37
advertisement

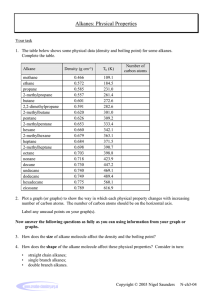
The Proton Transfer Reaction-Mass Spectrometer for Atmospheric Chemistry Tracers of Diesel Exhaust Emissions and Measurements of Trace gas and Aerosol properties. Debra Garvey, Matt Erickson and Tom Jobson Laboratory for Atmospheric Research Department of Civil and Environmental Engineering Objectives Introduction Diesel exhaust experiment It has previously been difficult to identify the emissions of diesel exhaust until the use of a technique referred to as the Dynamic Dilution System. The use of a Proton Transfer ReactionMass Spectrometer(PTR-MS) was vital during the experiment because the reagent ion, H3O+ , is highly reactive with organic compounds such as alkanes which are present in diesel exhaust emissions. The reaction which takes place is that the organic compound, R, would be protonated by the reagent ion and a mass reading plus one would be the result and it was expected that the new compound would dissociate into masses differing by fourteen which would represent a CH2. The alkanes used during the experiment ranged from lengths of six through seventeen carbons. Also Nitrogen gas was a necessity because the alkanes would react quickly with air, hence their ability to cause air pollution. The main purpose of the experiment was to determine the reaction time and to make sure that the sensitivity of the technique is high enough to detect different compounds in a complex mixture such as diesel exhaust. The reaction rate is the speed at which molecules collide and thus react with each other. For this technique the reaction time must be fast enough so that molecules react proving that the procedure is reliable. A high reaction rate displays the sensitivity of the instrument which is vital in the research of diesel exhaust emissions. During CABINEX, it is expected that with the emission of isoprene there would be a similar pattern with the production of formaldehyde and other air pollutants. To successfully detect and quantify the biogenic emissions, and the products which are formed, throughout a time period. Experiment Apparatus Liquid N2 Tank Cole Parmer Hamilton Rotameter (Liters per minute) Copper Pipeline Insulation: Glass wool and foil Clamps Heat suppliers: Heating tapes and ropes and variable autotransformers Septa Hamilton Syringe (50 uL) Harvard Apparatus PHP 2000 Infusion Syringe pump (20 uL/hr) Thermometers (Centigrade) Ionicon Analytik Proton Transfer Reaction-Mass Spectrometer (PTR-MS) Rotameter (LPM) PTR-MS MACL Measurement of Trace Gas and Aerosol Properties Trees are known to release biogenic compounds and among them are isoprene, C5H8. Isoprene is released from trees during photosynthesis with high intensities of sunlight. The isoprene in turn reacts with the hydroxyl radicals, OH, present in the atmosphere. From this reaction a host of compounds which cause air pollution, such as formaldehyde, are produced. The purpose of the PTR-MS is to detect the increase and decrease of isoprene over time with the difference of sunlight intensity. It is expected that the measurements of formaldehyde should follow the signal reading of isoprene just not as intense. This experiment was conducted at the University of Michigan Biological Station during CABINEX using the Mobile Atmospheric Chemistry Laboratory(MACL). The alkanes have this appearance due to their PA > PA of H2O and so they are easily detected. Hence why as their size increases so do their sum of ion signals. Below is the comparison of the PTR-MS with that of the electron ionization mass spectrum of Tetradecane. Insulated Pipeline N2 Gas CABINEX Tower and trailer inlet. Results and Discussion In this figure the PTR-MS detected monoterpenes at the expected masses of 137 and 81 amu. Syringe Pump Constant heat and insulation Procedure The N2 gas was supplied through the apparatus at a constant flow of 25 LPM and the alkane was injected through the system at 20 uL/hr. The alkanes were kept at a temperature of approximately 90 C or 30 C, for it was dependent upon their polarity and the required heat supply for sufficient signals. The pipeline required constant heat to prevent condensation which would increase dark counts on the PTR-MS signal readings. Each cycle was 120 seconds and the counts per second (cps) were recorded for each interest peak. After a number of cycles with stable readings, the next alkane was injected into the system. The long chain alkanes were injected at a mixing ratio of approximately 2ppmv while the smaller alkanes were injected at a higher concentration of ~3ppmv. Clearly the PTR-MS data demonstrates the production of VOCs from the reaction of biogenic emissions with the atmosphere during moments of intense sunlight. Conclusion: The sensitivity of the PTR-MS to the alkanes is efficient to detect the alkanes with a fast reaction time which is greatly important for gas phase emissions due to their ability to quickly react with the atmosphere. The VOCs detected by the PTR-MS also included acetaldehyde , MEK, PAN and multiple species to name a few. Acknowledgements: NSF REU program grant number 0754990, University Of Michigan Biological Station.