterns that affected the described portion of the genome
caused some difficulty in resolving genetic distances. Distorted loci are not uncommon
within mapping populations, especially those that involve a relatively wide cross such as
L. culinciiis x L. ervoides. Eujayl et al. (1997 & 1998) reported significant divergence
from the expected Mendelian segregation ratios within two lentil mapping populations. •
However, the results from their study did not indicate that a particular parent had more
influence as to which allele was donated at a particular locus. Results of the present
study clearly demonstrate the prevalence of the Lens culinaris allele within the distorted
areas. The population used in Eujayfs study (Lehs oreintalis x Lens culinaris), however,
is considered genetically narrower than the cross that was used in our study.
The use of wide crosses for generating linkage maps is based on the-fact that
segregation is usually observed among more of the loci tested. The significant genetic
divergence of the two parents Lc#7 (Lens culinaris) and Le#32 (Lens ervoides) (Muehcli
et al., 1991) would suggest a considerable amount of expected segregation. However,
with nine STS markers remaining monomorphic through our CAPS analysis, it appears
125
that the interspecific cross described may actually be genetically narrower than an
intraspecific f . Jafivwm cross in which these same STS markers co-segregated (Bfatmer
et ah, 2002).
•
Conserved Gene Svntenv
In most cases, the linkage clusters, in'lentil confirm the established genetics'
relationships as described by Weeden et al. (1992), Tadmor et al. (1997), Tahir et al.
(1994) (see figure 15 and 16A,B). However, the loci that displayed genetic
rearrangement with respect to pea are of particular interest. The 45 s: array that is
anchored to linkage groups IV and VII in pea, does not show linkage to either of these
respective linkage groups in lentil. Rather my results strongly suggest that the 45s array
is subterminal and linked to the translocation involving linkage groups I and II in-lentil.
The position of the 45 s array is in agreement with the FISH results of the NOR in lentil
(see Chapter 3). Furthermore the linkage between Est and 45srDNA is conserved in
Viciafaba (Torres et al., 1993), however the homology Of the Av locus in lentil to Est
locus in Vfaba must be determined.
The rearrangement of the 45s array was first described by Weeden et al. (1992)
based on RFLP data. This rearrangement was verified using PCR primers designed to
'
amplify the NTS of between transcribed arrays. Additional rearrangements include the
Lapl cluster assorting independently from TubAl cluster. Similarly, the three clusters
that are syntenic on pea.linkage group VII, specifically the Dimin-Apxl- f Amyl, SkdhEstl, Pgdp-Pepl clusters, show no linkage to each other in lentil. Two rearrangements ■
126
within the V/VI translocation complex were detected: Gpi appears to be linked to the
RbcS cluster and the. CipPor-PFruct segment shows linkage to markers that reside on
linkage group VI in pea. Neither of these linlcages has been reported in pea. Finally,
. regardless of the arrangement within the translocation, linlcage groups I and II have
changed in X. culinaris and linleage group II appears incomplete in both the parents. The
wild species (Z. ervoides) maintains the proposed consensus gene arrangement '
represented by pea at linkage groups I arid II, and Z. culinaris appears restructured.
Discrepancies in Lentil Genetic Mapping
1
'
Several linkages detected within our study challenge previous marker positioning
and chromosomal arrangements. The most intriguing of these is represented by the
addition of a second translocation. Tadmor et al. 1987 provided cytological evidence for
one translocation in which the satellited chromosome containing the NOR (and
presumably the 45s array) was clearly separated from the quadrivalent that was under
investigation. As previously stated, however, our evidence as well as Weeden et al.
(1992) place the 45s array within the translocation Vll complex and not on linkage groups
VII as seen in pea. The detection of an additional translocation between linkage groups
V and VI suggests that the photograph of the translocation documented by Tadmor and
characterized as a linkage group I/II interchange may actually have involved the
interchange between linkage groups V and VI, This novel translocation would explain
how the NOR would be cytologically distinct from a translocation while appearing
genetically linked to the translocation. One method to test this hypothesis would be to
127
examine a number of meiotic chromosome spreads from lines within the Lc#7 x Le#32
population for evidence of an additional quadrivalent, particularly those lines that exhibit
infertility; typically indicative of a translocation. Additional inconsistencies are
summarized as follows':
1. Tahir et al. (1993) placed Rrn on a different linkage group than Yc or Pgmp. My
results indicate that Yc:Est2:Pgmp:rDNA are syntenic within the I/II translocation
complex (see Figure 17A).
2. Weeden et al. (1992) indicated that Estl was syntenic to RbcS. My results
positioned of RbcS within the V/VI translocation complex. This area does not
share synteny with Estl which was anchored to Pisum linkage groups VII See
Figure 17B).
In summary, linkages were detected among 64 segregating markers,, 32 of which
had been previously anchored on the P. sativum Hnlcage map. Consequently, 10 linkage
groups were assembled that span over 300 discontinuous centimorgans. Our evidence
indicates that while the majority of loci evaluated share linkage synteny with pea, some
notable genetic rearrangements were revealed. Supplementary to this study, I generated
evidence demonstrating the presence of two reciprocal translocations between the two
parental lines. This, investigation represents the most.current documentation of conserved
gene synteny within the tribe Vicieae and begins to bring resolution to inconsistencies
regarding the genetics of this interspecies lentil cross.
128
Literature Cited
Comparative ,rak ag e
o f th e * = m d
Brauner S., R.L. Murphy, J.G. Walling, J. Przyborowski, N.F. Weeden. (2002) STS
Markers for Comparative Mapping in Legumes.' J. Amer. Soc. Hort. Sci. 127 :616-622.
Bonierbale M.W., BLI,. Iilaisted, S.D. Tanksley. (1988). IRiFLP maps based on a common
set ot clones reveal modes of chromosome evolution in potato and tomato. Genetics
Ellis, T.H.N., SJ . Poyser. (2002). An integrated and comparative view of pea genetic
and cytogenetic maps. New Phytologist 153:17-25.
Eujayl I., M. Baum, W. Erkskine, E. Pehu, F.J. Muehlbauer. (1997). The use of RAPD
markers for lentil genetic mapping and the evaluation of distorted Fg segregation.
Euphytica 96:405-412.
Eujayl I., M. Baum, W. Powell, W. Erkskine, E. Pehu. (1998). A. genetic linkage map
of lentil based on RAPD and AFLP markers using recombinant inbreed lines. Theor
Applied Genet. 97: 83-89.
Hall, A.E., A. Fieberg, D. Pruess. (2002). Beyond the Arabiclopsis Genome:
Oppurtunites for Comparative Genomics. Plant Physiol. 129: 1439-1447.
Hauge, B.M, SM. Hanley, S. Cartinhour, J.M. Cherry, H.M. Goodman. (1993). An
integrated genetic/RFLP map of Arabidopsis thaliana genome. Plant J. 3 :7 4 5 -7 5 4 .
Halt, G.E. (1979). Genetical and chromosomal realtionships among the wheats and their
relatives. Stadler Genet. Symp.
Havey M.J., F.J. Muehlbauer. (1989). Linlcages between restriction fragment length
isozyme and morphological markers in lentil. Theor. Appl. Genet 72:761-76.
Hollard, J.B,, S.J. Helland, N. Sharopova, D C. Rhyne. (2001), Polymorphism of PCRbased markers targeting exons, introns, promoter regions, and SSRs in maize and introns
and repeat sequences in oat. Genome. 44:1065-1076.
Iwata5H., T. Ujino-Ihara, K. Yoshimura, K. Nagasaka, U. Mukai, Y. Tsumura.
(2001). Cleaved amplified polymorphic sequence markers in sugi, Cryptomeria japonica
D- Don, and their location on a linkage map. Theor. Appl. Genet. 103:881-895.
129
KowdsJd S.T., T-H. Lan, K.A. Feldman, A.H. Peterson (1994). Comparative Mapping
oi Arabidopsis thaliana and Brassica oleracea Reveals Islands of Conserved
Organization. Genetics 138:499-510
Ladizinsky, G. (1979a) The origin of.lentil and its wild gene pool. Euphytica 28:179187.
Ladizinsky, G. (1979b). The genetics of several morphological traits in lentil
Heredity 70:135-137.
J
Ladizinsky, G., D. Braun, D. Goshen, FJ. Muehlbauer (1984). The biological species of
the genus Lens L. Bot Gaz. 154:253-261.
Ladizinsky, G., D. Cohen, F. J. Muehlbauer (1985). Hybridization of the genus Lens by
means of embryo culture. Theor. Appl. Genet. 70: 97-101.
Livingstone, K.D., G. Churchill, M.K. Jahn. (2000). Linkage mapping in populations
with karyotypic rearrangements. J. Heredity 9.1:423-428.
Muehlbauer,, F.J., N.F. Weeden, D.L. Hoffman. (1989). “Inheritance and linkage ■
relationships of morphological and isozyme. J. Heredity 80:298-303.
Mysore KS, Tuofi RF, Martin GB (2001). Arabidopsis: Genome sequence as a tool for
functional genomics in tomato” Genome biology 2:1003.1-1003.4
Polans, N.O., N.F. Weeden, D.L, Hoffman. (1989). Inheritance and linkage
relationships of morphological and isozyme loci in lentil Lens (Miller)! I Hered. 80:298303.
Prince^ J.P, E. Pochard, S.D. Tanksley. (1993). Construction of a molecular linkage
map of pepper and a comparison of synteny with tomato. Genome. 36:404-417
Simon, C.J., F.J. Muehlbauer. (1997). Construction of a chickpea linkage map and its
comparison with maps of pea and lentil. J. Hered. 88:937-945.
Sherman, LD., A.L. Fenwick, D.M. Namuth, N.L.V. Lapitan. (1995). A barley RFLP
map: alhgnment of three barley maps and comparisons to GramineaespecieS. Theor.
Appl. Genet. 91:681-690.
.
Small, RA., J.F. Wendel. (2000). Copy number liability and.evolutionary dynamics o f
the Adh gene family in diploid and tetraploid cotton (Gossypium). Genetics 155T 9131926.
Steele, K. P., and M. F. Wojciechowski. (2003). Phylogenetic systematics of tribes
Trifolieae and Vicieae (Fabaceae). Advances in Legume Systematics. (in press).
130
Tadmor Y., D. Zamir, G. Landizinsky (1987) Genedc mapping of an ancient
translocation in the genus Lens. Theor. Apph Genet. 73:883-892.
Tahir M., C.J. Simon, F.J. Meuhlbauer (1993). Gene map of lentil: a review. Lens
Newslett. 20:3-9.
TTanlcsley SI), IBerriatzky', TL, Lapitan, NL, Prince, JP (1988). Conservation of gene
repertoire but to gene order in Pepper and tomato. PNAS 85:6419-6423.
Tanksley, S.D., M.W. Ganal, J.P. Prince, M.C. de Vicente, M.W. Bonierbale, P. Broun
T-M- Fulton, J.J, Giovannonh S. Grandillo, G.B. Martin, R. Messeguer, J.C. Miller L ’
Miller,. A. H. Peterson, 0. Pineda, M.S. Roder, RA. Wing, W.Wu, N.D: Young. (1992).
High density molecular linkage maps of the tomato and potato genomes Genetics
132:1141-1160.
Taylor, C., K. Madsen, S. Borg, M.G. Moller, B. Boelt, P.B. Holm. 2001. The
development of sequence tagged sites (STSs) in Lolium perenne L.: the application of
primer sets derived from other genera. Theor. AppL Genet. 103:648-658.
Torres, A.M., N.F. Weeden, A. Martin. (1993). Linkage among isozyme, RFLP, and
RAPD, markers in Vicia faba. Theor. AppL Genet. 85:937-945.
Tsumura, Y, N. Toniaru. (1999). Genetic diversity of Cryptomeria japonica using co­
dominate DNA markers based on sequenced-tagged sites. Theor. AppL Genet 98 396404.
'
'
Vincent, J.L., M.R. Knox, T.H.N. Ellis, P, Kalo, G.B. Kiss, N.J. Brewin'. (2000).
Nodule-expressed Cyp-ISa cysteine protease genes map to syntenic genome regions in
Pisum andMedicago spp. Mol. Plant-Microb. Interact. 13:715-723.
Walling, LG. and N.F. Weeden.. 2002. Conserved synteny suggests that Gs in lentil is
homologous to D in Pea. Pisum Genetics.
Weeden, N.F,, FJ. Muehlbauer, G. Ladizinsky. Extensive conservation of linkage
relationships between pea and lentil genetic maps. J. Hered. 83:123-129.
Zamir D., G. Ladizinslcy. (1984). Genetics of allozyme variants and linkage groups in
lentil. Euphytica. 33:329-336.
131
CHAPTER 5
EXPERIMENTAL SUMMARY
The previous chapters described three separate investigations into Lens and
Pisum. Each investigation was designed to address a current issue that is of particular
interest and benefit to workers associated with these related legume species. Some of the
most current methods and technologies available to geneticists, including novel mapping
markers, a large insert genomic library, and fluorescent microscopy were integrated into
the experimental designs. The results of the first two experiments will provide a
foundation for future experimentation, while the third experiment brings resolution to
studies initiated prior to this investigation.
The intent of the first experiment was to assess the utility of a newly developed
Piswn sativum BAC library. At the time of experimentation, the coverage of the library
was estimated at one pea genome equivalent. Although the low coverage somewhat
limited the extent to which the library could be characterized, initial assessments could be
:
accomplished; the following three characteristics of the library were examined: first, the
abundance of chlbroplast DNA within the library was examined to determine the utility
of the library when screening for nuclear DNA. Second, the ability to locate nuclear
sequences of high copy number, in this case the 25s rDNA subunit, was investigated.
Finally, the ability to locate clones containing sequences of both low copy (gene family)
and single copy numbers within the library was investigated. The results from these three
experiments indicated that I) a minimum of 1,0% of the clones within, this BAG library
would contain cpDNA sequences', 2) 25 S rDNA (high copy) clones were obtainable and
132
represent approximately 0.35% of clones within, the library, and 3) one representative of
both a gene family and a unique sequence was found; RbcS representing a gene family
and Gsc representing a single copy gene. Although these results are of some value to
subsequent investigations, of greater value will be the data obtained regarding the
screening process itself. As indicated in Chapter 2, there were a substantial amount of
false positives detected using the filter hybridization procedure. False positives will
certainly encumber any future large-scale screening experiments by significantly
decreasing the precision and efficiency to which homologous BAC clones are readily
identified. Thus, any investigation using this library must address the issue of false
positives and adjust the experimental design accordingly. Portions of this experimental
design that may have contributed to the abundance of false positives have been, suggested
(see Chapter 2 Discussion), including the hybridization procedure or acquisition of
clones. Before the inception of a large-scale screening initiative using this library,
alternate methods of clone identification and verification should be considered.
The second experiment utilized the partially-characterized BACs obtained from
the first experiment to address one potential use of P. sativum BACs in future genomic
studies. This experiment questioned the utility of using FISH to detect low copy
sequences located within BACs on P. sativum chromosomes. The effect of adding
increasing amounts of blocking DNA to the FISH reaction was also investigated. A
series of FISH experiments was set up to evaluate the behavior of these BACs under
different hybridization conditions with the intent of developing a protocol for future
BAC-FISH experimentation specific to Pisum, Additionally, the value of several plasmid
133
probes was tested for possible future use as positive checks. The large amount of
repetitive sequences found in both the BAC insert and the pea genome significantly
cross-hybridized with each other. This result indicated that, when compared to species
with smaller genomes in which FISH is done routinely, the same approach in pea would
not be trivial. Methods to overcome the binding of repetitive seqeunces, such as
sequestration techniques' (i.e. Q t-I blocking DNA), were largely ineffective. However,
one FISH reaction using the RbcS BAC probe displayed relatively localized signals when
used in conjunction with large amounts of Q t-I blocking DNA. As discussed in Chapter
3; these data suggest two.future areas of investigation: I) a thorough characterization of
the insert sequence within the BAC probe, and 2) a more robust blocking DNA extraction
that would target Q t values significantly greater than Q t-I (e.g., Q t-100).
The final investigation addressed the hypothesis of conserved gene synteny
among members of the family Vicieae (Pisum and Lens). To permit the most thorough
investigation of a macrosyntenic gene arrangement between the two species, a novel
linkage map of Leris was generated primarily using a set of Pisum STS anchor markers
on a Lens RIL mapping population. These STS data were integrated with previously
generated isozyme, RFLP, and protein data, for most of which the homo log in pea has
been reported. The map was completed using RAPD markers tested on the (same lentil
mapping population used to test the other markers to fill gaps between loosely linked
markers. Linkages were detected among 64 segregating markers, 32 of which had been
previously anchored to the Pisum linkage map. These data allowed the assembly of 10
linlcage groups, eight linear and two complex, which span an estimated 300 discontinuous
134
centimorgans. The results confirm many of the previously established gene linkages, and
areas of conserved gene synteny between pea and lentil were detected. More
importantly, however, absence of synteny was observed in a number of single and ■
multilocus rearrangements that differed between the two species. Thus, evidence was
found that would reject the hypothesis of complete shared gene synteny between lentil
and pea. In addition to the comparative mapping results, data was obtained that further
characterized the cross that produced the RIL mapping population. An additional
translocation involving different chromosomes than those involved in the previously ■
reported interchange was mapped using our population. Although this translocation will
need to be further confirmed cytologically, the evidence generated from the mapping data
strongly indicates the presence of the additional interchange! The results of this
experiment represent both the most current documentation of conserved gene synteny
within the tribe Vicieae and most complete characterization of the original lentil cross.
In conclusion, the experiments'described appear, for the most part, to be'
unrelated. One exception being the clones obtained from the BAC experiment was used
in some of the FISH experiments. However one could envision a.future experiment in
which a lentil-anchored marker from the newly developed linkage map is used in the
positional cloning of its homolog in pea and subsequently physically localized on
chromosomes of pea or lentil using FISH. Whether the results obtained from each
experiment establish a new pathway of investigation or bring further resolution to
discrepant previous investigations, these results will nonetheless provide a significant
contribution to the current status of genetics in Lens m d Pisum.
135
APPENDICES
136
APPENDIX A
RAW BAC INSERT SEQUENCE
137
RAW B A C
C lo n e 8 9 -E lO F o r w a r d
p rim e rs
(832 bp)
5
Sequence
Sequence
DATA
a m p lifie d
u sin g
G sc
s p e c ific
ANGGGAGNAGCACNATGGAGGNGCAGGATCNGAGGGGGANGTTCTTTTATNGTATGA
CTAATTNAACTCAW TANTTAATTANGCNNNNTTTATAAAAAAAGTTATTTTTCCAAGAA
ACTTATGAGAGTTTCTTTGTTTTTTGTTTTCTTGTGTTTATTTGATGAAAAAAGTCCAC
ANGCTATTTTCAAGGACCCATTCAGAAGAGGGAATAATATTCTTGTAAGTTGTTCATTT
TTATTTTTTGAAAGAATTGGGTTATTAATTATAAATTGATTAAGAACATGGGTTCTAGT
TTATCTAATTATATTTATTTA TTCA TG TTA AA A A AA TTA ACTTTG TG G AA A A AA TA TTT
AGGTGATTTGTGATGTTTACACCCCAGCTGGAGAACCATTACCCACAAACAAGAGATAC
AATGCTGCCAAAATTTTCAGTCACCCTGATGTTGCTGCTGAAGTACCATGGTATGGGTG
TCATGTCGGATTTTTGTCGCATTCACACTGTCAAAATATTATTAACCTTTATCTTAATT
GCACATTTTTAAAAAAAAATTGAATTTTTTAAAATTATAAAATTTAAATTACAATTATT
ca a g a a ta a tta tta ccn ta a a ttta tttta ta ctg tca a ccca a tg a cg a cca ta ta t
CTCTGTTAATTCAAATTAAAATTTTTAAAATATTTATATGGACNTGATAAAATGATATA
ttttta tta g a a g a ta g ta ta a a a ctttttta g a g tg g cctttg g ta tccn cccta a a a
ATCCAAATTTTTAATGGGGATTTGGATTTTTCCTTTTAAAAACCCNAANCNAATTAAAG
GGGATN3 '
C lo n e 8 9 -ET0 R e v e rs e
p rim e rs .
(8 3 9 b p )
Sequence
a m p lifie d
u sin g
Gsc
s p e c ific
5 ' NAGAGGCNNCAATGCTGCGTNCNGTGC&TCGACGACGCCCNNCCATAAGCTTTATCA
GCACCAATTCCACAATAATATGGNGGNCTGGGNAATATAAAAATAATTAATAATTACAA
A A A T A A G c t t t aa t a t g a t t aa a a a a a c t G a a g a a a c a t a a t a a a a a a a t a a a g c a a a a
AAANNTGAAGAAAAATAATTAA A A A AA A A AA A G AA A A A AA A A NG TN TTTTN CTTA CCN G
TTTNCCNGGATANCCNCCAATNGGCCANCCTAGGGGCCNGTTAANGNCTTNCNGNAANA
aagnatacnctngttca
Attc
cn ta ccn gtncatataancaan caa aattaa ttttcat
tacaan attccn gttagn ctttataan aanaa aaaaccaaa tttangta attatta ttt
TNGGAANANCTTTAAAAANANGTTTTTAANAAANATNTTTTANGGANAGTNGGGATTAA
ttan cccttta ttn g cttg g g ttttta a a a n a a a a a n cca a a tcccn tta a a a ttta a a
TTTTANGGGGANACAANGACCCCCTAAAAAAGTTTTATACTANCNNCNAAAAAAAATAN
ATCATTTTANCNNGCCATAAAAATATTTTAAAAATTTTAATTTGAATTACCANAAATNT
ntg gccnccnttgg ttgacagg tancaaan aaattta tgnaa ataatta tncttngaa a
AATTGGNAATTTAAATTTTAANAANTTTAAAAAAATNCCATTTTTTTTTTAAAAAGNGG
G CN N TTA A A AN A A A AG G G TA A A TAA A N NTTTTTGN CCG G NG G G AA A N GCN CN CA A AA A N
C C N A A CA TG G N C C3'
138
C lo n e 103 K l F o rw a rd
p rim e rs .
(519bp)
Sequence
A m p lifie d
u sin g
RbcS
s p e c ific
5 ' GATTGCTNCTTCTNCAAGCAATGGTGGAAGAGTAAAGTGCATGCAGGTGACAGAAAC
ATATACATCTATTTGAATATCAGTAATGATTCAAGTTTGTTAACCGTTATTGTTGAATA
TGNAGGTGTGGCCTCCAATTGGAAAGAAGAAGTTTGAGACTCTTTCCTATTTGCCACCA
TTGACGAGAGATCAATTGTTGAAAGAAGTTGAATACCTTCTGAGGAAGGGATGGGTTCC
ATGCTTGGAATTTGAGTTGGGAGGTTTCATATTCATTCCTTTTTTCAATGATTCTATAA
ATAGTTTTGTTTGAAACCGTAATGAGTTGATTTTGACTGGTTTGGTTGCAGAAAGGATT
.T T G T G T A C C G T G A G C A C A A C A A G G T C A C C A G G A T A C T A T G A T G G G A A G A T A C T G G A C A A
TGTGGGAAGCTCCTATGTTTGGTAGCACTGATNCTTNTNAAGNCTTTNANGGACCTGGN
T N A N A N N T N G N T G C C N F rT T A N C C C C C A A C C T T N N N N N C C G T T C A N N N S '
C lo n e 1 0 3 -K l R e v e rs e
p r i m e r s . (737bp)
Sequence
A m p lifie d
u sin g
RbcS
s p e c ific
5 ' TTACAATCTACTATCATNCCACTTGACATCGTTGTCGAAACCGATGATACGGACGAA
AGCTTGNGGGTAATGCGNCAANAACTTCATNAAGCTCCTTNAACACTTGNTAAGCATNA
GGGGNTNCCAAACATAGGAAGCTTGGCACATTGACCAGTATCTTCCATCATATTATCCT
g g tg a cttg ttg tn ctca cg g t
AcAc a
aatcctttctgcaaccaaacagtcaaaatcaa
CTCATTACGGTTTCAAACAAAACTATTTATATACTCATTCNTTM NCNNAACCATCTCTN
NNTTTCCNTTNCTACTCCTNCNCCCCCNTTCCACNNCATNCTNCTTNTCCM TCANCNCC.
TCCTNNNTCCNCCCCTTCTTCCCCANCCCCCNTTCTNCNCCNTCNCCNCNANTACTCTN
CNNTCTCNTCTCTCTCTACCTTCTNNNTTTCNCCACNCTCCTCCCCCTCTNNCTNTCCC
NTNTM TCCTCCTCNCTTNTCCNCNNNANCCCNNTTCNTTNNCCNNCTNTNCCTCCNCCC
TCCNACCNTNTTCNATTANTCNCTTCTNNCCCNCNCCCCNNCNATTCNNNCTCTTTNTN
CCCCNCTCCNTNCM TTNCCTCNNNCCCCTNANCNNCNCCCNTCTTCNTCCNCTCCCTCN
'N N C C C T N C C C C C C T T A T T C N C T A T T C C C T C A N T T A C C N C C C C A C T T N C N N C T C T N T C A T
CC A C C A C G N W C N N TN N TC N C TTC TC C G 5'
139
APPENDIX B
RAW MARKER SEGREGATION FROM CROSS LC #7 x EE# 32
140
Raw
M ark er
.L.
S e g re g a tio n
cu lin a ris
L .cu lin a ri.s= A
A atc
A atp
Apx
Apy
pA m yl
pfruct
C lpP or
Dim in
Enol
Gpi
E stl
E st2
Gs
H isl
K9
Lapl
Lap2
Lb
L ectin
L ip(C opl)
Mnsod
Pl
P393
P628a
P628b
Pepl ■
Pgdp
Pgmp
PhyApo
P in l
PsPk2
Pxl
RbcS
RDNA
Rpl22
Sahh
's k d h
TubAl
Yc
O P A 2 /5 5 0
O P A 2 /7 4 0
O P A 2 /8 0 0
O P A 7 /3 0 0
O P A 9 /2 9 0
x.L.
D a ta
fo r
C ro ss
erv o id e s
L . erivoides= B
BB BB A B BBA B BA A BBB BB A B BBB BA A B- AABABAAAABBBBAA- -A A -A A
AAB BAB B BA BA A -BA A A B A BBB A A A A A A B BBA B A B A BBA B BA B-B ----- B B A B B B A B B B A A B B A A B B B A A B B A -B B A A A -B B A A A -A -B B ---------------------AAAAAAAB A A A -AAAAAAAAAAAABAAAABAAB AAAAAB AB AB A B B B - —
BAABAABBBBABBBABBAAABBAABBAAABBBAAAAAABABAAABABAAB BAAB BAB BAABAB BABBABAABABAAAAABAAAAAAAAB B BAAAB BAAA
• AAAABAABBAAAAAAAAAABAABABAAAAAAAAAAAAAAABB -A A A -A A A
B A - A B B B B B - B B B - B B B B A - - B - B - -B A A A B B -B A A A -A A - A A - B B B - - B
A A A A A BB A A B BA A B BB BA BA A A A B BA A A A -A A -A -A A A A A ---------------------A A A A B -A B B A B AAAAAABA - AABABAAABBAA- A A A B - BABBAABAAAAAABBBBABBBBBBBBABBAABBBBAAAABBABAABAABBABBAABABAAB
B A -B BAABBBAAABAAA -A B B B - AAAAAB BBBBABAABBABABABAB - BA
B A B B B A BB BB A A A BA A A A A B BBB A A A A A BBB BB A B A B BB BBA B B-BB-BB
AAAAABAAAAAABAAB -A B A A A A - B -A A B A A B - AAAAAB AAAB AB A -B B B
BAAABBABAAABBBABABBAAAAABBAAABBBAAAAA-- - - - A - A B - A - B
AAAAAAB B - BAAAAAAAABA A A - A A A B - AAABAAAABABABAABAAA-B A
A BA A A B A B- - B B B B B A B - - B A - B
A A ------------------------ '
--------------■----------------------------------------------- -r - A A - - A B --------- B - ------------- A ------ B - - A AAB B AAB B AAAA - AAAAAAAB AB AB AB A B -A B A B B B B B B ---------------------- -B A - - B A - BBAAAAAAAAAABBB-AAAAAB- B B B A B A -B B - - A B - B - B - B a b a b a a b b a a a a a a a a b a a a a a a a a a a a a b a b a b Ab b b b b b a b b a a b b a a
BBAABBABBAABBBA BBBBBAAAABBAAABBBAAAAA- :-------A - B B - B - B
a a Bb a a b a a a a a b a a a b a a a b b b b b a b b b a a a b b b b a b b b b b b b a - a b b a
A A B - BB BA A BA A BA A ABAA A A BBBBA BA B-A BBBBBA BBBBA BBA B A - BA
B A B - BAABBAAAABBAAAABA BBAA A BA B- B B B B B B B B B A B A A A B B B -B B
A - A A B B -A A B A B -B -B --B B A B -A B --A B A B --A B --A B B --B -A -A -A A
A A A A A B B A B B B B B B A B B B A B B B B A B A B A B A B B B B B B B A B A --B A A ----- AA
- A - B B A - B - B A A - - B A A - ------B - A A A - A ---------B B B - B B - - A - - B B B -------BABABAAAB BAAAAAAAAAB B B BAAABAAB BAAABAAB B B BAAABA B B - AAAABAAAABAAAAAAAAA- A A BA A A A A BA A A -A A -A -A A B BA A B A A A A A
AAAAAAAAAAAAAAAAAAA- A A - AAAAAAAAA- A A A A - AABBAAAAAAAA
AAAABAABAAAAAAAAA- AAAABAA- - A - - B A - A ----------------------------- -----AAAAB AABB AAAAAAAAB AB AAB AB AAAB B AAAAAAAAAB B BAABAA - AA
BAAABABBBBAAABAAABA-AAAAAAAABBBB - A B - A - B AB AAAB AAAAA -A A -A A .- -A A A A A A -A A A B A A BABAAAA- - A A - A A - - A B - BAAAABAAA
AAAAAAB B BAAAA
A A A -B B A A -A A ------- B A A A A B -B A B --------------------A A B B B B B B B B B B B B B A B B A B B B B B A -B B B A A B A -B -A A -B B B A A B A A -A B
BB BB BBB BBB BA A BB A B A A B BBB BA A B- B - B - B A A A A B B B ---------------------ABAAAB BAAAAABAB BBAB BAAABAB B BAAAAABABAAABAAB BAB BBBB
BAB BAAB B BBAB BBAB BA A - B B A A B B A A A B B B -A A A A -B B B AAABA BA A BABABBA BBBA A BBBBBAA - BBBB BA A A A BB B- A B A A - AABAAABB B A B BABABAAB BB B AAB BAABA - B B B B A A B B A A B A -A A A A -B B B A A B A A A A A BAAAAAABBBAAABAAABA- AAAAAAAABB B B -A B A A -B A B AAABAAAAA BBAB B BAAAAABAAABA A - AABBBA BA BA BA - B A B A -A B B A B A A B A B B -
141
OPAl O/9.5 O
OPB1/350
OPB1/500
OPBl/650
OPBI/850
OPB7/550
OPB8/900
OPB8/1000
OPB17/900 .
OPB17/78.0
OPB18/HOO
OPB18/1500
OPD5/950
OPD5/400
OPD5/HOO
OPD7/1150
OPD7/750
OPD7/550
OD16/1800
OPD16/950
OPD16/800
AAAABAAABABAA- AAAAA- AABBBAAAAAAA7AAAA- AABBAAAABAAAAAAB AAAAAAAAAAAAAA - AAB AAAAABAAA - AAAA - AAB AAAB AAAA AAAAB AB AAAAAAAAAAAA - AABAAAAAB AAA - AAAA - AAB BAABAAAA ABBABBBAAAAABAAABAA-AABBBABABABB- BABA-ABBABBAAAAAAABABBABBBBBBBBABBA-BBBBAAABBBAB- BBAA- BABBAABABABBAABAABBBBABBB- BBAA- BBAABBAAABBB-AAAA- BAB AAABABAABAB BAAB BBBABB BAB BAA- BBAABBAAABBB-AAAA- AABAAABABAABAB BAAB BABABB BAB BAA - BAAAABAAABBA-AAAA-AABAAABAAAA ABBABB BAAAAABAAABAA - AABBBABAB - BB - BABA - ABBABBAAAAA AAAAABBAAAAABAAABAA- AAAABAAAABAB- AAAA- ABBAAABBBBBABBBBB BAAAAABAAABAA- ABBBBABABABB- - A BA - ABBABBAAA-BAAAABAABAAAAAAAAAAA- AABABAAABBAA- - AAA-AABBAABAA-AA -A A -B -A A -A B A B A A A A B r AABABABAAAAB-AAAB- BABAABAABBBB - B B - B - B B -A B A A A B B A A -B B A A B B A A A B B B -A A A A -B B B A A A B A B A A A - B A - B - A A - A A BA AABAA-ABBBBABABA B B -B B B A -A B B A A B A B A A A BAAABAAABBAAAAAAAAA-BABAAAAAAAAA-AAAA-AABAAAAAAAAa a a a b a a a a a a a a a a a a a a -a a b a a a a a b a a a -a a a a -a a b a a a a a a a a -
AAAABBBBBBAAABAAAAB - AABAAAABAAAB -A A A B - BAABBBABBBB B AAAB AAAAAAAAB B AAAA - AAB AAAAAAB B B - AAAA - AB AAAAB B BAA BAAABABBBBA A A BA A ABA -A A BA A AA A BBBB-A BA B - BBBABABBBBB A B B A B BBA A A A A A A A A B A A -A A B BB A B A BA A A -BA BA -A BBA A BA BA B B-
Lc #7 x Le #32 Progeny. *Order same as above
2,3,5,7,9, 10,11,12,13,14,15,17,18,19,20,21,22,23,24,25,26,27,
33,34,35,3 6,3 7,38,41,42,44,45,46,47,48,49 ^51,5.2,53,54,55, 56,57,
58,62,63,64,65,66
MONTANA STATE UNIVERSITY - BOZEMAN