5-36 volume drop to specified values. The heat transfer and the... There are no work interactions involved other than the boundary...
advertisement

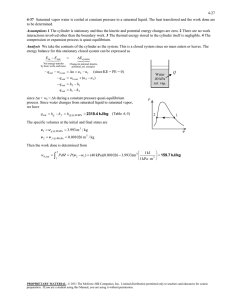
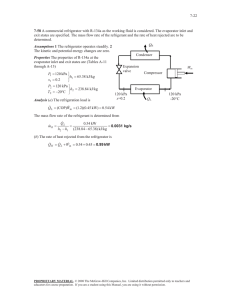
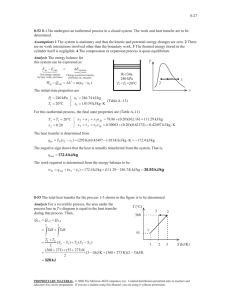
5-36 5-46 R-134a contained in a spring-loaded piston-cylinder device is cooled until the temperature and volume drop to specified values. The heat transfer and the work done are to be determined. Assumptions 1 The cylinder is stationary and thus the kinetic and potential energy changes are zero. 2 There are no work interactions involved other than the boundary work. 3 The thermal energy stored in the cylinder itself is negligible. 4 The compression or expansion process is quasi-equilibrium. Analysis We take the contents of the cylinder as the system. This is a closed system since no mass enters or leaves. The energy balance for this stationary closed system can be expressed as E E in out 'E system Net energy transfer by heat, work, and mass Change in internal, kinetic, potential, etc. energies Wb,in Qout Qout 'U m(u 2 u1 ) (since KE = PE = 0) Wb,in m(u 2 u1 ) The initial state properties are (Table A-13) P1 T1 600 kPa ½ v 1 ¾ 15qC ¿ u1 P 3 0.055522 m / kg 357.96 kJ/kg The mass of refrigerant is m V1 v1 1 600 kPa 2 0.3 m 3 0.055522 m 3 /kg 5.4033 kg v The final specific volume is v2 V2 m 0.1 m 3 5.4033 kg 0.018507 m 3 /kg The final state at this specific volume and at -30qC is a saturated mixture. The properties at this state are (Table A-11) x2 v 2 v f 0.018507 0.0007203 0.22580 0.0007203 v g v f u2 u f x 2 u fg P2 84.43 kPa 0.079024 12.59 (0.079024)(200.52) 28.44 kJ/kg Since this is a linear process, the work done is equal to the area under the process line 1-2: Wb,in Area P1 P2 (V 1 V 2 ) 2 (600 84.43)kPa § 1 kJ · (0.3 0.1)m 3 ¨ ¸ 2 © 1 kPa m 3 ¹ 68.44 kJ Substituting into energy balance equation gives Qout Wb,in m(u 2 u1 ) 68.44 kJ (5.4033 kg)(28.44 357.96) kJ/kg 1849 kJ PROPRIETARY MATERIAL. © 2008 The McGraw-Hill Companies, Inc. Limited distribution permitted only to teachers and educators for course preparation. If you are a student using this Manual, you are using it without permission.