Movement of Replicating DNA through a Stationary Replisome Please share
advertisement

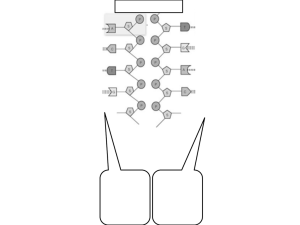
Movement of Replicating DNA through a Stationary Replisome The MIT Faculty has made this article openly available. Please share how this access benefits you. Your story matters. Citation Lemon, Katherine P, and Alan D Grossman. “Movement of Replicating DNA through a Stationary Replisome.” Molecular Cell 6, no. 6 (December 2000): 1321-1330. Copyright © 2000 Cell Press As Published http://dx.doi.org/10.1016/S1097-2765(00)00130-1 Publisher Elsevier Version Final published version Accessed Wed May 25 19:02:26 EDT 2016 Citable Link http://hdl.handle.net/1721.1/83846 Terms of Use Article is made available in accordance with the publisher's policy and may be subject to US copyright law. Please refer to the publisher's site for terms of use. Detailed Terms Molecular Cell, Vol. 6, 1321–1330, December, 2000, Copyright 2000 by Cell Press Movement of Replicating DNA through a Stationary Replisome Katherine P. Lemon and Alan D. Grossman* Department of Biology Massachusetts Institute of Technology Cambridge, Massachusetts 02139 Summary We found that DNA is replicated at a central stationary polymerase, and each replicated region moves away from the replisome. In Bacillus subtilis, DNA polymerase is predominantly located at or near midcell. When replication was blocked in a specific chromosomal region, that region was centrally located with DNA polymerase. Upon release of the block, each copy of the duplicated region was located toward opposite cell poles, away from the central replisome. In a roughly synchronous population of cells, a region of chromosome between origin and terminus moved to the replisome prior to duplication. Thus, the polymerase at the replication forks is stationary, and the template is pulled in and released outward during duplication. We propose that B. subtilis, and probably many bacteria, harness energy released during nucleotide condensation by a stationary replisome to facilitate chromosome partitioning. Introduction The production of viable progeny depends on the faithful replication and partitioning of genomic DNA. Bacteria do not appear to have a eukaryotic-like mitotic apparatus. Like many bacteria, B. subtilis has a single circular chromosome (4200 kb; Kunst et al., 1997) with a single origin (oriC) from which bidirectional DNA replication initiates (Kornberg and Baker, 1992). Separation of newly duplicated chromosome regions occurs while DNA replication of the rest of the chromosome proceeds (Webb et al., 1997; Teleman et al., 1998; Webb et al., 1998). Recent work with B. subtilis, E. coli, and C. crescentus demonstrates that the chromosome is oriented such that for most of a cell cycle the two sister origins reside on opposite sides of the cell, while the unduplicated terminus region is centrally located (Glaser et al., 1997; Gordon et al., 1997; Lewis and Errington, 1997; Lin et al., 1997; Mohl and Gober, 1997; Webb et al., 1997; Niki and Hiraga, 1998; Sharpe and Errington, 1998; Teleman et al., 1998; Webb et al., 1998; Jensen and Shapiro, 1999; Niki et al., 2000). Regions located between the origin and terminus on the circular chromosome appear to be positioned between them in the cell (Teleman et al., 1998; Niki et al., 2000). Efforts to associate chromosome movement with the act of DNA replication are confounded by two factors. First, the rate of bacterial DNA polymerization is estimated at 500–1000 nucleotides per * To whom correspondence should be addressed (e-mail: adg@ mit.edu). second (Baker and Bell, 1998). This is equivalent to moving of a single base pair a distance of ⵑ170–340 nanometers each second. Second, DNA inside cells is highly compacted (reviewed in Trun and Marko, 1998). The ⵑ4200 kilobase pair (kbp) circular genome of B. subtilis (Kunst et al., 1997) is neatly organized in a rod-shaped cell two to four microns in length and about half a micron in diameter. Here, we describe experiments that demonstrate that DNA is duplicated at a centrally located, stationary replisome. Previously, we visualized the B. subtilis replicative DNA polymerase using a functional fusion of the catalytic subunit, PolC, to green fluorescent protein (GFP) (Lemon and Grossman, 1998). PolC–GFP localizes to discrete positions predominantly at or near midcell or sites that will become midcell, indicating that the replisome is stationary and centrally located. We reasoned that if DNA replication is reversibly blocked at a specific chromosomal region, then that region should be centrally located and coincident with the DNA polymerase. When the replication block is relieved, that site should be duplicated and no longer coincident with the stationary replisome. A reversible DNA replication arrest occurs in B. subtilis during the stringent response (Levine et al., 1991, 1995; Autret et al., 1999) induced by amino acid starvation. Replication arrests in regions ⵑ100–130 kbp to the left (LSTer or left stringent terminus region) and ⵑ150–200 kbp to the right (RSTer) of oriC (Levine et al., 1991, 1995; Autret et al., 1999). DNA polymerase resumes chromosome synthesis in the STer regions when amino acids are added back to the cells. Starvation for an amino acid activates the production of the alarmone (p)ppGpp by the relA gene product, which induces the pleiotropic stringent response in many bacteria (Cashel et al., 1996; Wendrich and Marahiel, 1997), though stringent arrest of DNA replication at sites away from the origin has only been characterized in B. subtilis. We visualized the more precisely mapped LSTer region (Autret et al., 1999). During stringent arrest, the LSTer region was located at or near midcell. Under these conditions, the newly duplicated sister origins are already separated to opposite sides of the cell (Sharpe and Errington, 1998). After release from replication arrest, the LSTer region was duplicated, and the two copies were positioned away from midcell in opposite halves of the cell. We also visualized a region of the chromosome (270⬚) approximately midway between origin and terminus. Just prior to a burst of duplication of the 270⬚ region in a roughly synchronous population of cells, we observed an increase in the occurrence of coincidence of the 270⬚ region and DNA polymerase. Taken together, our results indicate that the stationary, centrally positioned DNA polymerase does indeed represent the location of active replication forks and suggest that the DNA is threaded through the polymerase during duplication and then extruded from the polymerase after duplication. Molecular Cell 1322 Table 1. Number of Foci of the LSTer Region per Cell before, during, and after Stringent Arrest Table 2. Number of Foci of the LSTer Region in Cells during Growth after Synchronization Percentage of Cells with Indicated Number of Focib Conditionsa (Number of Cells) Asynchronous growth (728) Stringent arrest (572) Release from arrest (504) 1 2 42 77 28 56 22 64 a Strain KPL549 [dnaB134(ts), hutM⫹-lacO, lacI-gfp] was grown in defined minimal medium at 30⬚C, and the number of foci of the LSTer region per cell was determined. Samples were taken (1) during asynchronous exponential growth, (2) 30 min after induction of stringent arrest in a synchronized population, and (3) 30 or 35 min after release from stringent arrest. b During asynchronous growth and stringent arrest, ⬍2.5% of cells had three or four foci. After release from arrest, 6.9% of cells had three foci and 1.4% had four foci. Percentage of Cells with Indicated Number of Focib Timea Number of Cells Asynchronous 0 15 30 45 60 75 856 406 442 348 449 394 566 1 2 45.2 62.3 64.3 52 39.7 31.2 27.2 51.5 37.4 35.7 46.8 58.1 65.5 69.3 a The replication cycle was synchronized by shifting cells to nonpermissive temperature (45⬚C) to block initiation of replication and to allow most ongoing rounds to finish. Samples of strain KPL549 [dnaB134(ts), hutM⫹-lacO, lacI-gfp] were taken at the indicated times (min) after shift back to permissive temperature, and the number of foci of the LSTer region per cell was determined. b At all times, less than 3.5% of cells had three or four foci. Results Visualization of the LSTer Region We chose the more precisely defined LSTer region over the RSTer region for analysis (Autret et al., 1999). The LSTer region contains two approximately equivalent arrest sites, LSTer1 and LSTer2, separated by about 27 kbp (Autret et al., 1999). Stalling of DNA polymerase in both the left and right STer regions is about 80% effective in a synchronized population of cells treated with arginine hydroxymate (Levine et al., 1991, 1995; Autret et al., 1999). We marked the LSTer region by inserting a tandem array of E. coli lac operator sites adjacent to hutM on the B. subtilis chromosome, which is ⵑ40 kbp from LSTer2 and ⵑ67 kbp from LSTer1. We chose this position for convenience and to minimize the possibility that the array would disrupt function of the LSTer region. The hutM-lacO array was visualized with a carboxyterminal fusion of Lac repressor to a variant of GFP (LacI-GFP) (Cormack et al., 1996). During exponential growth, most cells have a partly duplicated chromosome. The LSTer region is within 130 kbp of the origin and is duplicated relatively early (Autret et al., 1999). Indeed, during asynchronous exponential growth, most cells had two well-separate foci of the LSTer region (Table 1). The pattern of localization of the sister LSTer regions appeared to be similar to that of the origin region (Webb et al., 1997; Teleman et al., 1998; Webb et al., 1998) and the chromosome partitioning protein Spo0J (Glaser et al., 1997; Lewis and Errington, 1997; Lin et al., 1997; Sharpe and Errington, 1998; Teleman et al., 1998), which binds to sites (parS) in the originproximal 20% of the chromosome (Lin and Grossman, 1998). Synchronization of DNA Replication in a Population of B. subtilis In order for most of the cells to undergo replication arrest at a similar time, the population was first synchronized for DNA replication (Levine et al., 1991, 1995; Autret et al., 1999). A mutant that is temperature sensitive for initiation of DNA replication, dnaB134 (Mendelson and Gross, 1967; Karamata and Gross, 1970), was shifted to nonpermissive temperature to allow most ongoing rounds of replication to finish while preventing initiation of new rounds of replication. In B. subtilis, the replication initiation protein DnaB (not a helicase as is E. coli DnaB) is membrane associated and is required for origin enrichment in membrane fractions (Winston and Sueoka, 1980). Immediately after shiftdown to permissive temperature, ⵑ62% of the population was synchronized as judged by having a single focus of the LSTer chromosome region (Table 2), indicating a single chromosome per cell. This method gives at least one roughly synchronous round of replication in a population (Dworkin et al., 1972). The LSTer Region Is Centrally Located during Stringent Response–Induced Replication Arrest Stringent response was induced in a dnaB134(ts)-synchronized population of cells by addition of the amino acid analog arginine hydroxymate (250–500 g/ml) (Levine et al., 1991, 1995; Autret et al., 1999). Thirty minutes after induction of stringent DNA replication arrest, 77% of cells had a single, unduplicated focus of the LSTer region (Table 1). In contrast, in the absence of stringent arrest, the percent of cells (52%) (Table 2) with a single focus 30 min after synchronization was similar to that in a culture of asynchronously growing cells (45%) (Table 2). During stringent arrest, the single focus of LSTer was located at or near midcell (Figure 1A). DNA Polymerase Is Centrally Located during Stringent Response–Induced Replication Arrest To be sure that the manipulations to synchronize and induce stringent arrest did not affect DNA polymerase position, we observed DNA polymerase in live cells using a functional fusion of the Tau subunit to GFP. A dimer of Tau is the scaffolding that holds the leading and lagging strand polymerases together (Kelman and O’Donnell, 1995; Baker and Bell, 1998). The Tau-GFP fusion produced a slightly stronger signal than the PolCGFP fusion, and previous work indicated that during exponential growth, the position of Tau-GFP is similar Movement of DNA through a Stationary Replisome 1323 Figure 1. Position of the LSTer Region and DNA Polymerase during Replication Arrest Induced by the Stringent Response During stringent arrest, most cells have a single centrally located focus of the LSTer region. After release, most cells have two wellseparated foci of the LSTer region. (A) shows the position of the LSTer region (hutM-lacO) in cells with a single focus during stringent arrest (341 cells). In (B), the position of DNA polymerase in cells with a single focus of Tau-GFP during stringent arrest (402 cells) is shown. (C) shows the position of the LSTer region (hutM-lacO) in cells with two foci after release from stringent arrest by nutrient addback (114 cells). Position was determined by measuring the distance from midfocus to the cell pole. In cells with two foci, distance was measured from the same cell pole. (A) and (C), strain KPL549 [dnaB134(ts), hutM⫹-lacO, lacI-gfp]; (B), strain KPL502 [dnaB134(ts), dnaX-gfp]. to that of catalytic subunit fusion, PolC-GFP (Lemon and Grossman, 1998). In cells with a single focus of Tau-GFP during stringent arrest, the focus was located predominantly at or near midcell (Figure 1B), similar to the LSTer region position during stringent-induced DNA replication arrest (compare Figures 1A and 1B). After Release from Replication Arrest, Most Cells Had Two Well-Separated Foci of the LSTer Region DNA replication arrest was released by washing cells (to remove arginine hydroxymate) and resuspending them in a nutrient-rich medium. Within 30–35 min after release, most cells (64%) had two foci of the LSTer region (Table 1). Thus, a majority (67%) of the cells with a single focus during arrest appeared to replicate the LSTer region after release. Duplication of the LSTer region correlated with a resumption in growth as measured by an increase in the culture’s optical density. The two newly duplicated copies of the LSTer regions were well separated from each other and located at ⵑ35% and ⵑ65% of the cell length (Figure 1C). In addition to the shift from one to two foci, after release there was an increase in the proportion of cells with three and four foci (from 1% to 8%). Visualization of DNA Polymerase and LSTer in the Same Cells The experiments described above were done in live cells from two different strains; one contained the LacI-GFP fusion and the lac operator array near the LSTer region, and the other contained a functional Tau-GFP fusion. When DNA replication was stalled in the LSTer region, the majority of cells had a single focus of both the LSTer region and DNA polymerase at or near midcell (Table 1; Figures 1A and 1B). To directly show that the LSTer region and the polymerase colocalize during replication arrest, we visualized the LSTer region and the replisome in the same cells with and without the stringent arrest. The development of variants of GFP with sufficiently separable excitation and emission spectra permits the observation of two separate fusion proteins in the same cell (Tsien, 1998; Miller et al., 1999). DNA polymerase was visualized using a functional fusion of Tau to a yellow variant of GFP (YFP) (Ormo et al., 1996). The lacO array near the LSTer region (hutM-lacO) was visualized using a fusion of LacI to a cyan variant of GFP (CFP) (Heim and Tsien, 1996). During asynchronous growth, the majority of cells (83%) had two foci of the LSTer region (Table 3, line A). Immediately after synchronization of the replication cycle, ⵑ43% of the cells had two foci of the LSTer region and ⵑ55% had a single focus (Table 3, line B). One hour later after growth at permissive temperature, most of the cells (83%) had two foci of the LSTer region (Table 3, line C). In marked contrast, when stringent arrest was induced, only ⵑ47% of the cells had two foci and 49% had a single focus of the LSTer region (Table 3, line D). When stringent arrest was released, cells began to grow, and by thirty minutes a majority had duplicated and separated the LSTer region (Table 3, line E). The increase in cells with three and four foci after release (Table 3) indicates the occurrence of multifork replication under these growth conditions (see below). A clear pattern emerged when we observed cells that had a centrally located polymerase (either a single focus or two closely juxtaposed foci [Lemon and Grossman, 1998]) and one or two foci of LSTer. During exponential growth in an asynchronous culture, 86% of the cells had two well-separated foci of the LSTer region, one on either side of the central DNA polymerase (Figure 2). This same pattern was observed in the majority of cells (79%) that were growing without arrest 60 min after synchronization (Figures 2, 3A–3C, and 4). In contrast, when DNA replication arrest was induced (stringent response), there was a marked increase in the proportion of cells in which a single focus of LacI-CFP bound near the LSTer was either coincident with (31%) or touching (18.5%) a focus of DNA polymerase (Tau-YFP) (Figures 2, 3D–3F, and 4). Likewise, there were many fewer cells, only ⵑ36%, with the typical exponential growth pattern (Figures 2 and 4). The lacO array is inserted ⵑ67 kbp from the first arrest site (LSTer1) and ⵑ40 kbp from the second site (LSTer2). LSTer1 and 2 are approximately equivalent (Autret et al., 1999), so in about half the cells the DNA replication fork is blocked ⵑ67 kbp away from the lacO array that we visualized. It is likely that in cells in which the LacI-CFP focus touches the Tau-YFP focus, LSTer is coincident with the replication fork. Therefore, we have combined these two classes in Figures 2 and 4. When cells were released from stringent arrest, within 30 min the pattern shifted back toward that seen for cells growing without arrest. Approximately 53% of cells had two well-separated LSTer foci flanking a central Molecular Cell 1324 Table 3. Number of Foci of the LSTer Region in Cells Containing Tau-YFP Percentage of Cells with Indicated Number of Foci a Conditions (Number of Cells Counted) (A) Asynchronous (623) (B) Synchronous, 0 min (706) (C) Synchronous, no arrest (600) (D) Synchronous, stringent arrest (457) (E) Synchronous, release from arrest (696) 1 2 5.6 54.8 12.7 49 20 83.3 42.5 83 47.3 66.4 3 4 4.7 2 2.7 2.8 9 6.4 0.7 1.7 0.9 4.6 a Samples of strain KPL730 [dnaB134(ts), hutM⫹-lacO, lacl-cfp, dnaX-yfp] were taken under the indicated conditions, and the number of foci of the LSTer region per cell was determined. (A), asynchronous growth; (B), synchronous cells immediately after shift to permissive temperature after 45 min at 42⬚C (to prevent initiation of new rounds of replication and to allow most ongoing rounds to finish); (C), synchronous cells 60 min after shift to permissive temperature with no stringent arrest; (D), synchronous cells 60 min after shift to permissive temperature and during stringent arrest; and (E), synchronous cells 30 min after addition of nutrients to release stringent arrest. Nutrients were added 60 min after stringent arrest and shift to permissive temperature. DNA polymerase (Figures 2, 3G–3I, and 4). The pattern of localization of DNA polymerase and the LSTer region described here strongly supports the model that the DNA template is duplicated by a stationary, centrally located DNA polymerase. After duplication, the two sister chromosome regions then move in opposite directions apart from each other and away from the replisome. LSTer out toward each pole, both of which were either adjacent to or coincident with a focus of DNA polymerase (Figures 3J–3O). In these cells, DNA replication has probably reinitiated or is about to initiate at the two sister origins prior to cell division. We speculate that initiation occurs at the cell quarters, which are nascent Multifork Replication B. subtilis, like many other bacteria, is capable of doubling, under optimal growth conditions, in about a third of the time it takes to duplicate the genome. This is accomplished by reinitiating DNA replication from the sister origins before cell division so that there are multiple bidirectional replication forks per cell, essentially giving the replication cycle a headstart on the division cycle. When cells were released from stringent arrest by resuspension in a nutrient-rich medium, multifork replication began to occur in about 20% of cells, as indicated by cells with more than two LSTer foci (Table 3). After release, we observed cells that had a focus of Figure 3. Visualization of DNA Polymerase and the LSTer Region in the Same Cells Figure 2. Relative Positions of the LSTer Region and DNA Polymerase in the Same Cells Samples of strain KPL730 [dnaB134(ts), hutM⫹-lacO, lacI-cfp, dnaXyfp] were taken under the indicated conditions, and the relative positions of the LSTer region (hutM-lacO; gray circles) and DNA polymerase (triangles) were determined by fluorescence microscopy. Data are shown for cells with a central focus (or two closely juxtaposed central foci) of Tau-YFP and one or two foci of the LSTer region. The number of cells counted for each condition is indicated in parenthesis. The coincident class includes cells in which the LSTer and polymerase foci were coincident and those in which the foci were touching (see Results). Samples of strain KPL730 [dnaB134(ts), hutM⫹-lacO, lacI-cfp, dnaXyfp] were taken for microscopy various times after synchronization of the replication cycle. (A–C) Sixty minutes after synchronization. (D–F) Sixty minutes after synchronization and induction of the stringent response (replication arrest at LSTer). LSTer is coincident with the replisome. (G–I) Thirty minutes after release from stringent response, two copies of LSTer away from the centrally located replisome. (J–L) A cell undergoing (or about to undergo) multifork replication 30 min after release from stringent replication arrest. The central polymerase is from the initial round of replication, and additional replisomes have assembled at approximately the cell quarter positions. (M–O) Replisomes approximately at the cell quarters in a cell that seems to have finished the initial round (no replisome at midcell). Left panels (A, D, G, J, and M) show foci of DNA polymerase (TauYFP; orange) and cell outline (FM 4-64; gray). Middle panels (B, E, H, K, and N) show foci of LSTer (LacI-CFP; cyan) and cell outline (gray). Right panels (C, F, I, L, and O) show merged images of the left and middle panels. Scale bar is 1 m. Movement of DNA through a Stationary Replisome 1325 Figure 4. Analysis of the LSTer Region and DNA Polymerase Samples of strain KPL730 [dnaB134(ts), hutM⫹-lacO, lacI-cfp, dnaXyfp] were taken 60 min after synchronization of the replication cycle (no arrest), 60 min after synchronization and induction of the stringent response (arrest), and 30 min after release from the stringent response (release). Dark bars indicate the percent of cells with a single LSTer region that was coincident with a central DNA polymerase (as in Figure 3F). Light bars indicate the percent of cells with two well-separated foci of the LSTer region and a centrally located DNA polymerase (as in Figure 3I). Error bars indicate the 95% confidence interval. Figure 5. Analysis of the 270⬚ Chromosomal Region and DNA Polymerase midcell positions. Some of these cells also had a centrally located DNA polymerase that probably represents an initial round of replication that was not yet complete (Figures 3J–3L). Visualization of DNA Polymerase and the 270ⴗ Region of the Chromosome in Synchronized Cells It appears that in B. subtilis, the origin region is located at approximately midcell or a position that will become midcell, prior to duplication (Lewis and Errington, 1997; Teleman et al., 1998) (D. C.-H. Lin, P. S. Lee, and A. D. G., unpublished data). The LSTer region is close to oriC, less than 150 kbp away, whereas the distance from oriC to the terminus is about 2100 kbp. Thus, we could demonstrate the central position of the LSTer region during replication arrest and movement of the two sister copies away from the center after duplication. It was not possible to clearly demonstrate movement of the LSTer region to the cell center prior to duplication. Nor, given the size of the LSTer region, was it possible to place this region at a more origin-distal chromosomal site. Regions of the B. subtilis chromosome that fall between the origin and the terminus of the circular map are generally positioned between them in cells (Teleman et al., 1998). The 270⬚ region is halfway between oriC and the terminus. Sister origin regions are localized at approximately the cell quarter positions (D. C.-H. Lin, P. S. Lee, and A. D. G., unpublished data), while the terminus is at approximately midcell (Webb et al., 1997; Teleman et al., 1998; Webb et al., 1998). Thus, the 270⬚ region is usually positioned between the quarter and midcell (Teleman et al., 1998). We visualized the 270⬚ region using a tandem array of lac operators adjacent to cotS (Teleman et al., 1998) combined with our LacICFP fusion. DNA polymerase was again visualized with Tau-YFP. Immediately after synchronization (t ⫽ 0), most of the cells (62%) had a single focus of the 270⬚ region (Figure 5A). Between 60 and 75 min after synchronization, there was a marked increase in the number of cells with two foci of the 270⬚ region (Figure 5A), indicating that in Samples of strain KPL727 [dnaB134(ts), cotS⫹-lacO (270⬚), lacI-cfp, dnaX-yfp] were taken at the indicated times after shiftdown to permissive temperature, and the foci of DNA polymerase (Tau-YFP) and the 270⬚ region (LacI-CFP) were analyzed. (A) The percentage of cells with one focus (squares) or two foci (circles) of the 270⬚ region as a function of time after synchronization is shown. (B) The percentage of cells with the 270⬚ region coincident with (squares) or separate from (circles) a focus of DNA polymerase is shown. Only cells with a single focus of the 270⬚ region were counted. many cells the 270⬚ region was duplicated during this interval. We observed three general patterns when we compared the position of the 270⬚ region to that of DNA polymerase over the time course in cells with a single focus of 270⬚. (1) At all time points, 30%–35% of the cells had the 270⬚ region adjacent to a focus of DNA polymerase (Tau-YFP). This is not surprising, given that 270⬚ is usually positioned between the cell quarter and midcell (Teleman et al., 1998), while the replisome is at or near midcell (Lemon and Grossman, 1998). (2) We also observed cells in which the 270⬚ region was clearly separated from DNA polymerase and (3) cells in which the 270⬚ region was coincident with a focus of DNA polymerase. The percent of cells in which the 270⬚ region was coincident with DNA polymerase increased between 45 and 60 min after synchronization (Figure 5B). This increase occurred just prior to the shift from predominantly one focus of 270⬚ per cell to predominantly two foci of 270⬚ per cell (Figure 5A). The timing of this position change is consistent with movement of the 270⬚ region to the stationary DNA polymerase just prior to duplication. Discussion A Centrally Located, Stationary Bacterial Replisome Our results indicate that during replication in B. subtilis, DNA moves through a stationary replisome located at or near midcell. After a region of DNA is replicated, the two duplicated regions move away from the replisome Molecular Cell 1326 toward opposite ends of the cell. We propose that B. subtilis and probably many bacteria harness the energy released during replication by a stationary replisome to facilitate chromosome movement. In this extrusioncapture model, the centrally located stationary replisome pulls the DNA template in, duplicates it, and releases the two products into opposite halves of the cell. We speculate that once the origin regions are extruded from midcell, they are captured and maintained at approximately cell quarter positions until DNA replication reinitiates. DNA and RNA Polymerases Are Motor Proteins For years, people have thought of DNA polymerase as propelling itself along the DNA template during replication. A stationary DNA polymerase would instead spool the template through itself during replication. Measurements of the force generated by a single, stationary RNA polymerase during transcription (25 picoNewtons) (Gelles and Landick, 1998; Wang et al., 1998) indicate that RNA polymerase is a more powerful motor than either myosin or kinesin (for reviews see Block, 1998; Goldman, 1998; Cook, 1999; Mehta et al., 1999; Vale and Milligan, 2000). A replisome contains two types of nucleoside triphosphate-dependent molecular motors, the DNA polymerase and helicase. Given that the rate of DNA synthesis is approximately ten times faster than that of RNA synthesis, it seems likely that the replisome is at least as powerful a motor as RNA polymerase. Although the replisome has a mass of ⵑ1,000 kDa, the mass of the chromosome dwarfs that of the replisome. A stationary replisome would bind and release and move the chromosome in single base pair increments of ⵑ660 Da and ⵑ0.34 nm (Kornberg and Baker, 1992; Cook, 1999). During chromosome duplication, the same amount of energy is used whether the polymerase is stationary or moving. Given the limits of our understanding of the viscosity and crowding of the intracellular environment, it has been difficult to estimate if spooling the chromosome through the replisome would be more energetically cost effective than moving the replisome along the polymerase. We speculate that an added benefit of the stationary polymerase, combined with rapid recompaction of the DNA after duplication, is the ability to rapidly separate newly duplicated regions, thus limiting the possibility of sister chromosomes tangling in the crowded nucleoid mass. E. coli Probably Has a Stationary Replisome There is increasing evidence that the gram-negative bacterium E. coli also has a centrally located, stationary replisome. In E. coli, newly replicated DNA detected by pulse-labeling with [3H]thymidine, is centrally located in most cells (Koppes et al., 1999). Previously, we speculated (Lemon and Grossman, 1998) that the location of SeqA (Hiraga et al., 1998) also indicates that newly replicated DNA is found at or near midcell in E. coli. SeqA binds both the origin and newly replicated, hemimethylated DNA (Slater et al., 1995). Fluorescence microscopy indicates that the bulk of SeqA does not colocalize with oriC. Rather, SeqA localizes as discrete foci predominantly at midcell or the cell quarters (Hiraga et al., 1998; Onogi et al., 1999) in a pattern similar to that of the B. subtilis replisome (Lemon and Grossman, 1998). The SeqA foci probably represent the majority of SeqA bound to newly duplicated, hemimethylated DNA adjacent to the replication forks. We suspect that the extrusion-capture model for how DNA replication might power, at least in part, chromosome partitioning will apply to other bacteria. Is the Replisome Anchored? While there is no evidence to suggest that the replisome is membrane bound, it seems reasonable to expect it might be anchored in order to prevent movement during DNA replication. In B. subtilis, one of the proteins (DnaB) needed to initiate replication is a membrane protein and might play a role in helping to anchor the replisome. The bidirectional replication forks might also be maintained in place by the viscosity inside the cell. Recent findings indicate that large protein complexes may not diffuse freely throughout the cell (Elowitz et al., 1999). Head-to-Head Replication Forks In vitro data from E. coli replication indicates that the helicase for each fork is loaded onto the origin bubble in a such a way that they either must pass each other or, if stationary, remain head-to-head as each spools and unwinds DNA (Fang et al., 1999). Head-to-head stationary helicases spooling DNA might be envisioned in a manner similar to models for how RuvA and RuvB accomplish Holliday junction branch migration (Rafferty et al., 1996) (see also http://www.sdsc.edu/journals/ mbb/ruva.html). When a model of head-to-head helicases is drawn (Fang et al., 1999), it suggests that the leading and lagging strand DNA polymerase at each fork might use the same template strand (Figure 6). In other words, we suggest that each replication fork duplicates one entire template strand (one fork for Watson and one for Crick) instead of each fork duplicating both strands from half of the chromosome. Head-to-head bidirectional forks might help to facilitate chromosome separation and also help to limit movement of the replisome. The Replisome Is Probably Positioned during Initiation of Replication The simplest model is that DNA replication initiates centrally or at sites that will become central after cell division, and this establishes the position of the replisome. This model is supported by the pattern of origin region localization in both B. subtilis and E. coli which indicates that replication initiates at or near midcell or at positions that will become midcell. In E. coli, the origin region as detected by fluorescence in situ hybridization (FISH) appears to be centrally located in cells just prior to onset of DNA replication (Roos et al., 1999; Niki et al., 2000). B. subtilis origin-region DNA can be labeled with BrdU when spores of a thymine auxotroph are germinated. Initially, the BrdU-labeled origin regions form a single focus near midcell (Lewis and Errington, 1997). Later, cells have two well-separated origin regions. When the B. subtilis origin region is labeled with the lacO-LacIGFP system, the origin is centrally located in cells with a single focus (D. C.-H. Lin and A. D. G., unpublished data). Spo0J, which is an excellent marker of origin region position in B. subtilis (Lewis and Errington, 1997; Movement of DNA through a Stationary Replisome 1327 2000; Sawitzke and Austin, 2000). SMC may be loaded onto newly replicated DNA to establish these constrained (⫹) supercoils and compact the DNA. In addition to SMC, Spo0J, a member of the ParB protein family, is required for efficient partitioning (Ireton et al., 1994), probably by functioning to organize the origin region into a higher order structure (Glaser et al., 1997; Lin et al., 1997; Britton et al., 1998; Lin and Grossman, 1998; Britton and Grossman, 1999). Figure 6. A Model of Two Head-to-Head, Opposing, Stationary Hexameric Helicases Spooling the DNA Template through as They Unwind the Double Helix Adapted from Fang et al. (1999). Lin et al., 1997; Teleman et al., 1998)), binds to eight identified parS sites located in the origin-proximal 20% of the chromosome (Lin and Grossman, 1998). During exponential growth, most cells have two well-separated foci of Spo0J. In cells with a single focus, Spo0J, and hence the origin region, is predominantly at midcell (Teleman et al., 1998; P. S. Lee and A. D. G., unpublished data). Most cells in which the initiation of DNA replication is inhibited and old rounds have finished have a single focus of Spo0J-GFP located at approximately midcell (our unpublished data). Finally, our results with replication upon release from stringent arrest (Figures 3J–3O) indicate that during multifork replication initiation occurs at the cell quarters, positions that will become midcell upon division. Other Proteins Are Required for Efficient Chromosome Partitioning Although we have proposed that the replisome plays an important role in chromosome partitioning, it is certainly not alone. It is likely that proteins involved in chromosome organization, compaction, and supercoiling contribute to the orderly movement of sister DNA regions away from the central replisome. We speculate that SMC, which is clearly important for chromosome partitioning (Britton et al., 1998; Graumann et al., 1998; Moriya et al., 1998; Britton and Grossman, 1999; Hirano, 1999), may play such a role. It has recently been proposed that SMC and the E. coli analog MukB generate higher order compaction of bacterial DNA by introducing constrained positive supercoils (Holmes and Cozzarelli, The Common Theme of Stationary Replisomes A diverse array of organisms including prokaryotes, archaea, and eukaryotes may have stationary replisomes. Many of the components required for DNA replication are functionally conserved and homologous in all organisms (Baker and Bell, 1998). The term “replication factory” was first applied to the multiple foci of replication proteins observed in the eukaryotic nucleus using immunofluorescence microscopy (for review see Newport and Yan, 1996; Cook, 1999). The prevailing view is that these factories represent a clustering of many replicons that probably initiate replication synchronously during S phase (Newport and Yan, 1996). Recently, a fusion of GFP to the eukaryotic sliding clamp, GFP-PCNA, has been observed using time-lapse microscopy in a stable tissue culture cell line (Leonhardt et al., 2000). Replication foci (GFP-PCNA) assembled and disassembled during S phase but did not show directional motion (Leonhardt et al., 2000). This observation is consistent with earlier models suggesting that these eukaryotic “replication factories” might represent a conglomerate of stationary replisomes through which the DNA template moves (Hozak et al., 1993). Most eukaryotes use a mitotic spindle and mitotic motors for the separation of sister chromosomes. These components involved in chromosome partitioning have not been found in prokaryotes. Perhaps the differences in the distances needed to move chromosomes during partitioning partly explain the dramatically different mechanisms used by eukaryotes and prokaryotes. Experimental Procedures Growth Media and Antibiotics Rich medium was Luria-Bertani broth (LB), and defined minimal medium was S7 medium with 1% glucose and 0.1% glutamate (Vasantha and Freese, 1980; Jaacks et al., 1989) and required amino acids (phenylalanine [40 g/ml], tryptophan [40 g/ml], and threonine [240 g/ml]). Where needed, antibiotics were used at the indicated concentrations: chloramphenicol (cat), 5 g/ml; erythromycin, 0.5 g/ml and lincomycin, 12.5 g/ml together (mls); and spectinomycin (spc), 100 g/ml. Strains and Alleles All strains are derivatives of JH642, which is trpC2 pheA1 (Perego et al., 1988). Relevant alleles that we constructed for this work are described in the appropriate sections below. dnaB134, a temperature sensitive mutation that prevents initiation of replication at nonpermissive temperature (Levine et al., 1991, 1995; Autret et al., 1999), was used to synchronize the replication cycle in a population of cells (see below). cotS⫹:pAT25 (cotS⫹-lacO cat) is an array of Lac operators (on pAT25) integrated by single crossover into cotS in the 270⬚ region of the chromosome (Teleman et al., 1998). Construction of B. subtilis strains by transformation was by standard procedures (Harwood and Cutting, 1990). Strains (and relevant genotypes) used in experiments include KPL502 [dnaB134(ts), zhb83::Tn917(mls) (linked by transformation to dnaB), dnaX- Molecular Cell 1328 gfp(mut2) spc]; KPL549 [dnaB134(ts), hutM⫹-lacO cat, thrC::(PpenlacI⌬11-gfp(mut2) mls), polC-myc spc]; KPL727 [dnaB134(ts), cotS⫹-lacO cat (270⬚), thrC::(Ppen-lacI⌬11-cfp(W7) mls), dnaX-yfp(mut2) spc]; and KPL730 [dnaB134(ts), hutM⫹-lacO cat, thrC::(PpenlacI⌬11-cfp(W7) mls), dnaX-yfp(mut2) spc]. Lac Operator/Lac Repressor System for Visualizing Regions of Chromosomal DNA The Lac repressor/lac operator system for visualizing chromosome regions was first developed for budding yeast and Chinese hamster ovary cells (Robinett et al., 1996; Straight et al., 1996; Belmont and Straight, 1998) and was adapted for use in B. subtilis and E. coli (Gordon et al., 1997; Webb et al., 1997). To mark the LSTer region for visualization, we inserted a tandem array of lac operator sites next to hutM. pAT12 contains a tandem array of lac operators and a chloramphenicol resistance (cat) cassette (Webb et al., 1997). A 3⬘ fragment of hutM extending past the stop codon was PCR amplified using primers oKPL148 and 149, which added a 5⬘ AatII site and a 3⬘ BamHI site. The hutM fragment was digested with AatII and BamHI and cloned between the AatII and BamHI sites of pAT12 to give pKL167. pKL167 was integrated by single crossover onto the B. subtilis chromosome by standard methods (Harwood and Cutting, 1990). The resulting strain has a tandem array of lac operators downstream of an intact hutM. To mark the 270⬚ region, a tandem array of lac operators (lacO) inserted by single crossover at cotS (Webb et al., 1997) was transformed into JH642 using chromosomal DNA from AT52 (Teleman et al., 1998). We found that a carboxy-terminal fusion of LacI (minus the last 11 codons to remove the tetramerization ability) to GFP, or its variants, produces a signal that is significantly brighter during exponential growth than the previously described amino-terminal fusion protein (GFP-LacI) (Robinett et al., 1996; Webb et al., 1997). Here we describe the construction of both LacI-GFP and LacI-CFP. The 3⬘ end of lacI, absent the last 11 codons, was PCR amplified from pDH88 (Henner, 1990) using primers (oKPL 134 and 135) that added an EcoRI site at the 5⬘ end and an XhoI site at the 3⬘ end. The resultant product was then fused in-frame to the 5⬘ end of gfpmut2 with a five-codon linker (LEGSG) to give plasmid pKL153. pKL153 was integrated into strain KPL417 by single crossover into a copy of lacI (driven by the Ppen promoter; Yansura and Henner, 1984) that had been introduced into the B. subtilis chromosome at the thrC locus with pPL97 (P. Levin, J. Quisel, F. Aragoni, and A. D. G., unpublished data) to give strain KPL420. The resulting Ppen-lacI(⌬11)gfpmut2 expression unit was PCR amplified using primers (oKPL145 and 147) that added EcoRI and BamHI to the 5⬘ and 3⬘ ends, respectively, and subcloned into pDG795 (provided by P. Stragier) (Ireton et al., 1994) to give pKL160 and into pUC9 to give pKL178. pKL160 was integrated by double crossover at thrC on the B. subtilis chromosome, resulting in strain KPL471 that expresses LacI-GFP. A cyan variant of gfp, cfpW7 (Heim and Tsien, 1996), was swapped for the gfpmut2 of pKL178 using the XhoI and SphI sites yielding pKL189. pKL189 was digested with EcoRI and BamHI, and the Ppen-lacIcfpW7 fragment was inserted into pDG795 to give pKL190. pKL190 was integrated by double crossover at thrC on the B. subtilis chromosome, resulting in strain KPL682 that expresses LacI-CFP. We found that in B. subtilis, the W7 CFP variant (Heim and Tsien, 1996) with the normal jellyfish codon usage produced a brighter fusion protein than the ECFP variant that is optimized for mammalian codon usage. Tau-YFP We constructed yfpmut2 by site-directed mutagenesis of gfpmut2 (S65A, V68L, S72A) (Cormack et al., 1996), changing amino acid 203 from threonine to tyrosine (Ormo et al., 1996). The yfpmut2 gene was then subcloned in-frame behind a 3⬘ fragment of dnaX (Tau), replacing gfpmut2 in pKL147 (Lemon and Grossman, 1998). The resulting construct, pKL183, was integrated by single crossover onto the B. subtilis chromosome, such that the fusion is the only functional copy of the gene and is under control of the endogenous promoter. Stringent Response Experiments Stringent response was induced similarly to previous descriptions (Levine et al., 1991, 1995; Autret et al., 1999). All strains were temper- ature-sensitive mutants (dnaB134) (Mendelson and Gross, 1967; Karamata and Gross, 1970) for initiation of DNA replication. Cells were grown at 30⬚C in defined minimal medium. To synchronize the cells for DNA replication, an exponentially growing culture was shifted to 45⬚C for 30 min (single-labeled strains) or 42⬚C for 45 min (double-labeled strains). Arginine hydroxymate (250–500 g/ml) (Sigma) was prepared and added immediately prior to shift back to 30⬚C. For experiments with the double-labeled strain, the culture was split into two flasks just before shift back to 30⬚C, and arginine hydroxymate was added to one but not the other. To release cells from stringent response, cells were removed from medium with arginine hydroxymate by pouring the culture onto a sterile 0.2 m Nalgene bottle top filter. Cells were washed with 2–3 volumes of medium and resuspended in fresh growth medium plus the following additions: arginine (600 g/ml), a standard mix of all 20 amino acids (Harwood and Cutting, 1990), adenosine (20 g/ml), guanosine (20 g/ml), thymidine (50 g/ml), and cytidine (50 g/ml). All data presented in the tables and figures are cumulative from at least two experiments, with each experiment giving similar results. Live Cell Microscopy Slides of live cells were prepared essentially as described (Lemon and Grossman, 1998). Briefly, an aliquot of cells was removed from an exponentially growing culture and stained with the vital membrane stain FM4-64 (140–280 ng/ml) (Molecular Probes) prior to placement on a pad of 0.5%–1% agarose in a solution of 1⫻ T⬘ base, 1 mM MgSO4 (Harwood and Cutting, 1990). The following filter sets were used: a Cy3 filter set for FM4-64, Chroma filter set 41029 for YFP, and Chroma filter set 31044 for CFP. Images were acquired using a Nikon E800 microscope equipped with a CCD camera (Hamamatsu, model C4742-95) and Improvision OpenLab software. Images were colorized and merged using Improvision OpenLab software and cropped in Adobe Photoshop 3.05 or 5.5. Acknowledgments We thank A. Teleman, C. Webb, and R. Losick for providing the lacO array plasmid, the cotS::lacO strain, and advice on their use. We thank R. Tsien for the W7 variant of cfp. We thank P. Levin, J. Quisel, and F. Aragoni for providing pPL97. We thank J. C. Lindow, T. A. Baker, F. Solomon, S. Bell, R. Losick, and K. Bacon Schneider for comments on the manuscript. This work was supported by Public Health Services grant GM41934 and by the Merck/MIT Collaborative Program. Received August 2, 2000; revised October 4, 2000. References Autret, S., Levine, A., Vannier, F., Fujita, Y., and Seror, S.J. (1999). The replication checkpoint control in Bacillus subtilis: identification of a novel RTP-binding sequence essential for the replication fork arrest after induction of the stringent response. Mol. Microbiol. 31, 1665–1679. Baker, T.A., and Bell, S.P. (1998). Polymerases and the replisome: machines within machines. Cell 92, 295–305. Belmont, A.S., and Straight, A.F. (1998). In vivo visualization of chromosomes using lac operator-repressor binding. Trends Cell Biol. 8, 121–124. Block, S.M. (1998). Kinesin: what gives? Cell 93, 5–8. Britton, R.A., and Grossman, A.D. (1999). Synthetic lethal phenotypes caused by mutations affecting chromosome partitioning in Bacillus subtilis. J. Bacteriol. 181, 5860–5864. Britton, R.A., Lin, D.C., and Grossman, A.D. (1998). Characterization of a prokaryotic SMC protein involved in chromosome partitioning. Genes Dev. 12, 1254–1259. Cashel, M., Gentry, D.R., Hernandez, V.J., and Vinella, D. (1996). Washington, D.C.: Am. Soc. Microbiol., 1458–1496. Cook, P.R. (1999). The organization of replication and transcription. Science 284, 1790–1795. Cormack, B.P., Valdivia, R.H., and Falkow, S. (1996). FACS-opti- Movement of DNA through a Stationary Replisome 1329 mized mutants of the green fluorescent protein (GFP). Gene 173, 33–38. Dworkin, M., Higgins, J., Glenn, A., and Mandelstam, J. (1972). Synchronization of the growth of Bacillus subtilis and its effect on sporulation. In Spores V, H.O. Halvorson, R. Hanson, and L.L. Campbell, eds. (Washington D.C.: ASM Press), pp. 233–237. vedo, V., Bertero, M.G., Bessieres, P., Bolotin, A., Borchert, S., et al. (1997). The complete genome sequence of the gram-positive bacterium Bacillus subtilis. Nature 390, 249–256. Lemon, K.P., and Grossman, A.D. (1998). Localization of bacterial DNA polymerase: evidence for a factory model of replication. Science 282, 1516–1519. Elowitz, M.B., Surette, M.G., Wolf, P.E., Stock, J.B., and Leibler, S. (1999). Protein mobility in the cytoplasm of Escherichia coli. J. Bacteriol. 181, 197–203. Leonhardt, H., Rahn, H.P., Weinzierl, P., Sporbert, A., Cremer, T., Zink, D., and Cardoso, M.C. (2000). Dynamics of DNA replication factories in living cells. J. Cell Biol. 149, 271–280. Fang, L., Davey, M.J., and O’Donnell, M. (1999). Replisome assembly at oriC, the replication origin of E. coli, reveals an explanation for initiation sites outside an origin. Mol. Cell 4, 541–553. Levine, A., Vannier, F., Dehbi, M., Henckes, G., and Seror, S.J. (1991). The stringent response blocks DNA replication outside the ori region in Bacillus subtilis and at the origin in Escherichia coli. J. Mol. Biol. 219, 605–613. Gelles, J., and Landick, R. (1998). RNA polymerase as a molecular motor. Cell 93, 13–16. Glaser, P., Sharpe, M.E., Raether, B., Perego, M., Ohlsen, K., and Errington, J. (1997). Dynamic, mitotic-like behavior of a bacterial protein required for accurate chromosome partitioning. Genes Dev. 11, 1160–1168. Goldman, Y.E. (1998). Wag the tail: structural dynamics of actomysin. Cell 93, 1–4. Gordon, G.S., Sitnikov, D., Webb, C.D., Teleman, A., Straight, A., Losick, R., Murray, A.W., and Wright, A. (1997). Chromosome and low copy plasmid segregation in E. coli: visual evidence for distinct mechanisms. Cell 90, 1113–1121. Graumann, P.L., Losick, R., and Strunnikov, A.V. (1998). Subcellular localization of Bacillus subtilis SMC, a protein involved in chromosome condensation and segregation. J. Bacteriol. 180, 5749–5755. Harwood, C.R., and Cutting, S.M. (1990). Molecular Biological Methods for Bacillus. In eds. (Chichester, England: John Wiley & Sons), pp. Heim, R., and Tsien, R.Y. (1996). Engineering green fluorescent protein for improved brightness, longer wavelengths and fluorescence resonance energy transfer. Curr. Biol. 6, 178–182. Henner, D.J. (1990). Inducible expression of regulatory genes in Bacillus subtilis. Methods Enzymol. 185, 223–228. Hiraga, S., Ichinose, C., Niki, H., and Yamazoe, M. (1998). Cell cycledependent duplication and bidirectional migration of SeqA-associated DNA-protein complexes in E. coli. Mol. Cell 1, 381–387. Hirano, T. (1999). SMC-mediated chromosome mechanics: a conserved scheme from bacteria to vertebrates? Genes Dev. 13, 11–19. Holmes, V.F., and Cozzarelli, N.R. (2000). Closing the ring: links between SMC proteins and chromosome partitioning, condensation, and supercoiling. Proc. Natl. Acad. Sci. USA 97, 1322–1324. Hozak, P., Hassan, A.B., Jackson, D.A., and Cook, P.R. (1993). Visualization of replication factories attached to nucleoskeleton. Cell 73, 361–373. Ireton, K., Gunther, N.W., IV, and Grossman, A.D. (1994). spo0J is required for normal chromosome segregation as well as the initiation of sporulation in Bacillus subtilis. J. Bacteriol. 176, 5320–5329. Jaacks, K.J., Healy, J., Losick, R., and Grossman, A.D. (1989). Identification and characterization of genes controlled by the sporulation regulatory gene spo0H in Bacillus subtilis. J. Bacteriol. 171, 4121– 4129. Jensen, R.B., and Shapiro, L. (1999). The Caulobacter crescentus smc gene is required for cell cycle progression and chromosome segregation. Proc. Natl. Acad. Sci. USA 96, 10661–10666. Karamata, D., and Gross, J.D. (1970). Isolation and genetic analysis of temperature-sensitive mutants of B. subtilis defective in DNA synthesis. Mol. Gen. Genet. 108, 277–287. Kelman, Z., and O’Donnell, M. (1995). DNA polymerase III holoenzyme: structure and function of a chromosomal replicating machine. Annu. Rev. Biochem. 64, 171–200. Koppes, L.J., Woldringh, C.L., and Nanninga, N. (1999). Escherichia coli contains a DNA replication compartment in the cell center. Biochimie 81, 803–810. Kornberg, A., and Baker, T.A. (1992). DNA Replication, Second Edition (San Francisco, CA: W.H. Freeman and Co.). Kunst, F., Ogasawara, N., Moszer, I., Albertini, A.M., Alloni, G., Aze- Levine, A., Autret, S., and Seror, S.J. (1995). A checkpoint involving RTP, the replication terminator protein, arrests replication downstream of the origin during the Stringent Response in Bacillus subtilis. Mol. Microbiol. 15, 287–295. Lewis, P.J., and Errington, J. (1997). Direct evidence for active segregation of oriC regions of the Bacillus subtilis chromosome and colocalization with the Spo0J partitioning protein. Mol. Microbiol. 25, 945–954. Lin, D.C., and Grossman, A.D. (1998). Identification and characterization of a bacterial chromosome partitioning site. Cell 92, 675–685. Lin, D.C.H., Levin, P.A., and Grossman, A.D. (1997). Bipolar localization of a chromosome partition protein in Bacillus subtilis. Proc. Natl. Acad. Sci. USA 94, 4721–4726. Mehta, A.D., Rief, M., Spudich, J.A., Smith, D.A., and Simmons, R.M. (1999). Single-molecule biomechanics with optical methods. Science 283, 1689–1695. Mendelson, N.H., and Gross, J.D. (1967). Characterization of a temperature sensitive mutant of Bacillus subtilis defective in deoxyribonucleic acid replication. J. Bacteriol. 94, 1603–1608. Miller, D.M., III, Desai, N.S., Hardin, D.C., Piston, D.W., Patterson, G.H., Fleenor, J., Xu, S., and Fire, A. (1999). Two-color GFP expression system for C. elegans. Biotechniques 26, 914–918, 920–911. Mohl, D.A., and Gober, J.W. (1997). Cell cycle-dependent polar localization of chromosome partitioning proteins in Caulobacter crescentus. Cell 88, 675–684. Moriya, S., Tsujikawa, E., Hassan, A.K., Asai, K., Kodama, T., and Ogasawara, N. (1998). A Bacillus subtilis gene-encoding protein homologous to eukaryotic SMC motor protein is necessary for chromosome partition. Mol. Microbiol. 29, 179–187. Newport, J., and Yan, H. (1996). Organization of DNA into foci during replication. Curr. Opin. Cell Biol. 8, 365–368. Niki, H., and Hiraga, S. (1998). Polar localization of the replication origin and terminus in Escherichia coli nucleoids during chromosome partitioning. Genes Dev. 12, 1036–1045. Niki, H., Yamaichi, Y., and Hiraga, S. (2000). Dynamic organization of chromosomal DNA in Escherichia coli. Genes Dev. 14, 212–223. Onogi, T., Niki, H., Yamazoe, M., and Hiraga, S. (1999). The assembly and migration of SeqA-Gfp fusion in living cells of Escherichia coli. Mol. Microbiol. 31, 1775–1782. Ormo, M., Cubitt, A.B., Kallio, K., Gross, L.A., Tsien, R.Y., and Remington, S.J. (1996). Crystal structure of the Aequorea victoria green fluorescent protein. Science 273, 1392–1395. Perego, M., Spiegelman, G.B., and Hoch, J.A. (1988). Structure of the gene for the transition state regulator, abrB: regulator synthesis is controlled by the spo0A sporulation gene in Bacillus subtilis. Mol. Microbiol. 2, 689–699. Rafferty, J.B., Sedelnikova, S.E., Hargreaves, D., Artymiuk, P.J., Baker, P.J., Sharples, G.J., Mahdi, A.A., Lloyd, R.G., and Rice, D.W. (1996). Crystal structure of DNA recombination protein RuvA and a model for its binding to the Holliday junction. Science 274, 415–421. Robinett, C.C., Straight, A., Li, G., Willhelm, C., Sudlow, G., Murray, A., and Belmont, A.S. (1996). In vivo localization of DNA sequences and visualization of large-scale chromatin organization using lac operator/repressor recognition. J. Cell Biol. 135, 1685–1700. Roos, M., van Geel, A.B., Aarsman, M.E., Veuskens, J.T., Woldringh, C.L., and Nanninga, N. (1999). Cellular localization of oriC during Molecular Cell 1330 the cell cycle of Escherichia coli as analyzed by fluorescent in situ hybridization. Biochimie 81, 797–802. Sawitzke, J.A., and Austin, S. (2000). Suppression of chromosome segregation defects of Escherichia coli muk mutants by mutations in topoisomerase I. Proc. Natl. Acad. Sci. USA 97, 1671–1676. Sharpe, M.E., and Errington, J. (1998). A fixed distance for separation of newly replicated copies of oriC in Bacillus subtilis: implications for co-ordination of chromosome segregation and cell division. Mol. Microbiol. 28, 981–990. Slater, S., Wold, S., Lu, M., Boye, E., Skarstad, K., and Kleckner, N. (1995). E. coli SeqA protein binds oriC in two different methylmodulated reactions appropriate to its roles in DNA replication initiation and origin sequestration. Cell 82, 927–936. Straight, A.F., Belmont, A.S., Robinett, C.C., and Murray, A.W. (1996). GFP tagging of budding yeast chromosomes reveals that proteinprotein interactions can mediate sister chromatid cohesion. Curr. Biol. 6, 1599–1608. Teleman, A.A., Graumann, P.L., Lin, D.C.H., Grossman, A.D., and Losick, R. (1998). Chromosome arrangement within a bacterium. Curr. Biol. 8, 1102–1109. Trun, N.J., and Marko, J.F. (1998). Architecture of the a bacterial chromosome. ASM News 64, 276–283. Tsien, R.Y. (1998). The green fluorescent protein. Annu. Rev. Biochem. 67, 509–544. Vale, R.D., and Milligan, R.A. (2000). The way things move: looking under the hood of molecular motor proteins. Science 288, 88–95. Vasantha, N., and Freese, E. (1980). Enzyme changes during Bacillus subtilis sporulation caused by deprivation of guanine nucleotides. J. Bacteriol. 144, 1119–1125. Wang, M.D., Schnitzer, M.J., Yin, H., Landick, R., Gelles, J., and Block, S.M. (1998). Force and velocity measured for single molecules of RNA polymerase. Science 282, 902–907. Webb, C.D., Teleman, A., Gordon, S., Straight, A., Belmont, A., Lin, D.C., Grossman, A.D., Wright, A., and Losick, R. (1997). Bipolar localization of the replication origin regions of chromosomes in vegetative and sporulating cells of B. subtilis. Cell 88, 667–674. Webb, C.D., Graumann, P.L., Kahana, J.A., Teleman, A.A., Silver, P.A., and Losick, R. (1998). Use of time-lapse microscopy to visualize rapid movement of the replication origin region of the chromosome during the cell cycle in Bacillus subtilis. Mol. Microbiol. 28, 883–892. Wendrich, T.M., and Marahiel, M.A. (1997). Cloning and characterization of a relA/spoT homologue from Bacillus subtilis. Mol. Microbiol. 26, 65–79. Winston, S., and Sueoka, N. (1980). DNA-membrane association is necessary for initiation of chromosomal and plasmid replication in Bacillus subtilis. Proc. Natl. Acad. Sci. USA 77, 2834–2838. Yansura, D.G., and Henner, D.J. (1984). Use of the Escherichia coli lac repressor and operator to control gene expression in Bacillus subtilis. Proc. Natl. Acad. Sci. USA 81, 439–443.